To identify predictors associated with success of a Nutrition Support Team (NST) for the elderly.
A total of 101 patients who received NST intervention in 2015 were divided into two groups by NST outcome. Patients who achieved therapeutic targets were classified in the success group. Patients who discontinued treatment because of worsening condition or death were classified in the non success group. We assessed patient characteristics, laboratory data, and nutritional support methods. Prior to this study, we retrospectively extracted NST outcome predictors for patients treated in 2014, and identified cutoff values of quantitative variables using receiver operating characteristic analysis. The extracted predictors were as follows: % total energy expenditure (66.0%), albumin (2.4 g/dL), total lymphocyte count (1,195/µL), C-reactive protein level (2.00 mg/dL), transthyretin level (8.5 mg/dL), Controlling Nutritional Status (CONUT) score (8), and the presence of Peripheral Parenteral Nutrition (PPN). We performed univariate and multivariate logistic regression analysis to identify the factors relevant to NST outcome.
We conducted the multivariate logistic regression analysis adjusted for age, sex, the presence/absence of PPN, and CONUT. A CONUT score ≥ 8 was an independent risk factor for NST outcome (Odds Ratio [OR]: 3.72, 95% Confidence Interval [CI]: 1.26-10.95). Moreover, the presence of PPN tended to be associated with NST outcome (OR: 2.63, 95% CI: 0.96-7.20).
To increase the success rate of NST for elderly, it is important to identify the elderly at risk of non success NST outcome.
Nutrition support team, Outcome, Elderly, Nutritional assessment, Malnutrition
Malnutrition in the elderly is a risk factor for poor clinical outcomes, negatively affecting the rate of disease remission or cure, and is associated with an increase in complications, mortality rate, and length of hospital stay [1-5]. The incidence of malnutrition in the elderly is high [6-11]. When elderly patients experiencing illness at home or in the nursing home are admitted to a higher function hospital in an urban area, it is difficult to improve their nutritional status to a healthy threshold, because disease status, as well as malnutrition, are more difficult to improve in the elderly than in the young [12,13]. Considering that the world population is aging [14], it is anticipated that there will be a large scale challenge in effectively improving malnutrition in the elderly.
The concept of a Nutrition Support Team (NST) originated in the USA with the development and spread of total parenteral nutrition by Dudrick in 1968 [15-17]; NST thereafter spread worldwide, including to Europe and North America. Since its inception, many studies have been performed to evaluate the effects of NST [18-22]; these have revealed several positive findings about its efficacy; for example, it has had an impact on performing the appropriate nutrition assessment and therapy, improvement in patient’s nutrition status, decrease in the complications caused by enteral nutrition or parenteral nutrition, shortening hospitalization periods, and saving medical expenses. However, factors associated with NST outcome have not been documented.
This study was conducted at a higher function hospital, which admits serious patients who require hospitalization or surgery in Suminoe-ku, an area with a population of approximately 125,000 in the southwestern part of Osaka city. In this hospital, malnourished patients receive intensive nutritional care from an NST; the average age of patients served by NST is above 80 years. Based on the experience of this hospital in operating NST, we aimed to investigate predictors for success or non-success of NST in order to increase the efficiency of NST therapy for the elderly.
Therefore, the purpose of this study was to identify NST outcome predictors for the elderly and verify the validity of predictors.
Registered dietitians in the hospital identified patients who met two or more of the factors (1)-(3); (1) Dietary intake less than 50%, (2) Albumin ≤ 2.7 g/dL, (3) Body mass index ≤ 18.5 kg/m2. Nurse and the bedsore commission identified patients who were eligible for NST therapy. Terminal stage patients who were not clinically indicated for NST intervention as determined by a primary care provider were excluded. Furthermore, patients clinically indicated for NST intervention as determined by a primary care doctor were included. These groups composed the total cohort of possible study subjects.
Herein, 178 patients who received care from an NST in this hospital in, Osaka City, Japan, from January 2015 to December 2015, were enrolled. After excluding patients who ended NST treatment because of changing hospital or hospital discharge for social reasons, before receiving a final NST evaluation (73 patients), and those who were younger than sixty-five years (4 patients), 101 patients were enrolled in the study. Next, the patients were divided into two groups by NST outcome, the success group and non-success group (Figure 1).
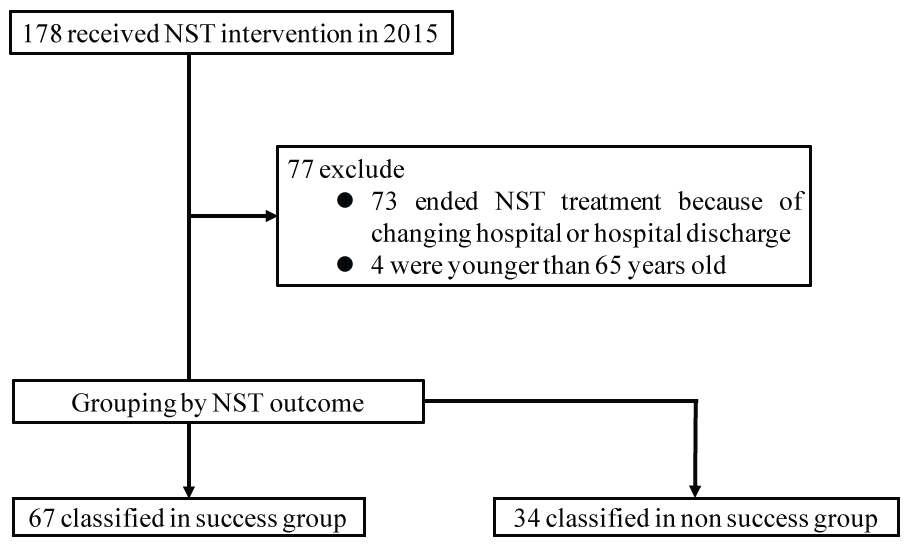 Figure 1: Flow chart of the subjects in this study. View Figure 1
Figure 1: Flow chart of the subjects in this study. View Figure 1
This study was conducted in accordance with the Declaration of Helsinki and was approved by the ethics committee of Osaka City University.
In this hospital, the therapeutic target suitable for each patient was selected based on the five main therapeutic targets at the time of NST intervention. An example of the therapeutic targets are TEE is filled by oral intake, Alb ≥ 2.7 g/dL, amount of oral intake ≥ 50%, TEE is filled by PEG, and body weight increase.
NST outcome was determined as below. Patients who achieved the therapeutic target were classified in the success group. In addition, patients whose nutrition status improved to a level at which they did not need further intensive nutritional care by an NST was classified in a success. Patients who discontinued treatment from NST because of worsening condition or death were classified into the non success group.
We analyzed clinical data, including sex, age, Body Mass Index (BMI; kg/m2), Charlson Co-morbidity Index (CCI; point) [23], Subjective Global Assessment (SGA; point), the presence/absence of dysphagia (presence/absence), and the energy fill-rate to total energy expenditure (%TEE; %).
BMI was calculated based on patient’s height and weight. Type and level of severity of co-morbidities in subjects were evaluated by CCI. We classified subjects into four groups by CCI, low (0 points), medium (1-2 points), high (3-4 points), and very high (≥ 5 points). SGA in this hospital was scored as presented in Table 1. The higher the total score, the higher the risk of nutritional disorder. We classified subjects with an SGA of 0 points as normal nutritional condition, 1-2 points as light malnutrition, 3-4 points as moderate malnutrition, and 5-10 points as severe malnutrition. %TEE was calculated by using the following formula: %TEE = {total energy intakes [kcal]/(basal metabolic rate [kcal] × activity factor × stress factor)} × 100. Basal metabolic rate was calculated by using the Simple expression for Japanese [24]; man: 14.1 × body weight [kg] + 620, women: 10.8 × body weight [kg] + 620.
Table 1: Subjective global assessment metrics. View Table 1
We measured the serum Albumin (Alb; g/dL), Total Lymphocyte Count (TLC; /μL), Total Cholesterol (T-cho; mg/dL), C-Reactive Protein (CRP; mg/dL), Transthyretin (TTR; mg/dL), Hemoglobin (Hb; g/dL), and Triglyceride (TG; mg/dL).
We calculated the Controlling Nutritional Status (CONUT) score [25,26] by using the values of Alb, TLC, and T-cho, which are used to calculate a total score representing nutritional status. In general, the level of under nutrition for a patient was classified with a CONUT score of 0-1 as normal, 2-4 as light, 5-8 as moderate, and 9-12 as severe; however, we treated CONUT score as the quantitative variable and did not use it to assess the level of under nutrition in subjects.
Method of supplying nutrition were classified within seven groups, Total Parenteral Nutrition (TPN), Peripheral Parenteral Nutrition (PPN), nasal feeding, Percutaneous Endoscopic Gastrostomy (PEG), diet (including therapeutic diet), oral supplementation, and rehabilitation for oral intake (diet for dysphagia patient). Oral supplementation included, for example, jelly, juice, ice cream rich in nutrients (calorie or protein or trace element); protein modified rice cracker or cookie; yogurt; or enteral nutrient. We documented the method of supplying nutrition for each patient at the time of NST intervention.
In preparation for this study, we retrospectively extracted data on factors for patients treated in 2014, to predict NST outcome and identify the cut-off values of extracted quantitative variable by Receiver Operating Characteristic (ROC) analysis. The seven factors, including %TEE, the presence/absence of PPN, Alb, TLC, CRP, TTR and CONUT, were significantly different between the success group and non-success group. ROC analysis was used to obtain the cut off values of quantitative variables other than the presence/absence of PPN, Area Under the Curve (AUC), and 95% Confidence Interval (95% CI). The cutoff values (AUC, 95% CI) of each factor were as follows: %TEE, 66.0% (0.63, 0.51-0.74); Alb, 2.4 g/dL (0.72, 0.61-0.82); TLC, 1195/µL (0.68, 0.57-0.80); CRP, 2.00 mg/dL (0.68, 0.57-0.79); TTR, 8.5 mg/dL (0.73, 0.60-0.87); and CONUT score, 8 (0.67, 0.55-0.79).
Data were expressed as the median (25th-75th percentile) or as the number of patients (percentage). Among the success group and non-success group, quantitative variables were compared by the Mann-Whitney U test, and categorical variables were compared by the Fisher’s exact test or the Chi-square test.
Univariate and multivariate logistic regression analysis were used to identify the relevant factors for NST outcome and to obtain the crude and adjusted Odds Ratio (OR) and 95% CI. Age, sex, BMI, CCI, %TEE, the presence/absence of PPN, Alb, TLC, CRP, TTR, and CONUT were chosen as potential factors. Quantitative variables were divided into two groups. Age, BMI, and CCI were divided as follows: age ≥ 85 or < 85 years, BMI ≥ 18.5 or < 18.5 kg/m2, and CCI ≥ 3 or < 3 points. %TEE, Alb, TLC, CRP, TTR, and CONUT were divided by using the cut off value. In the multivariate logistic regression analysis, the 4 factors, including age, sex, the presence/absence of PPN, and CONUT, were selected based on the results of univariate logistic regression analysis.
IBM® SPSS® Statistics 23 software (IBM Japan, Tokyo, Japan) was used for analysis, except for the univariate and multivariate logistic regression analysis, which was performed using SAS 9.4 (SAS Institute Inc., Cary, NC, USA). A p-value of less than 0.05 was considered indicative of statistical significance.
The characteristics of subjects at the time of NST intervention are showed in Table 2. No significant difference was seen among the groups. The median age of the success group was 85.0 years and that of the non success group was 84.5 years. Both groups were categorized as underweight (the median value of success group was 18.2 kg/m2, non-success group was 17.5 kg/m2).
Table 2: Characteristic of subjects. View Table 2
The results of laboratory evaluations at the time of NST intervention are shown in Table 3. The values of Alb, T-cho, and TTR in the success group were significantly higher than those in the non success group. The values of CRP and CONUT in the success group were significantly lower than those in the non success group. The values of TLC, Hb, and TG were not significantly different among the groups.
Table 3: Laboratory evaluations of patients. View Table 3
The methodology used to supply nutrition at the time of NST interventions are summarized in Table 4. Although no significant difference was seen regarding methods, the rates of absence PPN and presence diet in the success group tended to be high. Nasal feeding was not used to supply nutrition for any patients.
Table 4: Prevalence of methods used to supply nutrition at time of NST intervention. View Table 4
Table 5 shows the results of univariate and multivariate logistic regression analysis, which were used to determine the factors associated with NST outcome. Unadjusted univariate logistic regression analysis suggested that 2 factors, including CRP ≥ 2.00 and CONUT ≥ 8, were significantly associated with NST outcome, while male, the presence of PPN and TTR < 8.5 tended to be related to NST outcome. In the multivariate logistic regression analysis, adjusted for sex, age, PPN, and CONUT, a CONUT score ≥ 8 was found to be an independent risk factor for NST outcome (OR: 3.72, 95% CI: 1.26-10.95). Moreover, the presence of PPN tended to be associated with NST outcome (OR: 2.63, 95% CI: 0.96-7.20). CRP ≥ 2.00 was significantly associated with NST outcome in univariate analysis; however, it was excluded from the factors used in multivariate logistic regression analysis in order to avoid co-linearity. Sex, age, BMI, CCI, %TEE, Alb, and TLC were not associated with NST outcome.
Table 5: Result of univariate and multivariate logistic regression analysis for NST outcome. View Table 5
In this study, at the time of NST intervention, a CONUT score of ≥ 8 was negatively associated with NST outcome in the elderly and the presence of PPN had the second most negative association. In a higher function hospital, which admits serious patients who require hospitalization or surgery in an urban area, if the elderly patient with a CONUT score of ≤ 7 or the absence of PPN present at the time of NST intervention, short term NST participation could improve their nutritional status. In our study, 66% of the elderly patients who were malnourished (Table 2 and Table 3) showed an improvement in their nutritional status (of 101 subjects, 67 were classified into the success group).
The results of the univariate analysis showed that Alb or TLC was not separately associated with NST outcome, but CONUT score was significantly associated with NST outcome. This result shows that comprehensive nutrition evaluation on the basis of CONUT seems to be useful in identifying high risk patients. In addition, as described in our methods, we treated CONUT as a quantitative variable and identified the cut off value as 8, which means that 5-8 indicates moderate malnutrition. When patients are evaluated using CONUT, the consideration of score is important, in addition to the assessment of the level of under nutrition as determined using the original method. Although focusing on a different population, another study of terminal liver disease patients needing liver transplantation reported that the cumulative survival rate in patients with a CONUT score ≤ 7 was significantly higher than in patients with CONUT score ≥ 8 [27]. The cut off value in the previous study is nearly equal to that in our study. Regarding the cut off value of CONUT for prognosis prediction of serious patients, a CONUT score of 7-8 would be valid.
In regard to parenteral nutrition, TPN is usually used when the intestines are not functioning, and when patients need hyperosmotic parenteral nutrition for more than two weeks [28]. This occurs in patients experiencing, for example, lack of digestive and absorptive function due to a digestive disorder, ileus, severe acute pancreatitis, or chemotherapy. This means that the use of TPN is limited regardless of disease severity. Therefore, TPN appeared not to be a significant factor. In contrast, PPN is widely used [29] when nutrition supply by oral or enteral nutrition is not sufficient, for example, when nutritional requirements are not met with enteral nutrition despite functioning intestines, or when patients are expected to resume oral intake in a short period of time such as in cases of gastroenteritis or during preparation for surgery. This means that patients who were using PPN at the time of NST intervention would have been in a severely malnourished condition. Therefore, we postulate it would be difficult to sufficiently improve nutrition status in the short-term for patients on PPN in this condition. In this study, we found two NST outcome predictors for the elderly: (1) CONUT score ≥ 8 and (2) The presence of PPN. At the time of NST intervention, more comprehensive and targeted nutrition therapy is recommended for patients with one or both of these risk factors, in order to ensure NST therapy is successful. Based on these findings, we plan to enhance the NST intervention method for patients who meet these criteria, and devise an improved NST intervention method in order to improve the success rate of NST.
This study has limitations. This study has a small sample size because it was conducted at a single institution, and therefore may not be widely generalizable. In the future, we plan to verify the validity of the above NST outcome predictors, (1) CONUT score ≥ 8, and (2) The presence of PPN, at the time of NST intervention with a prospective cohort and multicenter study with additional higher function hospitals in urban areas.
In conclusion, our results indicate that at the time of NST intervention, (1) CONUT score ≥ 8 and (2) The presence of PPN were found to be negatively associated with NST outcome for the elderly. Identifying older patients at risk of non success NST outcome is important to increase the success rate of NST therapy.
The authors are grateful to the participants, the staffs of nutritional section of Minami osaka Hospital, and the members of the same hospital. This study does not have any support or relationship that may pose a conflict of interest.
M Hiramatsu, K Kato, C Momoki, Y Yasui, and D Habu contributed to study conception and design. M Hiramatsu, Y Oide, C Kaneishi, A Mori, and T Fukuda contributed to data acquisition. M Hiramatsu, K Kato, CMomoki, and D Habu contributed to analysis and interpretation of data, and drafting and revision of the manuscript. All authors have approved the final version of the manuscript.