25-OH vitamin D levels above 30 ng/mL activate HCAP-18, which impacts the immune response to respiratory viruses. While emerging data suggests a link between low levels of 25-OH vitamin D and an increased incidence of respiratory infections, the relationship between these levels and severity of infection is unknown. We hypothesized that infants with insufficient 25-OH vitamin D levels would have more severe viral respiratory infections.
Healthy infants admitted with fever and cough or congestion were prospectively enrolled and 25-OH vitamin D levels were measured. Presumptive bacterial infections were excluded. Nasopharyngeal specimens were collected for determination of viral etiology by polymerase chain reaction (PCR). Demographics and severity markers were recorded. Levels were categorized as sufficient (≥ 30 ng/mL), or insufficient (< 30 ng/mL).
We enrolled 90 subjects. 25-OH vitamin D was < 30 ng/mL in 66%. Respiratory syncytial virus (RSV) was the most common virus. On multivariable analysis, breastfeeding was the only risk factor found to be associated with insufficiency. Administration of antibiotics and intravenous fluid, and PICU admission were not significantly different in infants with insufficient levels. However, infants with insufficient vitamin D had increased durations of both hospitalization (1.4 days, 95% confidence interval (CI) = 1.0-1.9 days) and oxygen use (2.2 days, 95% CI = 1.4-3.5 days), and used a higher level of oxygen supplementation (odds ratio = 4.58, 95% CI = 1.58-13.23).
Infants with 25-OH vitamin D insufficiency had more severe viral respiratory illnesses. Breastfeeding was associated with insufficiency. Vitamin D supplementation during pregnancy might be a strategy to reduce risk and severity of respiratory illness in infants.
Bronchiolitis, Vitamin D insufficiency, Pediatrics
The prevalence of 25-OH vitamin D insufficiency (level of < 30 ng/mL) in persons aged > 1 year in the United States is 31% [1]. Growing evidence suggests that in addition to calcium homeostasis, vitamin D also plays key roles in innate and adaptive immunity and thus vitamin D deficiency has been hypothesized to be a culprit in the risk and severity of acute respiratory infection (ARI) in both adults and children. Vitamin D receptors are widely expressed amongst cells of the innate and adaptive immune system, including dendritic cells and macrophages. When stimulated, these cells produce human cathelicidin protein (HCAP-18), which augments the microbial killing capacity of phagocytes and activates anaphylactoid factors that promote neutrophil and monocyte migration to the site of infection [2]. In addition to its immunologic activating effects, vitamin D also suppresses the release of potentially damaging pro-inflammatory cytokines and chemokines, to include IL-1β, IL-6, IL-8, and TNF-α [3].
Conversion of vitamin D to the active form in humans begins with vitamin D3 production in the skin upon exposure to sunlight. Given the effects of vitamin D on immunity, it is plausible that the seasonality of ARI may be driven in part by periods of active vitamin D insufficiency during the winter and early spring months attributable to lower rates of skin conversion to D3. Although several studies have now shown an increased incidence of ARI in subjects with low 25-OH vitamin D levels [4-9], very little is known regarding 25-OH vitamin D levels and their relationship to the severity of ARI. We therefore investigated the correlation between 25-OH vitamin D levels and severity of viral respiratory tract infections in a prospective cohort of otherwise healthy children hospitalized at a community based children's hospital in Tacoma, WA. We hypothesized that children with lower 25-OH vitamin D levels would have more severe disease.
From February 14, 2014 to May 11, 2015, we recruited otherwise healthy children < 2 years of age with a febrile presumed viral, lower respiratory illness admitted to the Mary Bridge Children's Hospital (MBCH). MBCH is a tertiary care children's hospital with 82 beds located in Tacoma, WA. Inclusion criteria included a fever and at least one of the following symptoms: cough, congestion, increased work of breathing, or wheezing. Exclusion criteria included chronic medical illness; primary diagnosis of a bacterial infection; and history of asthma, immunodeficiency or prematurity. Respiratory infections were categorized as bronchiolitis if the subjects had increased work of breathing or wheezing in addition to the cough and congestion. All treatment decisions were made by the inpatient pediatric hospitalist attendings, independent of this study. All subjects' parents or guardians provided signed informed consent prior to enrollment. The study was approved by the Infectious Disease Institutional Review Board of the Uniformed Services University of the Health Sciences (IDCRP-045), and by the Multicare Institutional Review Board.
At enrollment, demographics, medical history, clinical symptoms, vital signs and physical exam findings were recorded by clinical research personnel using a standard clinical record form, and a nasopharyngeal swab (Nylon-flocked, Copan Diagnostics, Corona, CA) for respiratory viral detection was collected. A serum sample of approximately 3 mL was obtained for 25-OH vitamin D analysis. 25-OH vitamin D levels of < 30 ng/mL were considered vitamin D insufficient (VDI). Levels ≥ 30 ng/mL were considered to be vitamin D sufficient (VDS). Although there is a debate about the cutoff level for vitamin D sufficiency, we chose to use the 30 ng/mL cutoff based on the work of Bahn, et al. who determined that a level of at least 32 ng/ml is needed for activation of the immune response [10].
Both the serum sample and the nasal swab from each subject were sent at room temperature to Laboratories Northwest, co-located on campus, for 25-OH vitamin D assay of the serum, and detection of viral pathogen. The 25-OH vitamin D levels were obtained using the Abbott Architect Chemoluminescent Microparticle Immunoassay (Abbott Laboratories, Abbott Park, IL). Viral pathogens were detected with the eSensor® Respiratory Viral Panel (Genmark Diagnostics, Carlsbad, CA), a multiplex polymerase chain reaction (PCR) based assay that tests for Influenza viruses A and B, Adenovirus, Human parainfluenza viruses 1-3, Human rhinovirus, Respiratory syncytial virus (RSV) A and B, and Human metapneumovirus. If phlebotomy was unsuccessful, the subject was removed from the study.
Primary outcome measures included the use of intravenous fluids and antibiotics, need for intensive care and oxygen supplementation, maximum amount of oxygen flow needed, and the number of days of fever, of oxygen requirement, of intensive care, and of hospitalization.
First, we examined bivariable associations between demographics and potential risk factors by 25-OH vitamin D level (VDI vs. VDS). We performed chi-square tests (exact test, if appropriate) for dichotomous and categorical variables and Wilcoxon rank-sum tests for continuous variables. We then performed multivariable logistic regression to examine factors associated with vitamin D levels, including variables that were p ≤ 0.05 in the bivariable analysis. Next, we examined differences in clinical severity by categorical 25-OH vitamin D levels (VDI vs. VDS) using chi-square tests (exact test, if appropriate) for categorical outcomes and Wilcoxon rank-sum test for continuous outcomes. We adjusted p-values in the bivariable analysis to control for multiple comparisons using false discovery rate (FDR) [11]. We performed multivariable analysis for clinical outcomes that were significantly different across 25-OH vitamin D categories with adjustment of potential confounders: ordinal logistic regression for ordinal outcomes (i.e. volume level of oxygen use), and linear regression for duration of hospitalization and oxygen supplementation. We fit the linear models with log transformed duration data; however, for intuitive understanding, we presented the exponentiated values of point estimates and 95% confidence intervals (CIs) in the linear models. Tests with p ≤ 0.05 (2-sided) were considered statistically significant. Analyses were performed using SAS 9.3 (SAS Institute, Inc., NC).
We approached 158 eligible subjects for participation in the study. Subjects were only enrolled during the viral respiratory season. Fifty (32%) declined, with 90% citing the need for a blood sample. Of the 108 subjects enrolled, 14 (12.9%) were administratively withdrawn due to inability to obtain a blood specimen and 4 (0.2%) declined collection of nasal swab, leaving 90 subjects who completed the study. There were no demographic differences among those who declined, withdrew or completed the study. Twenty-two (24%) patients required intensive care during their stay. Mean age of enrollees was 8 months, with 43% female. Seventy-one percent were Caucasian, 6% Pacific Islander, 4% African-American, 4% Native American, and 15% more than one race.
The median 25-OH vitamin D level was 24.5 ng/mL (interquartile range (IQR): 18-34 ng/mL) (Figure 1). Fifty-nine (66%) had levels < 30 ng/mL (VDI), including 19 (20%) with levels ≤ 12 ng/mL (the lowest limit of our assay), with only 31 (34%) having sufficient levels of 25-OH vitamin D (VDS, ≥ 30 ng/mL). Testing for viral etiology showed that 58 (64%) were positive for RSV. Other viruses detected, including co-detections, were Rhinovirus (n = 9), Human metapneumovirus (n = 7), Influenza virus (n = 4), Parainfluenza virus (n = 2), and Adenovirus (n = 1). Seven children had co-detection of two viruses. No virus was detected in 18 subjects.
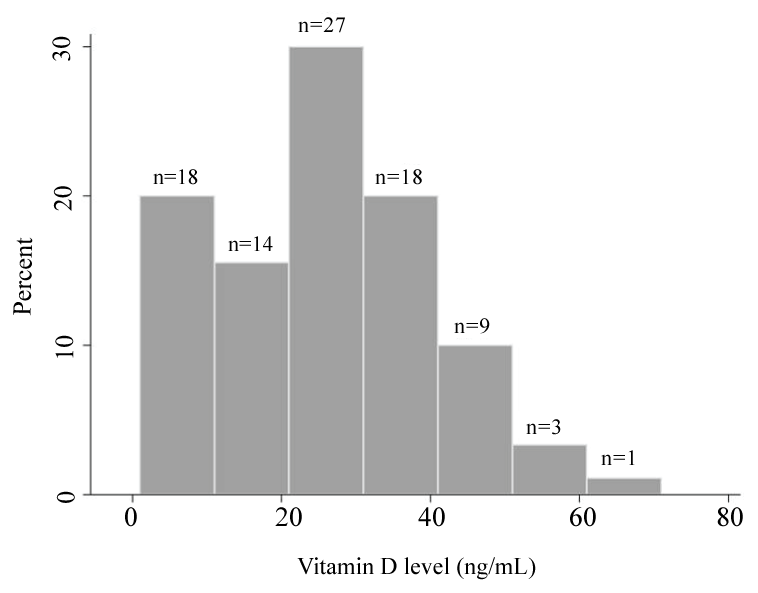 Figure 1: Distribution of 25-OH vitamin D levels.
View Figure 1
Figure 1: Distribution of 25-OH vitamin D levels.
View Figure 1
Two demographic features were associated with VDI: younger age and breastfeeding (Table 1). Supplementation with vitamin D had no impact on 25-OH vitamin D levels, with similar proportions of VDI and VDS subjects reportedly receiving supplements. There were no differences in 25-OH vitamin D levels by sex, race/ethnicity, tobacco exposure, parental education, or BMI. Looking at viral etiologies, RSV was more commonly detected in VDI as compared to VDS subjects (73% versus 48%; p = 0.021, Table 1). On multivariable analysis, only breastfeeding was significantly associated with VDI (Table 2).
Table 1: Association between demographics, risk factor and level of vitamin D among 90 children hospitalized. View Table 1
Table 2: Multivariable analysis of potential confounders, including age, RSV detection and breastfeeding status, and 25-OH Vitamin D sufficiency among 90 children hospitalized for viral respiratory tract infections. View Table 2
Associations between 25-OH vitamin D categories and clinical severity are shown in Table 3. We noted that the proportion of intravenous fluid supplementation, intensive care treatment, artificial ventilation, and antibiotic treatment were all markedly higher in subjects with VDI when compared to VDS subjects, but the differences were not statistically significant after accounting for multiple comparison. Among those who received oxygen support (N = 68), VDI subjects used significantly higher level oxygen support compared to VDS subjects (p = 0.020). The difference remained significant in multivariable ordinal logistic regression: VDI subjects were more likely to use higher levels of oxygen support (OR = 4.58; 95% CI 1.58-13.23; data not shown) adjusting for breastfeeding, age and RSV detection.
Table 3: Association between vitamin D level and clinical severity outcomes. View Table 3
Figure 2 displays further evidence of more severe disease in VDI subjects. We found that VDI subjects had significantly longer median days of hospitalization (5 versus 3 days, p = 0.030), and of oxygen support for subjects requiring oxygen (4 versus 1 days, p = 0.001) when compared to VDS subjects. However, the median duration of parent-reported fever was similar between the two groups (2 versus 2 days, p = 0.830).
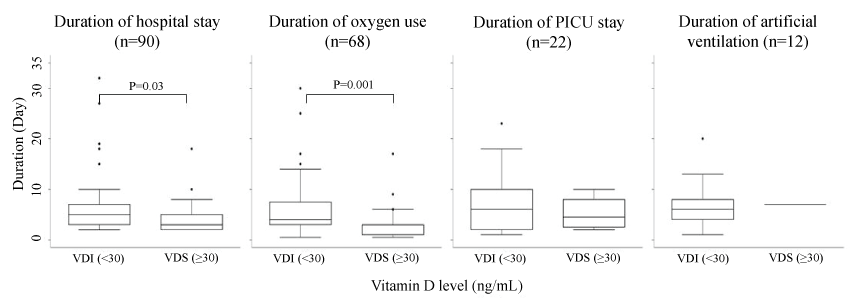 Figure 2: Duration of receiving medical support among ARI pediatric cases who needed such treatment shown by vitamin D level as box-and-whisker plots.
Note: The boxes represented the median (thick line) and 25th and 75th percentile (box end) of duration in each group. The whisks indicate 1.5 times of interquartile range below 25th percentile and above 75th percentile. Difference of duration between vitamin D groups were tested using Wilcoxon rank sum test. P-value was adjusted for multiple comparison.
View Figure 2
Figure 2: Duration of receiving medical support among ARI pediatric cases who needed such treatment shown by vitamin D level as box-and-whisker plots.
Note: The boxes represented the median (thick line) and 25th and 75th percentile (box end) of duration in each group. The whisks indicate 1.5 times of interquartile range below 25th percentile and above 75th percentile. Difference of duration between vitamin D groups were tested using Wilcoxon rank sum test. P-value was adjusted for multiple comparison.
View Figure 2
Multivariable log-transformed linear regression analysis verified that VDI patients had longer duration of hospitalization (1.4 days longer, 95% CI: 1.0, 1.9 days) and oxygen use (2.2 days longer, 95% CI: 1.4, 3.5 days) compared to VDS subjects with adjustment of breastfeeding, age and RSV detection (data not shown).
We found that infants with 25-OH vitamin D levels < 30 ng/mL suffered from more severe viral respiratory infections according to several objective measures when compared to those infants with sufficient levels, even when adjusting for age, breastfeeding and RSV status. We had a high level of VDI in our population, but we did not find any risk factors associated with VDI other than breastfeeding and younger age, suggesting that the most important predictor of neonatal vitamin D status in our population was likely related to maternal vitamin D stores.
Pathophysiological evidence has been mounting that a sufficient level of 25-OH vitamin D is needed to protect against severe RSV and other viral respiratory tract infections. According to Bahn, et al. the activation of HCAP-18, which is critical in the immune response to respiratory viruses, requires a minimum 25-OH vitamin D level of 32 ng/mL [10]. Randolph, et al. found that children hospitalized with RSV bronchiolitis were more likely to carry the GC1s haplotype, which is associated with higher levels of 25-OH vitamin D binding protein, and consequently, less freely available 25-OH vitamin D [12]. Additionally, Hansdottir, et al. found that human airway epithelial cells infected with RSV and exposed to 25-OH vitamin D resulted in a decreased inflammatory response with minimal impact on the capacity for viral clearance [13].
From a clinical perspective, increased rates of viral respiratory tract infections have been shown in both infants and children with insufficient levels of 25-OH vitamin D [4-9]. Several studies, all outside the U.S., have shown that newborns with lower cord blood levels of 25-OH vitamin D are at increased risk for acute nasopharyngitis [4], lower respiratory tract infections [5,7,9], and RSV bronchiolitis [6]. Another study of pregnant Norwegian women detected a decreased risk of lower respiratory tract infections during the first 3 years of life in those infants with higher maternal mid-pregnancy 25-OH vitamin D levels [14]. A prospective study of 743 Canadian children ages 3-15 years also found a correlation between lower serum 25-OH vitamin D levels and rates of laboratory-confirmed viral respiratory tract infection [8]. A limitation of such studies has been the difficulty in proving causal association, particularly when potentially confounding factors such as younger age and season have also been associated with both increased rates of viral respiratory tract infections, and vitamin D insufficiency.
More definitive evidence of VDI and the risk of viral respiratory infection stems from prospective studies of vitamin D prophylaxis [15-17]. Grant, et al. demonstrated a reduction in primary care visits for ARI among New Zealand infants randomized to receive prophylaxis with high dose vitamin D, and whose moms were also prophylaxed during pregnancy [15]. Goodall, et al. also found a reduction in laboratory confirmed upper respiratory tract viral infections and viral loads in Canadian adults given vitamin D3 [16]. In another study, Swedish adults with frequent respiratory infections given vitamin D3 prophylaxis also had a reduction in disease burden [17]. Conversely, two other studies of vitamin D prophylaxis failed to show any effect on the rate of ARI: no reduction in upper respiratory tract infection was observed among U.S. adults aged 45-75 years who were given vitamin D3 supplementation [18]; similarly vitamin D3 supplementation among adults in New Zealand had no impact on the incidence nor severity of respiratory infections [19].
Few studies have addressed whether vitamin D insufficiency is correlated with an increased severity of symptoms with ARI. One prospective study of 28 Japanese children hospitalized with lower respiratory tract infections did show a significant correlation between VDI and use of supplementary oxygen and artificial ventilation [20]. A study of 300 German adults with community acquired pneumonia showed that a modestly more severe course of illness was associated with low levels of 25-OH vitamin D [21]. By contrast, no such association was found in a large study investigating 25-OH vitamin D levels and the duration of respiratory tract infections among elderly Norwegians [22].
Recently, Beigelman, et al. reported a study with a similar design to ours, investigating the relationship of vitamin D levels and severity of RSV bronchiolitis amongst hospitalized infants, but with opposite results [23]. Of note is that the 25-OH vitamin D assay used in their study was different than our assay, which may also affect the validity of comparing results of our studies. They found no differences in severity as measured by duration of hospitalization, lowest oxygen saturation level and bronchiolitis severity score in their population related to vitamin D status. However, there are several important differences in our populations-the mean age of their subjects was lower, at 4 versus 8 months; they had a larger percentage of African American participants (42% versus 4%); and most notably a very low number of subjects had vitamin D insufficiency compared to our subjects (median level of 36.8 ng/mL versus 24.5 ng/mL). The relationship of vitamin D to bronchiolitis severity might vary according to age. Racial differences in vitamin D metabolism may also exist. Finally, given the critical threshold of 30 ng/mL needed to trigger the immunologic effect of 25-OH vitamin D, [10] Beigelman's study may not have had enough subjects with low vitamin D levels to discern an effect on severity.
Our study provides novel information in that it is one of very few to date investigating whether lower levels 25-OH vitamin D levels correlate with the severity of viral respiratory infections. Although our numbers were small, we were able to demonstrate higher severity of the infection among these infants, even after controlling for viral etiology, age and breastfeeding status. Specifically, infants with VDI had significantly longer durations of hospitalization and oxygen use, and also required a higher level of oxygen support. These findings augment those of the Japanese study [20] and provide support for the importance of maintaining 25-OH vitamin D levels ≥ 30 ng/mL to not only prevent respiratory infections in children, but lessen their severity.
Previous work has revealed that 25-OH vitamin D levels in newborns directly correlates with maternal levels during pregnancy [24], and that supplementation of women during pregnancy with higher dose 25-OH vitamin D may result in less VDI in newborns, as well as in decreased rates of asthma in childhood [25]. Our findings further highlight the importance of addressing the issue of maternal VDI during pregnancy. Higher supplementation beyond prenatal vitamins may be warranted, particularly as there was no difference in levels associated with those infants who had been prescribed supplemental vitamin D compared to those without. Potentially there was poor compliance with administering the vitamin D to the infants.
Our study had several limitations. A larger study might have allowed a number of sub-group analyses to further tease out severity impact. 25-OH vitamin D levels were measured at the time of clinical presentation/enrollment, and the correlation and impact of levels over the duration of illness could not be determined. Lastly, our population was confined to otherwise healthy subjects in the Pacific Northwest, and so our findings may not be applicable to other populations in other regions of the US.
Our findings suggest that achieving sufficient levels of 25-OH vitamin D in healthy infants and toddlers might be an effective intervention to reduce severe viral respiratory infections. Further interventional studies in either infants or pregnant females would be helpful in assessing this strategy to prevent viral respiratory infections that are associated with severe clinical outcomes in infants and children.
We are indebted to the support from the Multicare Institute of Research for successful completion of this project. We are also thankful for the support of the intravenous therapy team at Mary Bridge Children's Hospital. We are most indebted to the families who volunteered to participate in this project. The views expressed are those of the author(s) and do not reflect the official policy or position of the US Army Medical Department, Department of the Army, Department of Defense or the U.S. Government. The investigators have adhered to the policies for protection of human subjects as prescribed in 45CRF46.
None of the authors have conflicts of interest to report. All subjects' parents or guardians provided signed informed consent prior to enrollment. The study was approved by the Infectious Disease Institutional Review Board of the Uniformed Services University of the Health Sciences (IDCRP-045), and by the Multicare Institutional Review Board.
This work was supported by the Infectious Disease Clinical Research Program (IDCRP), a Department of Defense (DoD) program executed through the Uniformed Services University of the Health Sciences. This project has been funded in part, with federal funds from the National Institute of Allergy and Infectious Diseases, National Institutes of Health (NIH), under Inter-Agency Agreement [Y1-AI-5072]. The remainder of the funding was provided by an unrestricted research grant from the Multicare Institute of Research and Innovation.