Aging and hypertension are common factors in increased central arterial stiffness and the prevalence of atrial fibrillation (AF). Individualized weighing of the risk-benefit of anticoagulation is recommended in patients with AF who have low established risk scores or, conversely, are at increased risk for bleeding. Parameters of arterial stiffness and wave reflection could improve risk stratification.
The objective of the present study was to determine whether pulse pressure (PP), pulse wave velocity (PWV), and pulse wave morphology, considered markers of vascular stiffness, change during episodes of paroxysmal AF in elderly patients.
Elderly patients with paroxysmal AF (n = 23) attended biannual appointments for four years, totaling 28 sinus rhythm appointments and 33 AF appointments. Brachial and central PP and PWV were assessed using Applanation tonometry (TA). In patients under AF, brachial PP was reduced (from 67.1 ± 23.7 to 56.9 ± 17.7 mmHg; P = 0.027) and PWV was maintained (from 11.0 ± 0.5 to 10.2 ± 0.5 m/s; P = 0.313). These patients showed early reflected wave return, assessed by the inflection point (Pi) during ventricular systole (from 39.8 ± 15.5 to 34.0 ± 11.5 mm; P = 0.048). No changes were observed in the augmentation index (from 26.7 ± 15.2 to 29.8 ± 17.4%; P = 0.224). In the multivariate analysis, only age influenced PWV (P < 0.001). The arterial vascular stiffness markers (PWV and PP) had different behaviors during episodes of paroxysmal AF, and these biomarkers could be conducted to different risk stratification in patients with AF.
Aging promotes marked changes in the cardiovascular system, especially in large arterial vessels. All arterial vessels layers undergo important modifications, such as the proliferation of collagen fibers, accumulation of glucose degradation products, and reduction in elastin, among others [1]. Such changes promote a loss of elasticity of the great vessels and a consequent increase in arterial stiffness [2]. In these circumstances, the arterial pulse wave velocity (PWV) increases as vascular stiffness progresses as a result of aging [2,3]. Measurement of the PWV in the carotid-femoral section (c-fPWV) using applanation tonometry (AT) is considered the gold standard for the detection of vascular stiffness [4]. The AT is also able, among other measures, to identify the morphology of the wave generated by the ventricular contraction and the reflected wave that returns to the heart, in addition to being able to measurethe central pressures and identify parts of the cardiac cycle [5,6]. c-fPWV can detect vascular stiffness before the patient becomes hypertensive [7], and it is directly related to cardiovascular risk (CVR) [8].
The association of certain arrhythmias, such as atrial fibrillation (AF), with vascular stiffness, is cited by several authors [9,10]. Some of the predisposing factors of AF are the same factors that promote arterial stiffness, such as aging and systemic arterial hypertension (SAH), and the prevalence increases with age [11], high morbidity, and mortality [12]. The constant search for biomarkers that can predict AF episodes and parameters of arterial stiffness and wave reflection that could improve risk stratification may, in the future, indicate the best pathway for the treatment and prevention of AF [13]. In this regard, c-fPWV and arterial pulse pressure (PP) have been associated with several markers of vascular rigidity with inconsistent results [14-16] and are considered in the stratification of CVR [17]. Examples of biomarkers include everything from pulse and blood pressure to basic chemicals to more complex laboratory tests of blood and other tissues. As quantifiable medical signs, cfPWV, and PP fall into the category of biomarkers that modern laboratory science allows us to measure reproducibly [18]. This study aimed to verify whether these two biomarkers (PP and c-fPWV), would change their values during episodes of paroxysmal AF in elderly patients.
This retrospective study used the Study of the Pulse Wave Velocity in the Elderly from the Urban Area in Brazil (EVOPIU for its acronym in Portuguese) database of patients evaluated from 2014 to 2016, and the Ethics Committee of the Federal University of Uberlândia approved it (UFU; CAAE-37440114.3.0000.5152) with support from FAPEMIG (Minas Gerais Research Foundation). EVOPIU followed up1, 192 patients older than 60 years, for four years (2014 to 2019), with semiannual clinical and laboratory evaluations including AT to evaluate c-fPWV, central pressures, and pulse wave morphology. Further details on EVOPIU were previously published [19]. For the present study, patients who presented with paroxysmal AFduring follow-up, i.e, with a repetitive pattern in AF, sometimes with AF and sometimes with sinus rhythm (SR) detected by an electrocardiogram (ECG) performed at each medical visit, were included. All patients were evaluated at least once regarding SR and once in AF. Three patients were evaluated more than once in SR and AF throughout the study.
Aortic PWV was measured with AtCor Sphigmocor Xcel device, model EM4C (AtCor Medical, Sydney, NSW, Australia) by sequentially recording electrocardiogram-gated carotid and femoral artery pressure waves, using the intersecting tangent algorithm to determine the foot of the waveform. Each result already being calculated as the ensemble average over (on average) 11 beats (standard deviation between repeat PWV measurements were 0.97 m/s before and 0.66 m/s after. For patients with unstable heart rates, the 20-second capture may be required to obtain a valid measurement. The automatic capture section always happens when the signal quality of the waves is valid (the signal turns to green). Once the SphygmoCorXCEL system detects seconds of regular carotid pulse period, the femoral cuff will be inflated [20].
After AT, if the occurrence of AF was confirmed, the patient was referred to the outpatient clinic for treatment. In the subsequent medical visit, only those patients without a previous history of cardioversion or using antiarrhythmic drugs specific for AF were included in the study. Patients whose c-fPWV measurements were not concomitant with an ECG were excluded from the study. Of the 1,192 patients included in the EVOPIU database, 2,689 ECGs were examined during a 4-year follow-up, and 26 patients with AF compatible with the inclusion criteria were found. Three patients were excluded because they did not perform an ECG concomitant with the c-fPWV measurement. The 23 patients included in the present study attended semiannual consultations, totaling 28 consultations in SR and 33 consultations in AF. The following were evaluated: systolic (bSBP), diastolic (bDBP), pulse (bPP), and mean (bMAP) brachial pressures; centralsystolic (cSBP), diastolic (cDBP), pulse (cPP), and mean (cMAP) pressures; pressure amplitude (PA); augmentation index (Aix); and c-fPWV. For wave morphology, ventricular ejection (VT) duration in milliseconds, the relative time of ventricular ejection (RTVE) in %, the height of the reflected wave inflection on the ventricular ejection wave (P1) in mmHg, final systolic pressure (FSP) in mmHg, and amplification of pulse pressure (bPP/cPP) were evaluated. Figure 1 shows the details of the arterial pulse wave [6].
The data were tested for normality using the Shapiro-Wilk test and for homogeneity with the Levene test. For the differences between means, Student's t-test or the Mann-Whitney test were used according to the distribution. c-fPWV was adjusted considering it as the dependent variable and bMAP, age, and sex as the independent variables. Univariate and Stepwise multivariate were used to assess possible associations. An alpha error of 0.05 was considered significant. For the statistical calculations, the program STATA v. 16.1 was used.
Table 1 shows the clinical and laboratory characteristics and the AT values for the patients when experiencing SR and AF. The mean interval between visits was 11.1 ± 1.4 months. Table 2 shows the univariate and multivariate analyses in which c-fPWV is the dependent variable. (Figure 1) shows the morphology of the pulse wave of an elderly patient and the respective parameters evaluated in the present study. The average ages of patients with and without AF are similar because some patients were first examined with AF and then at a sinus rhythm, while other patients had the opposite clinical sequence. The percentage of patients using antihypertensive drugs was similar in both periods, with the exception of calcium channel blockers that reduced their use during the PA period. We found significant increases in HR during PA compared to the period in sinus rhythm in addition to reductions in bPP and Pi height.
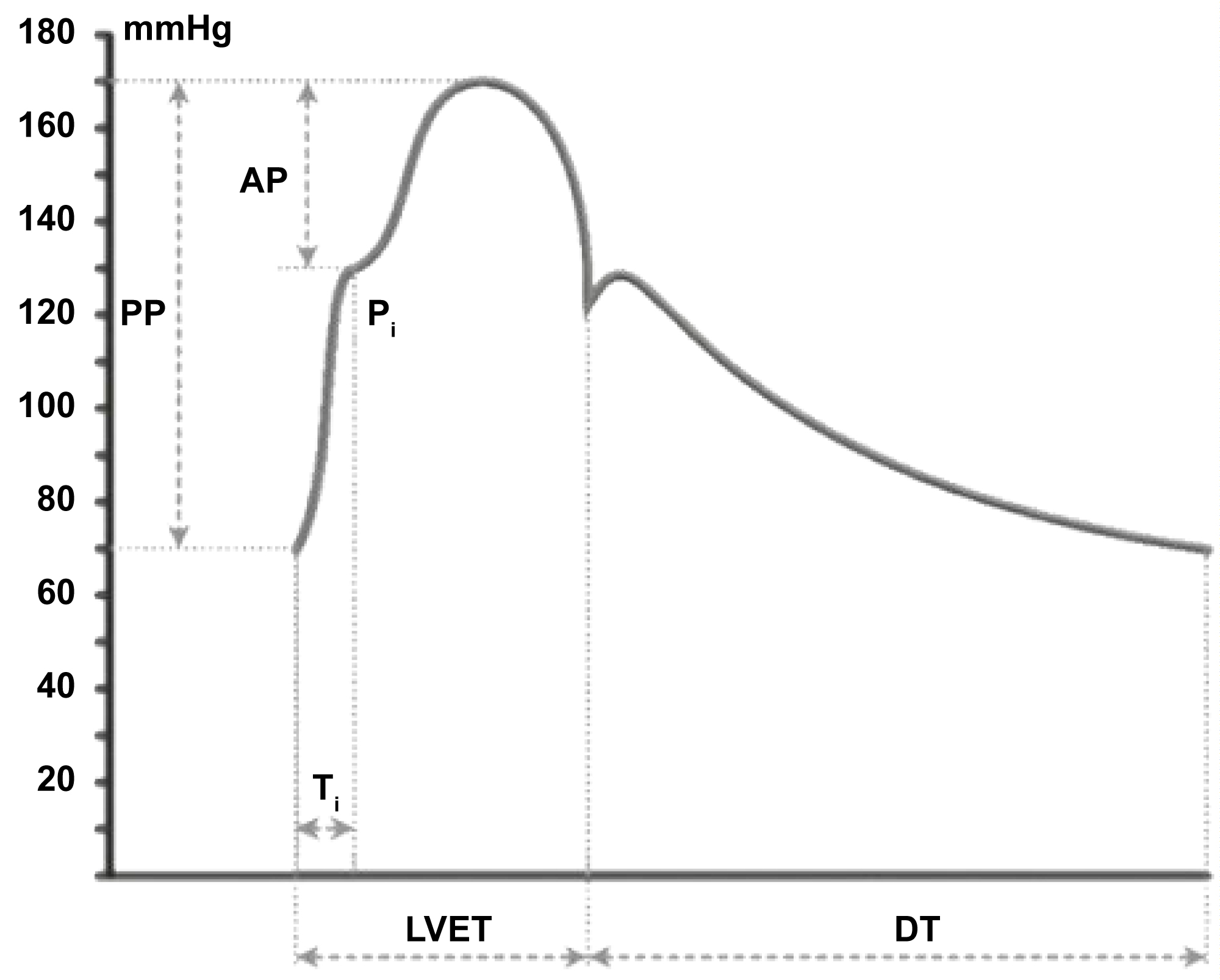 Figure 1: Some parameters defined in pulse wave analysis in elderly people [6]: PP: Pulse Pressure; AP: Augmented Pressure, defined by wave reflection; Pi: Inflection Point; TI: The Timing of the Reflected Wave; LVET: Left Ventricular Ejection Time; DT: Diastolic Time; Aix: Augmentation Index (AIx = AP/PP).
View Figure 1
Figure 1: Some parameters defined in pulse wave analysis in elderly people [6]: PP: Pulse Pressure; AP: Augmented Pressure, defined by wave reflection; Pi: Inflection Point; TI: The Timing of the Reflected Wave; LVET: Left Ventricular Ejection Time; DT: Diastolic Time; Aix: Augmentation Index (AIx = AP/PP).
View Figure 1
Table 1: Clinical and Laboratory Characteristics of Patients with/without AF (n = 23). View Table 1
Table 2: Association between c-f PWV and independent variables in elderly with Atrial Fibrillation (n = 23). View Table 2
The data obtained show that during episodes of AF, the adjusted values of c-fPWV remain unchanged, while bPP decreases significantly. Additionally, the reflected wave arrives earlier in the heart at the beginning of ventricular systole (P1) during episodes of AF (Table 1). Since c-fPWV is a gold standard method to measure the arterial stiffness and, therefore, with structural alteration of the vascular wall [4], it would be rational to think that c-fPWV values do not change during paroxysmal AF. By adjusting the c-fPWV values by bMAP, we eliminated the possibility that blood pressure reductions or elevations could be interpreted as real changes in the structure of large arterial vessels. Even so, the c-fPWV values adjusted or not remained constant during episodes of AF compared to those found during the SR period. Chu, et al. demonstrated that patients with AF had a higher PWV than that found in the control group with SR [21].
However, these authors studied patients with permanent AF, and after PWV adjustment by heart rate (HR), the values were similar to those of the control group. Lee, et al. also found higher PWV in 33 patients with chronic AF, regardless of age; however, the authors do not cite PWV adjustment by MAP [9].
On the other hand, PP has long been described as a measure of the stiffness of large arterial vessels [22]. However, it is potentially influenced by other factors related to cardiac function, such as HR, systolic volume and ventricular ejection pattern [23], which seems to have occurred in our study, where the VE time was shortened about the total cardiac cycle during the AF period (Table 1). Besides, AF may be associated with significant hemodynamic consequences, such as several detrimental effects on cardiac hemodynamic data [24-26] including loss of the atrial contribution to ventricular filling with a reduction in end-diastolic pressure and volume in the left and right ventricles [27], and consequently reduction in systolic and pulse blood pressures, an increase in the mean diastolic pressure in the atria [27], a reduced interval for passive diastolic filling [28-30] and possible atrioventricular valvular regurgitation [28].
Some antihypertensive drugs may affect cSBP and cPP. The CAFÉ (Conduit Artery Functional Evaluation) study showed that reductions in cSBP and cPP were higher in patients who used vasodilators than in those who used diuretics and beta-blockers (BB) [31]. In contrast to our study, in the AF period, patients showed a significant decrease in bPP and a tendency to reduce cPP levels, although the use of BB remained constant between the AF and SR periods. This fact was possibly due to the presence of arrhythmia rather than the possible effects of anti-hypertensive medication.
An essential element, which characterizes the systolic phase of the pulse waveform, is the inflection point (Pi), which represents the point where forward and backward waves meet. When it appears on the pulse wave, it is "shoulder shaped." Ti defines the time delay in the backward wave, and Pi is the particular inflection point. Thus, the lower the Pi height, the earlier the reflex wave arrives during ventricular systole [6]. Regarding the morphology of the pulse wave, it was observed that the height of P1 in mmHg was lower in AF than in SR. This finding demonstrates that the reflected wave returns earlier to the heart during periods of AF. Increased HR (from 67.3 ± 14.8 to 76.5 ± 17.0 bpm; P = 0.013) and the relative reduction in the ventricular ejection period (from 33.2 to 36.9%; P = 0.013) may explain this fact.
Our data differ from those presented by Wilkinson, et al. [32], who demonstrated an increase in Ti (correspondent an increase of Pi) of patients with cardiac pacemakers after increasing the number of regular heartbeats per minute. However, the changes detected by these authors were acute; the Ti mean values were shown without a separation between young and elderly patients, and the results of AIx were not normalized to the frequency of 75 bpm. Moreover, for the elderly, it is known that arterial stiffness causes an increased pulse wave velocity through the arterial system. Therefore, if the forward (centrifugal) pressure wave travels faster owing to increased arterial stiffness, similarly, the backward (centripetal) pressure wave goes back to the center at a higher speed. Under reduced arterial viscoelasticity, the earlier superimposition of the two waves, in the protomesosystolic phase of the cardiac cycle, produces a further increase in systolic blood pressure and pulse pressure values [6]. Finally, due to irregular beating frequencies of the heart in AF patients, the Pi height could be different from those authors.
As expected, the PWV values found in our study are quite high, exceeding by more than 30% of the values found in elderly individuals with good vascular health [7]. On the other hand, the age of patients with AF persists associated with arterial stiffness, even after multivariate analysis, where the central and peripheral pressures are the other dependent variables (Table 2).When the multivariate analysis was performed, the age of the patients was the independent variable that most influenced c-fPWV.
However, each of the systemic pressure variables was also significant in the univariate analysis. We believe that the age factor, when present in the analyses, is so dominant that it would hide the participation of other independent variables. Cecelja, et al. [33], in a systematic revision study, showed that age and blood pressure were significantly independently associated with c-fPWV in 90% of studies, but in 90% of the total studies, the patients had under 60-years-old.
Our study has some limitations, such as the small number of paroxysmal AF patients, opening the possibility of "effect size" in the statistical analyses (type II error). The absence of measures of ventricular function could have excluded possible patients with heart failure, since central PP, AIx, but also aortic c-fPWV, key measures of arterial function, are susceptible to the left ventricular performance [31]. It could have detected asymptomatic patients with heart failure, which would make the studied sample more homogeneous concerning heart function. However, patients were their controls, which could make the error constant both with and without AF.
C-fPWV and brachial PP are two different entities. C-fPWV represents the aortic component (central), and brachial PP represents the peripheral component. Both represent the stiffness of arterial walls. Instead, bPP to be easily obtained in the clinical practice, it cannot represent the real conditions of the vessels about the stiffness in patients with AF fibrillation. In conclusion, the present datasuggest that the markers of vascular stiffness (c-fPWV and PP) had different behaviors in paroxysmal AF: while c-fPWV did not change, a reduction in bPP was observed in the periods in which the patient presented with AF and these biomarkers could conduct to different risk stratification in patients with AF. Future studies with a larger number of patients should be conducted.
FAPEMIG (Fundação de Apoio à Pesquisa do Estado de Minas Gerais).
None.
None.