Paraquat, Poisoning, Hemoperfusion
A 52-year-old caucasian male farmer from Walterboro, South Carolina presented to the Medical University of South Carolina Health-University Medical Center in August, one hour after ingestion of 20 oz of a 50/50 water/paraquat preparation, with suicidal intent, complaining of nausea and emesis of blue colored fluid. His vital signs were normal on admission other than an increased blood pressure of 159/92 mmHg. Upon presentation, physical examination was within normal limits. Plasma levels of paraquat were obtained and the patient underwent gastrointestinal evacuation via nasogastric tube and was given 50 grams of activated charcoal along with sorbitol. The patient was then transferred to the academic medical center's medical intensive care unit for further care.
The patient was immediately started on Continuous Venovenous Hemofiltration (CVVH) using hemoperfusion charcoal filter but was unable to continue dialysis as the filter clotted. Subsequently, CVVH was restarted without the hemoperfusion filter. By hospital day 2, the patient's creatinine (Cr) had increased to 2.1 mg/dl (baseline 1.1 mg/dl). He was started on cyclophosphamide 200 mg IV per day for 2 days and methylprednisolone 1 gram per day for 3 days. On hospital day 3, the patient's Cr increased to 3.4 mg/dl despite CVVH and subsequently developed blood tinged sputum, nausea and vomiting. A chest radiograph was obtained that showed new alveolar infiltrates (Figure 1). His oxygen saturation decreased to 93% requiring 2 liters of oxygen per minute. An arterial blood gas showed a PaO2 of 70, therefore, charcoal hemoperfusion was restarted. On hospital day 5, the patient further deteriorated with increased dyspnea, hypoxemia (oxygen saturation now 92% on 50% FiO2), PaO2 62 and basilar crackles on lung exam. The patient was started on dexamethasone 5 mg IV every 6 hours. After a total of 6 days of treatment with CVVH and a Cr plateau of 2.5 mg/dl, the patient's paraquat level was found to be 0.034 mg/mL, highly indicative of survival, and dexamethasone taper was then initiated.
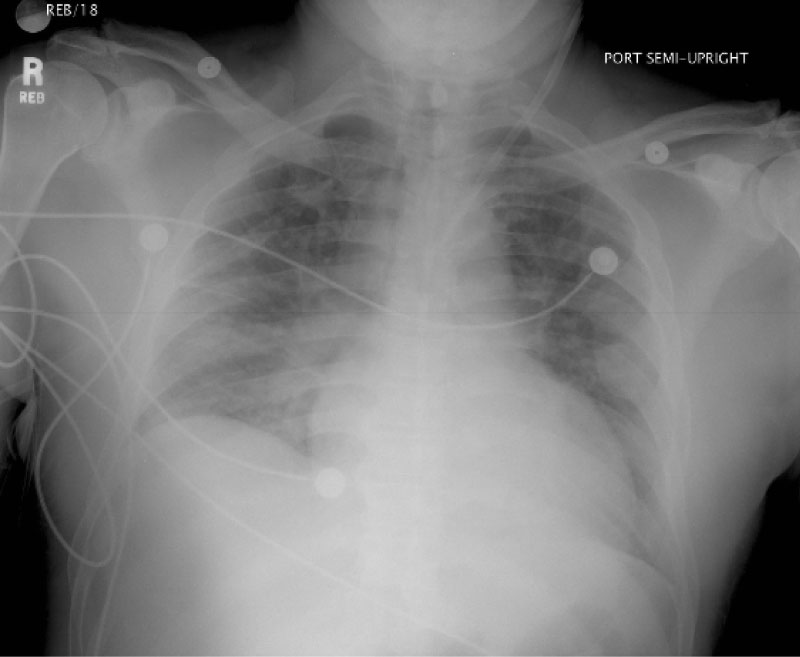 Figure 1: Chest X-ray from Hospital Day 4 demonstrating alveolar infiltrates and pulmonary edema indicative of the proliferative phase of paraquat induced lung disease. X-ray performed two days after initiation of Cyclophosphamide and Methylprednisolone. View Figure 1
Figure 1: Chest X-ray from Hospital Day 4 demonstrating alveolar infiltrates and pulmonary edema indicative of the proliferative phase of paraquat induced lung disease. X-ray performed two days after initiation of Cyclophosphamide and Methylprednisolone. View Figure 1
The patient continued to improve, received psychiatric evaluation and treatment, and eventually made a full-recovery. He was discharged on hospital day twenty-one with intensive psychiatric treatment related to his suicide attempt.
Paraquat is a toxic chemical that is used for weed and grass control. In the United States, paraquat is available primarily as a liquid in various strengths. Deliberate self-poisoning with pesticides such as paraquat continues to be a problem in the United States and abroad. One-hundred toxic paraquat exposures were reported in 2016 [1,2]. Therefore, awareness and education of health professionals about the serious consequences of exposure to paraquat and a general understanding of treatment is important. In the U.S. paraquat has "restricted use" and can only be used by licensed applicators. In our case, the patient was a farmer and that enable him to receive paraquat access through his line of work. Because of the high toxicity of paraquat, the form marketed in the United States has security features such as a blue dye, sharp odor, and an emetic agent. These characteristics are similar to the fluid that our patient reported ingesting, making our suspicion for actual paraquat ingestion stronger.
Mortality for paraquat poisoning correlates with the amount of paraquat in the blood [2-4]. Fatal outcomes are usually associated with plasma levels greater than 0.2 mg/mL at 24 hours after ingestion and 0.1 mg/mL at 48 hours [2]. Laboratory testing includes urine testing that detects paraquat concentrations of 1 mg/mL or above, gas chromatography (1 microgram/mL) and radioimmunoassay (< 0.1 microgram/mL). When comparing exposure modes, the least to most fatal forms of exposure are: inhalation, ingestion, and intravenous exposure [3-5].
It is hypothesized that paraquat induces end organ damage through the production of oxygen free radicals that oxidize NADPH and causing cell death [6,7]. Patients with paraquat poisoning should be monitored for the development of mucosal injury, acute renal failure, liver injury, and respiratory failure. The most damaging clinical effect of paraquat poisoning is pulmonary toxicity, leading to pulmonary fibrosis and respiratory failure [8]. This fibrosis typically develops days to weeks after ingestion and is responsible for respiratory failure which is the primary cause of death with ingestion of these agents [8]. This is because the agent is primarily concentrated in the lung, which has the highest oxygen tensions found in the body [9]. Furthermore, as paraquat excretion occurs primarily in the kidney, acute tubular necrosis may occur soon after ingestion, thus leading to decreased paraquat excretion and further toxicity. Based on the fact that our patient developed Acute Renal Failure (ARF) within the first day of admission, we were concerned about the magnitude of paraquat ingestion, as well as worsening toxicity secondary to impaired excretion of paraquat with ARF.
There are currently no generally accepted guidelines on the treatment of patients with paraquat poisoning. Treatment options for the paraquat ingestion consist of prevention of Gastrointestinal (GI) absorption, removal of paraquat from the blood, and alternative therapies such as immunotherapy and antioxidants. These interventions are discussed below and summarized in Table 1.
Table 1: Paraquat ingestion treatments. View Table 1
Prevention of absorption from the GI tract should be considered as an intervention in patients who present soon after ingestion and include treatment with cathartics, activated charcoal, diatomaceous earths, and gastric lavage. Of these options, gastric lavage followed by a dose of activated charcoal is typically employed, although the evidence for beneficial effects of gastric lavage and charcoal is poor with most studies showing no benefit [10-12]. Determining if, how, and when to implement treatments to reduce paraquat absorption should be based on the individual case and take into account the likely amount of paraquat ingested and timing since ingestion [10-14]. It is possible, but not certain, that early gastric lavage and charcoal treatment in our patient lead to overall decreased plasma paraquat absorption and a better clinical outcome.
Removal of paraquat from the blood can be performed through Hemoperfusion (HP), Hemodialysis (HD) and Continuous Venovenous Hemofiltration (CVVH). It has been suggested that removal of paraquat from the blood with hemoperfusion provides better clearance of paraquat than hemodialysis, and that the use of hemoperfusion within 12 hours of poisoning may reduce mortality [15]. CVVH may improve survival, reduce organ dysfunction, and prolong the survival time for patients to receive additional treatments [16-19]. In one study, survival rates for patients who received early hemoperfusion were more than double those who did not receive hemoperfusion (92.1% vs. 42.9%) [18]. Successful outcomes, however, have not been uniformly observed elsewhere, when hemodialysis or hemoperfusion were used alone or in combination [20-22]. Despite this controversy, if available and after assessing the likely amount of paraquat ingested, it is reasonable to initiate an early trial of hemodialysis or hemoperfusion for 4 to 6 hours daily, recognizing that treatment may be required for at least 2 to 3 weeks.
Other strategies have been developed for management of acute paraquat toxicity including immunosuppression and antioxidant therapy. Although there is a scarcity of good evidence, a recent systematic review with meta-analysis also found that patients who received antioxidant or immunosuppressant therapy in addition to hemoperfusion (such as cyclophosphamide, methylprednisolone, Vitamin E, Vitamin C, N-acetyl cysteine) had better survival rates compared to those who received hemoperfusion alone [23]. For acute paraquat poisoning, it is also important to limit oxygen therapy unless the PaO2 is less than 50 as this may contribute to the production of oxygen free radical species and further end organ damage.
The evidence of the effectiveness of immunosuppressant therapy is conflicting. However, the majority of evidence to date is supportive of this treatment for paraquat poisoning. Although the mechanism for immunosuppressants in paraquat poisoning is unclear, it is thought this decrease in inflammation reduces the introduction of oxygen free radicals produced by paraquat ingestion. A survival rate of 75% in 72 patients treated with high-dose cyclophosphamide (5 mg/kg/d) and dexamethasone (24 mg/d) for 2 weeks was reported in one study [24]. However, a subsequent study did not demonstrate the efficacy of this approach [25]. In one randomized control trial, patients with moderate paraquat poisoning had a decrease in mortality rate in the study group (31% vs. 86% in control group) when treated with cyclophosphamide, methylprednisolone and dexamethasone, compared to the control group who received only dexamethasone [26]. A Cochrane Review of patients with moderate to severe paraquat poisoning showed participants receiving glucocorticoids and cyclophosphamide demonstrated a lower death rate, although the overall quality of the studies was noted to be low [27].
Insufficient data exists to advocate the use of antioxidants such as Vitamin C, acetylcysteine, and deferoxamine. However, acetylcysteine is used routinely for renal protection in patients with compromised renal function prior to imaging studies and appears clinically safe [28]. Thus, it may be used in paraquat toxicity with a low adverse reaction profile and is presumed to work on the basis that it replenishes glutathione stores and may inhibit free radical production [29]. High-dose long-term antioxidants may increase the PaO2, enhance liver function, reduce lung fibrosis, and minimize lung dysfunction in patients with paraquat poisoning [30].
Paraquat poisoning is a rare, but potentially fatal clinical presentation. There are currently no generally accepted guidelines on the treatment of patients with paraquat poisoning. While there is a paucity of evidence for effective treatment of paraquat toxicity, early treatment to induce vomiting or with gastric lavage, charcoal, and diatomaceous earths may be helpful by reducing absorption. Once absorbed, hemoperfusion, hemodialysis and CVVH may be useful at lowering blood levels of paraquat. Alternative therapies that could be considered in treatment of paraquat poisoning include immunosuppressant and antioxidant therapies. In our case, the patient was able to achieve a positive outcome. A possible explanation for this includes vomiting and early gastric lavage/charcoal leading to decreased absorption of paraquat, and CVVH to decrease the paraquat level after absorption. If our patient did not improve or had higher levels of paraquat after CVVH, immunosuppressant or antioxidant therapy could have been initiated. Knowing about the clinical presentation, evaluation, and treatment options for paraquat poisoning is important to reduce morbidity and mortality in these cases.
This is original work that has not been published elsewhere. No funding was required for the preparation of this manuscript. The authors have no conflicts of interest to report.