Bone marrow mesenchymal stem cells (BM-MSCs) are the best adult stem cells that can be used for autologous regenerative medicine. These cells are easily manipulated for use in cell therapy. Therefore, rapid and simple method for culture MScs is needed. In this paper, we are showing simple and robust method for culturing the stem cells. The cells were flushed from the bone marrow of the thighbones of mice and cultured directly in tissue culture flask. Purification of BMSCs were based on the ability of mesenchymal stem cells to adhere to plastic surfaces. BM-MSCs tested immunocytochemically for CD34, CD44, CD90, and CD105. Results showed a high percentage of positive cells towards CD44, CD90, and CD105, and a negative reactivity towards the hematopoietic stem cells markers CD34. Conclusion, the direct culture method is best to culture high number of pure BM-MSCs in very short time and high viability rate.
Mesenchymal stem cell, Isolation, Culturing, Detection
Mesenchymal Stem cells (MSCs) are differentiated mainly from mesoderm embryonic germ layer give rise to skeletal and connective tissue, and also able to differentiate into neural and myogenic cells [1].
It's also isolated from many adult tissues like stromal bone marrow, marrow aspirates, periosteum, trabecular bone, synovium, skeletal muscle and deciduous teeth and adipose tissue these cells have capacity to differentiate into connective tissue including bone, fat, cartilage and muscle. It has ability to expand in culture while maintaining their multipotency [2,3]. MSCs have plastic adhesion ability to express CD105, CD90, lack expression of CD45, CD34, CD14 or CD11b, CD19 and HLA-DR surface molecules and must differentiation to osteoblasts, adipocytes and chondrocytes in vitro [4].
MSCs derived from umbilical cord have potential to differentiate into multiple lineages of mesoderm in addition to ectodermal and endodermal lineages [5].
All MSC isolation procedure depend on the plastic adherence property of cells a on the surface and start to extended and differentiated into many cell type. Positive selection only selected CD34+ cells were others sub cultured in a 25 cm2 culture flask [6], but different procedure used for their purification like negative isolation in which Adherent cells from adult C57Bl/6J mouse bone marrow were depleted of granulo-monocytic cells and subsequently allowed to grow on fibronectin-coated dishes in presence of fetal bovine serum and growth factors [7]. Therefore, direct method for isolate mouse BM-MSCs and culture in there native environment showing strongly positive for CD44 and CD90, negative CD45 and CD31 [8].
Albino mice used in this study were obtained from the Iraqi center for cancer research and medical genetics/Al-Mustansiriya University. Mice age was from 4-8 week. It was subjected to unified condition of temperature, light and feeding. A bone marrow cell was isolated from the femur of 4-8-week-old male albino mouse with weights ranging from 10-15 g.
The mouse was killed by cervical dislocation, placed the mouse on its back on a cutting board and soaks it with 70% ethanol and 10% iodine solution, followed by making a long transverse cut through the skin in the middle of the abdominal area. Reflected skin from the hindquarters and the hind legs, then remove the muscles and placed the bone in a Petri dish containing medium MEM free serum. A flushing method was used to flush the bone marrow cells from bones using a 1 ml syringe containing 1 ml of growth culture media (MEM) [9].
Freshly isolated whole bone marrow stromal cells were re-suspended in 5 ml growth culture medium MEM supplemented with 20% FBS, 1% Ampicllin/Streptomycin. Cells obtained from three mice (2 femurs and 2 tibias) were seeded in 24 ml tissue culture flasks. The cultured cells were incubation at 37 ℃ and left to adhere 24 hours, non-adherent cells were removed, Mesenchymal stem cells were selected by adherence during first 24 h and maintained in growth culture media. Media changed 3 times a week and when the cultures reached 80-100% confluence [10] with modification.
Passaging of the cells were done in suspension culture at cell density of 8.3 × 106 cell/ml concentration. the first time to passage occurs at 5th after culture, the culture medium was aspirated and the cells were washed three times with MEM free serum and detached by incubation with 1 ml of trypsin-versene for 5-10 minutes at 37 ℃ temperature. Then 5 ml of culture media containing 20% FBS was added and mixed to obtain a single cell suspension.
Method of Daring and Morgan in Freshny [11] was used to measure the cell viability by using trypan blue dye which allows distinguishing between healthy cells with uncompromised membrane integrity (unstained) and dead ones (stained blue).
The following CD markers (primary antibody) for detection of MSCs: (mouse anti-human CD105+, mouse anti-human CD90+, Mouse anti-human CD44+ and Mouse anti goat CD 34- [12].
After MSCs were dispersed with trypsin-versene, and suspended in MEM growth media the cell were re-cultured in multi-well tissue culture plates (8 well) in MEM supplemented with 20% FBS, the plates were incubated at 37 ℃ to allow the cells for development a monolayer of adherent cells within 1-3 days, after that the media was aspirated and the cell were fixed by 4% paraformaldehyde for 10 min. In all steps the sections were placed in the humid chamber and at room temperature (20-25 ℃) and done according to kit manufacturer procedure (Santa Cruze biotechnology, USA). The cells were incubated with 1% hydrogen peroxide for 10-15 minutes and wash with PBS for 5 min three times. Aliquot of 1.5% blocking serum was added to cell section for one hour then discarded. Incubated with 125 µl of diluted primary antibody at a ratio (1:50) for one hour at room temperature or overnight at 4 ℃ every primary anti body manufacturer procedures) then washed about 30 min with 1.2 ml biotinylated secondary antibody added. A liquot of 650 µl of AB enzyme reagent was added to cell section. Three drops of peroxidase substrate was added to cells for 10 min, or until desired stain intensity develops, every previous step end with washing. Hematoxylin stain was added to cell section for 5-10 seconds and immediately washed with distilled water. Finally 1-2 drops of permanent mounting medium was added and examined by light microscopy at magnification powers 40× and 100× [13].
Mice bone marrow isolated from the femurs and tibias of young albino mice by simple and easy flushing all stromal cells method. Then, direct culturing cells in aplastic tissue culture flask, MEM media with 20% FBS. After 24 hours the mesenchymal stem cells have round shape or quasi-circular shapes attached on the surface of flask and other cell like hematopoitic stem cell, still suspended in media. The media was discarded and changed with fresh 20% serum MEM media simply without any additional growth factors.
In this study MSC cultured directly without washing step to preserve cell activity in MEM media at concentration 8.3 × 106 cell/ml and show benefit isolation and growth. After 24 h the media was discarded and freshly media added, this step represent a benefit method for purification of culture from other cell type and dead cell, fresh MEM media added and the growth observed. The attached cell have circular mononeuclear structure, after 48 h the cell begun small spindle structure formed.
This structural shape and sequentional transformation of mesenchymal stem cell similar in all isolation protocol for research in the first day of primary culture [8]. While MSC research used Dulbecco's media alone or with high glucose as a stem cell culture media after washing with media and centrifugation then re-suspended in media, incubated for 72 h of p0 [14].
Bone marrow mesnchymal stem cells are multipotency, ease of purification and amplification, therefore be an ideal stem cell source for cell therapies. Some researchers isolated and maintained MSC in media without additional growth factors together with passage-dependent reseeding following trypsinization [15].
Attached cell represent 80% in the few days after culturing which give rise to mesenchymal stem cell and the other 20% still floating in culture, increasing serum percentage in culture increase the number of cell significantly from 5%, 10%, 15% and 20% [16].
The cultured media changed every 3-4 days and in the third day of culture the cells have small spindle like shape starting propagation. At the end of 5th day of culture the number of cell increase forming colonies distributed in flask, 70-80% field forming in concentration 2.5 × 106 cell/ml, cell have short fibroblast like structure with two polar cell shape and some cell have triangular shape in a homogenized monolayer (Figure 1, Figure 2 and Figure 3).
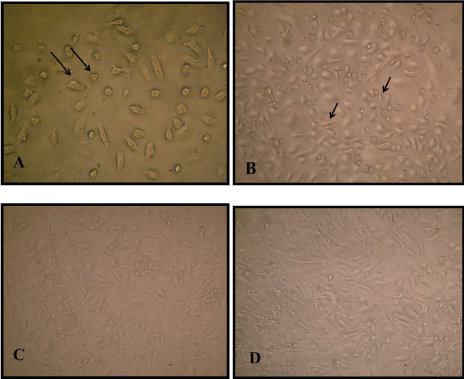 Figure 1: A) Primary culture Mesenchymal stem cells after 24 h showed round attached cell (10 × 20); B) After 48 h culturing MSCs have small spindle like shape or triangular structure (10 × 10); C, D) Monolayer primary MCSs culture (10 × 10).
View Figure 1
Figure 1: A) Primary culture Mesenchymal stem cells after 24 h showed round attached cell (10 × 20); B) After 48 h culturing MSCs have small spindle like shape or triangular structure (10 × 10); C, D) Monolayer primary MCSs culture (10 × 10).
View Figure 1
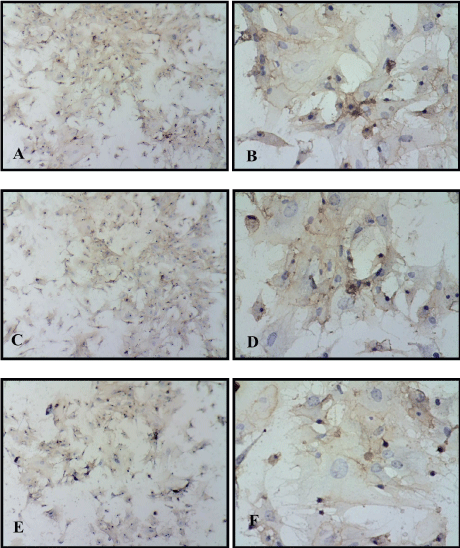 Figure 2: A,B) Immuonocytochemical study of mesenchymal stem cells for CD105 (10 × 20) (10 × 40); C) Positive CD90 (10 × 20) (10 × 40); E,F) Positive for CD44 (10 × 20) (10 × 40) brown of chromogen accumulated. View Figure 2
Figure 2: A,B) Immuonocytochemical study of mesenchymal stem cells for CD105 (10 × 20) (10 × 40); C) Positive CD90 (10 × 20) (10 × 40); E,F) Positive for CD44 (10 × 20) (10 × 40) brown of chromogen accumulated. View Figure 2
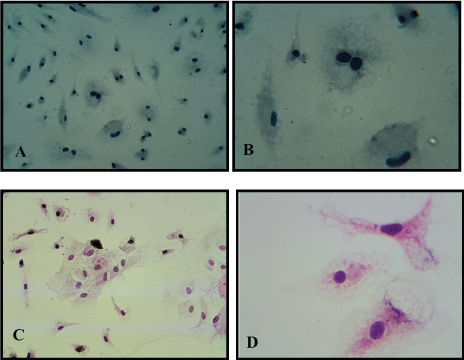 Figure 3: A,B) MSC immuonocytochemical study of mesenchymal stem cells for negative for CD34 (10 × 20) (10 × 40); C,D) Hematoxlyne and eosin stain for MSC (10 × 20) (10 × 40). View Figure 3
Figure 3: A,B) MSC immuonocytochemical study of mesenchymal stem cells for negative for CD34 (10 × 20) (10 × 40); C,D) Hematoxlyne and eosin stain for MSC (10 × 20) (10 × 40). View Figure 3
Mutlak [17], results at the end of 5th day of cultivation, a numerous fibroblast like cells formed and gradually grow to form small individual colonies like fibroblat morphologicaly.
When the cells reached confluent percentage about 70-80% in the flask about 2.5 × 106 cell/ml, they should be sub cultured [18]. Under the same conditions the cell began to propagate and form homogenize layer of mesenchymal stem cell in continues passaging; start from passage 1 used in the differentiation into motor neuron cells.
Scientific work on MSCs morphology and size indicate that the cells changed from small, spindle-like cells in the early passage to large and polygonal types in later passages. Their growth was highest in the third passage, followed by a gradual decrease. There were no special modifications of cell surface antigens or the karyotype of the MSCs from the first to the tenth passage [19].
Bonab, et al. [20] showed that multiple MSC passaging more than 9 passages will reduce the population doubling of cell from 7.7 to 1.2 in the 10th passage and telomere length decreased from 9.19 Kbp to 8.7 kbp in the 9th passage results in differentiation potential dropped from the 6th passage on.
Mesenchymal stem cells phenotypic characteristic detected in culture passage one by culturing passage 0 cell on multi well tissue culture slide, when reached monolayer, fixed with 4% paraformaldehyde and using immunocytochemistry test for detection specific CD markers for MSCs, these CD markers were CD105, CD90 and CD44 as positive markers and CD34 as negative marker and the results are shown in the Figure 2 and Figure 3. Mice bone marrow MSC detected using chromogen accumulation on the secondary antibody of the primary antibody of CD105, CD90 and CD44 showed dark brown color while cell for negative CD34 was still blue, the color of hematoxylin, with no brown stain.
The results matches with The International Society for Cellular Therapy in assuring identity of MSCs using CD70, CD90, and CD105 as positive markers and CD34 as a negative marker [12]. Bone marrow, adipose tissue and umbilical cord MSC have CD markers positive to CD105, CD90 and CD44 using flow cytometry analysis while negative CD34 for all type sources in more than 95% of cells [3].
This project was done by the foundation of Iraqi center for cancer and medical genetic research.