Background: Antibodies towards Preferentially Expressed Antigen in Melanoma (PRAME) have shown promise to discriminate between melanoma in situ (MIS) and benign melanocytic lesions. Paget's disease, extramammary Paget's disease, and squamous cell carcinoma in situ may mimic the morphologic features of MIS, and PRAME expression in these lesions has not yet been described.
Methods: Three cases of Paget's disease, 11 of extramammary Paget's disease, 15 squamous cell carcinoma in situ cases, and two collision tumors (extramammary Paget's disease and squamous cell carcinoma in situ) were entered into this study.
Results: One of 15 cases of squamous cell carcinoma in situ showed focal, weak PRAME positivity. One case of Paget's disease of the breast showed weak, diffuse positivity as did one collision tumor. Five of 11 cases of extramammary Paget's disease displayed positive nuclear PRAME staining with three showing various degrees of cytoplasmic positivity (negative nuclear staining).
Conclusions: Although PRAME has shown utility in the diagnosis of MIS, it is not lineage specific. PRAME shows variable expression in mimickers of MIS, and other immunostains should be used to confirm the diagnosis of pagetoid lesions. Positive PRAME staining does seem to suggest a non-squamous tumor lineage, however.
PRAME, Melanoma in situ, Paget's disease, Extramammary, Squamous cell carcinoma in situ
Melanocytic neoplasms may show atypical features which raise concern for premalignant or malignant biologic behavior. Pagetoid spread, the upward scatter of melanocytes throughout the full thickness of the epidermis, is one such concerning feature. Although it is often seen in benign nevi from special sites such as the thigh or acral locations [1-3] or as a component of sun-damaged skin, [4] it is also a feature which is frequently observed in melanoma in situ (MIS).
Neoplasia involving other cell lineages may also show pagetoid spread [5]. Paget's disease and extramammary Paget's disease are characterized by upward scattering of intraepidermal glandular cells. Squamous cell carcinoma in situ may also show pagetoid spread of dysplastic keratinocytes as individual keratinocytes or in neoplastic nests at all levels of the epidermis. To add further confusion, cells in each of these entities may occasionally have pigment and may be mistaken for MIS [6,7]. Uncommonly, MIS may be amelanotic [8-10].
It is prudent to scrutinize lesions showing concerning degrees of pagetoid spread for attributes that belay cell lineage. Intracytoplasmic mucin may increase suspicion for Paget's disease or extramammary Paget's disease. Intercellular desmosomes and dyskeratotic cells may lead one to suspect a keratinocyte neoplasm such as squamous cell carcinoma in situ [11,12].
Definitive diagnosis of these lesions may involve investigation using immunohistochemistry. Markers such as Melan A/MART1 and SOX-10 predictably stain melanocytes [13] while Carcinoembryonic Antigen (CEA), Ber-EP4, and CK7, among others, can be utilized to highlight glandular cells in lesions of Paget's disease and extramammary Paget's disease [14,15]. Keratinocyte stains such as CK5/6 and p63 reliably mark the keratinocytes of squamous cell carcinoma in situ [16,17].
Immunohistochemistry also plays a role in evaluating melanocytic lesions to determine if they are dysplastic or represent more nefarious MIS. One such stain that has garnered much attention in recent years is PRAME. PRAME is a relatively predictable differentiator of melanocytic lesions as it is expressed in most MIS and invasive melanoma cases and is negative in most dysplastic nevi and benign nevi [18]. We investigated PRAME expression in the mimickers of MIS.
A search of the pathology database at Epiphany Dermatology yielded a total of 31 cases using the term "Paget". These included 15 Pagetoid Squamous Cell Carcinoma in Situ (Figure 1), 2 Collision Tumor (Pagetoid Squamous Cell Carcinoma in Situ and Extramammary Paget's Disease) (Figure 2), 3 Paget's Disease of the Breast, and 11 Extramammary Paget's Disease (Figure 3). Only cases with unequivocal diagnoses were included for this study to determine the frequency of PRAME expression using IHC. A representative sample of 15 melanoma in situ cases were included as a control group. The tumors were reviewed by at least 2 dermatopathologists (M.H. and C.D.C.) with agreement on the diagnoses. Only cases with agreement on the diagnosis were included herein.
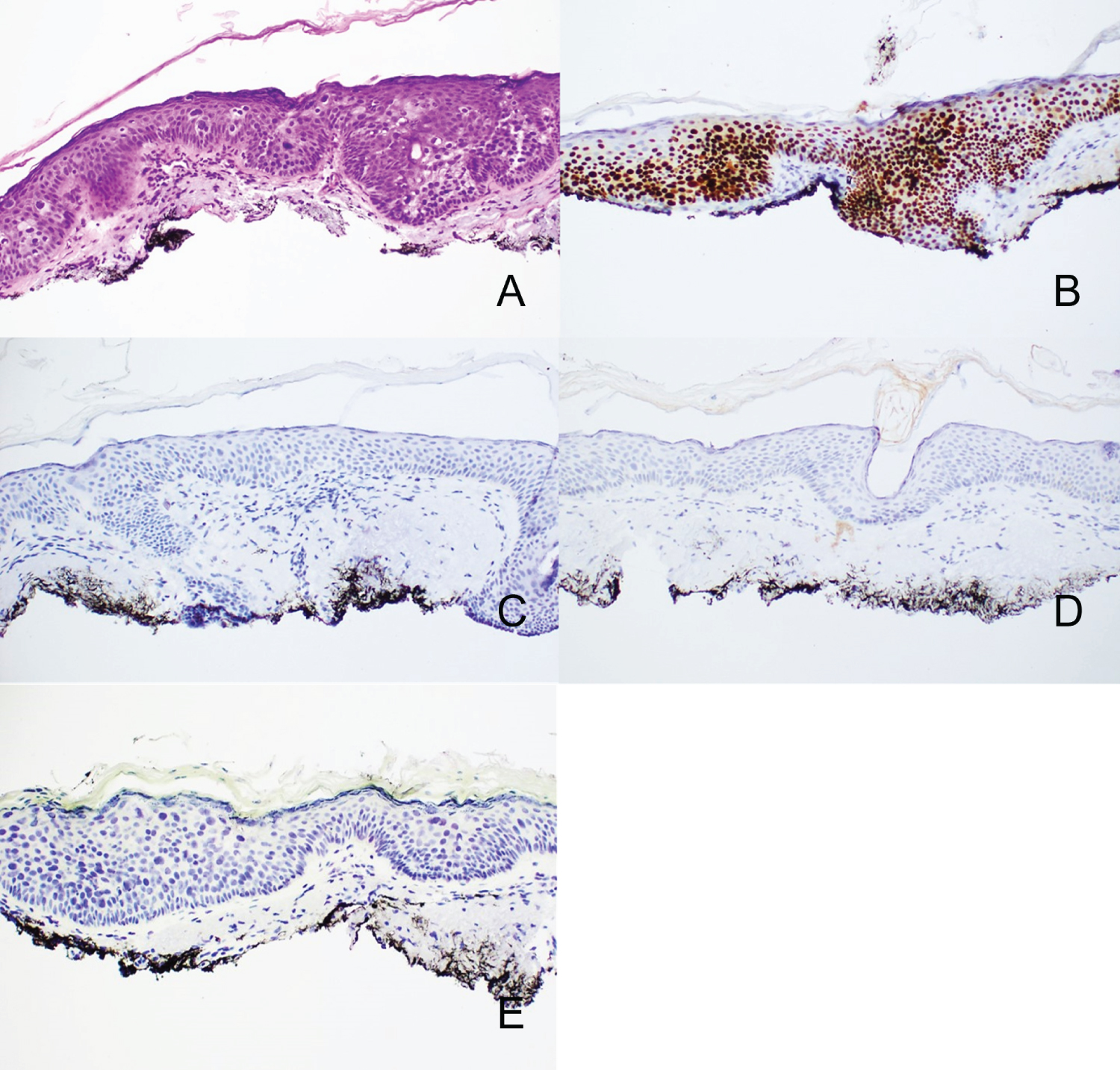 Figure 1: A) Pagetoid Squamous Cell Carcinoma in Situ seen with H&E staining (10x). B) Atypical cells are positive with p63 but negative with C) Ber-EP4 D) CK7 and E) Focal weak intensity PRAME expression is noted in this particular case.
View Figure 1
Figure 1: A) Pagetoid Squamous Cell Carcinoma in Situ seen with H&E staining (10x). B) Atypical cells are positive with p63 but negative with C) Ber-EP4 D) CK7 and E) Focal weak intensity PRAME expression is noted in this particular case.
View Figure 1
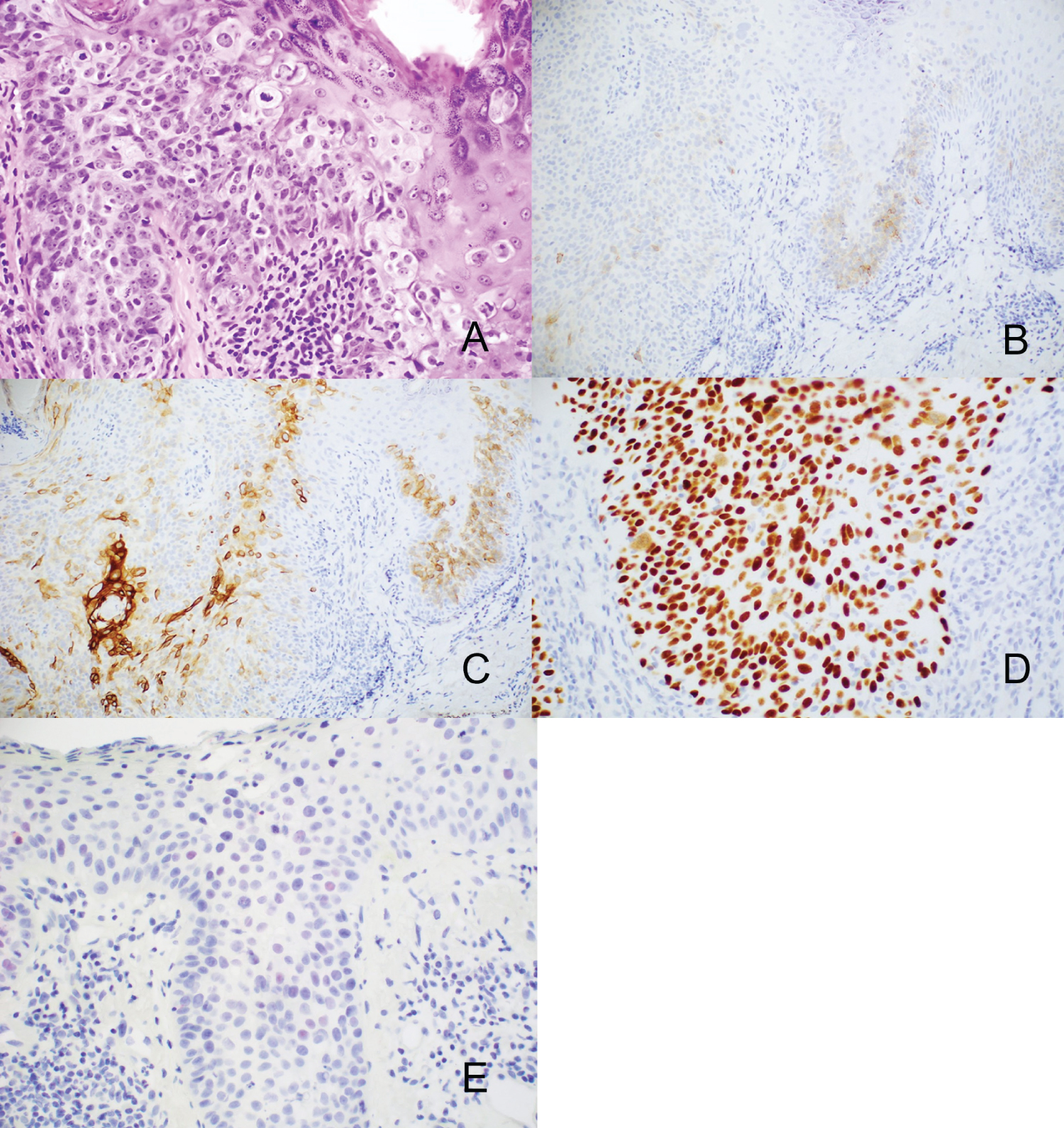 Figure 2: A) Collision tumor of Extramammary Paget's Disease and Squamous Cell Carcinoma in Situ with H & E (10x). Larger cells are Extramammary Paget's Disease, and smaller cells are Squamous Cell Carcinoma in Situ. B) Areas of larger cells are positive with Ber-EP4 and C) CK7, while the smaller atypical cells are positive with D) p63, and the larger cells are negative with p63 E) Focal weak intensity expression of PRAME is seen in a few of the larger nuclei.
View Figure 2
Figure 2: A) Collision tumor of Extramammary Paget's Disease and Squamous Cell Carcinoma in Situ with H & E (10x). Larger cells are Extramammary Paget's Disease, and smaller cells are Squamous Cell Carcinoma in Situ. B) Areas of larger cells are positive with Ber-EP4 and C) CK7, while the smaller atypical cells are positive with D) p63, and the larger cells are negative with p63 E) Focal weak intensity expression of PRAME is seen in a few of the larger nuclei.
View Figure 2
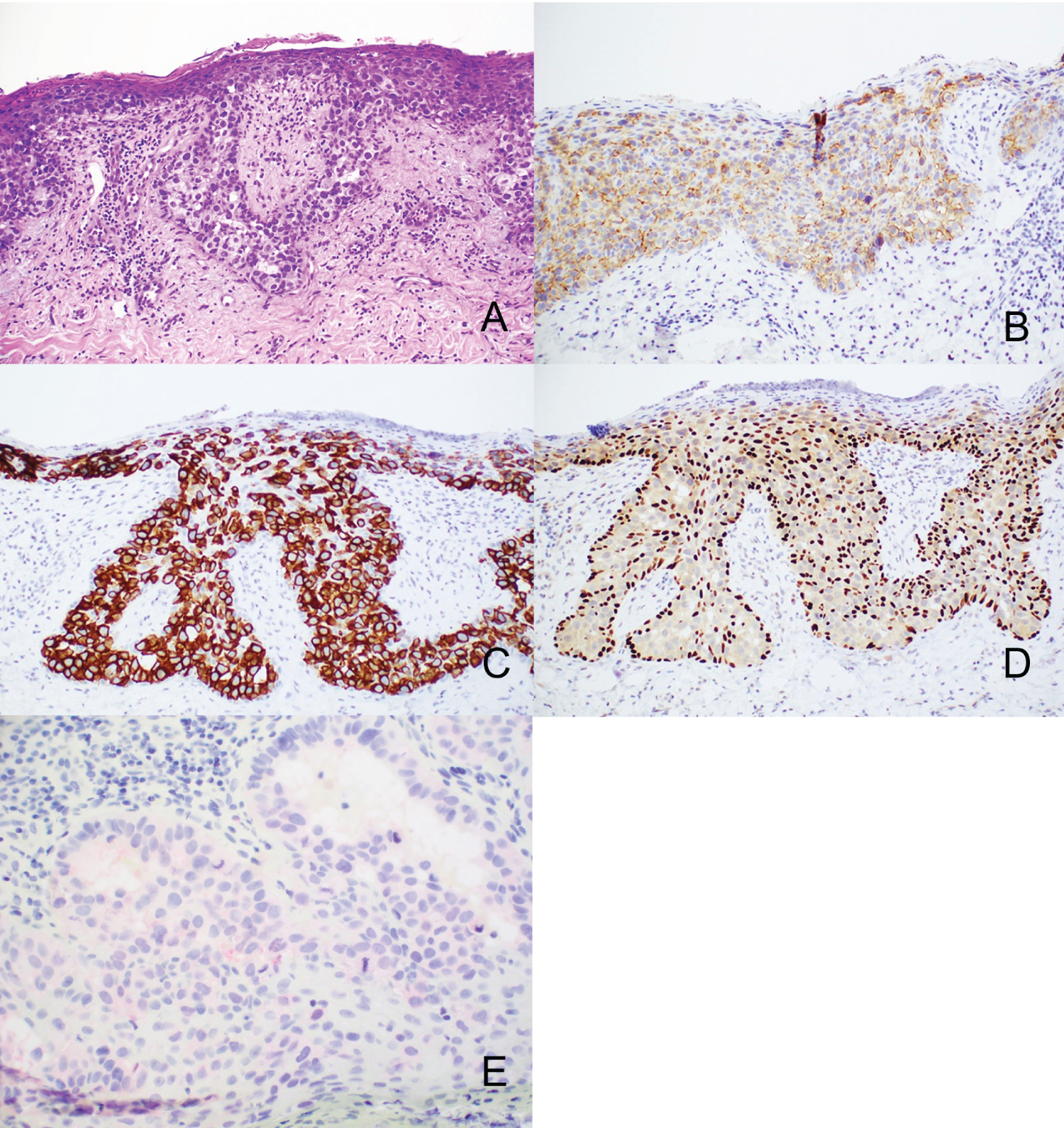 Figure 3: A) Extramammary Paget's Disease seen on H&E (10x). The tumor cells are positive with B) Ber-EP4 and C) CK7. D) p63 is positive in keratinocytes but negative in the larger tumor cells. E) PRAME shows weak cytoplasmic staining.
View Figure 3
Figure 3: A) Extramammary Paget's Disease seen on H&E (10x). The tumor cells are positive with B) Ber-EP4 and C) CK7. D) p63 is positive in keratinocytes but negative in the larger tumor cells. E) PRAME shows weak cytoplasmic staining.
View Figure 3
Five micrometer thick tissue sections were cut from formalin-fixed and paraffin-embedded tissue blocks. A commercially available antibody to PRAME (MAb EPR20330; Biocare Medical) was used on an automated Leica-Bond stainer platform. The staining result was recorded as the percentage of immunoreactive tumor cells with nuclear labeling per total number of tumor cells. Zero indicated no staining at all. Staining of 1%-25% of tumor cells was scored as 1+, 26%-50% of tumor cells was scored as 2+, 51%-75% of tumor cells was designated as 3+, and 76% or higher was recorded as 4+. Intensity of staining in positive nuclei was designated as weak, moderate, or strong.
In total, our patients ranged in age from 42-94 years of age encompassing 18 men and 13 women (Table 1). All cases of Paget's disease of the breast occurred on the nipple-areolar complex. The remaining cases show no particular site predilection although 24/28 of these cases were biopsied from sites of sun-damaged skin.
Table 1: Patient demographics and tumor sites. View Table 1
One case of Paget's disease of the breast showed weak intensity 2+ positivity (Figure 4) while the other two cases were negative (Table 2). Similar findings were present in the collision tumors with one showing 1+, weak positivity and the other being negative. Four cases of extramammary Paget's disease showed 1+, weak staining, one showed 4+, moderate staining, and, interestingly, three showed various degrees of cytoplasmic positivity with negative nuclear staining. The remaining three extramammary Paget's disease cases were negative. Only 1/15 pagetoid squamous cell carcinoma in situ showed any degree of positivity with that being 1+ and weak intensity.
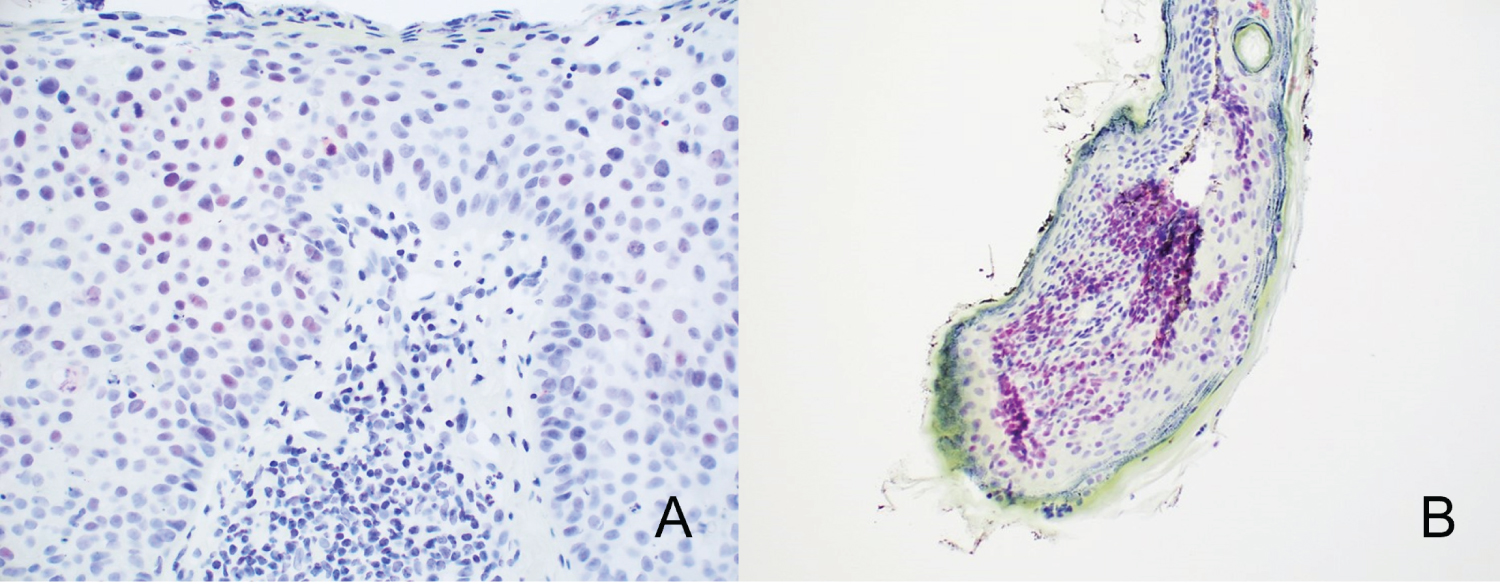 Figure 4: Variable staining is seen in extramammary Paget's disease. A) Foci of weak positive nuclei (pink/red nuclei) set within a background of negative nuclei (blue nuclei). The remainder of the tumor nuclei were negative (10x). B) Moderate intensity staining is seen in nearly all tumor nuclei.
View Figure 4
Figure 4: Variable staining is seen in extramammary Paget's disease. A) Foci of weak positive nuclei (pink/red nuclei) set within a background of negative nuclei (blue nuclei). The remainder of the tumor nuclei were negative (10x). B) Moderate intensity staining is seen in nearly all tumor nuclei.
View Figure 4
Table 2: Number of positive cases and associated intensity of staining. View Table 2
The melanoma in situ control group had 1+ staining in 2 cases, 2+ expression in 2 cases, 3+ positivity in 6 cases, 4+ staining in 2 cases (Figure 5). 3 cases of MIS were negative for PRAME.
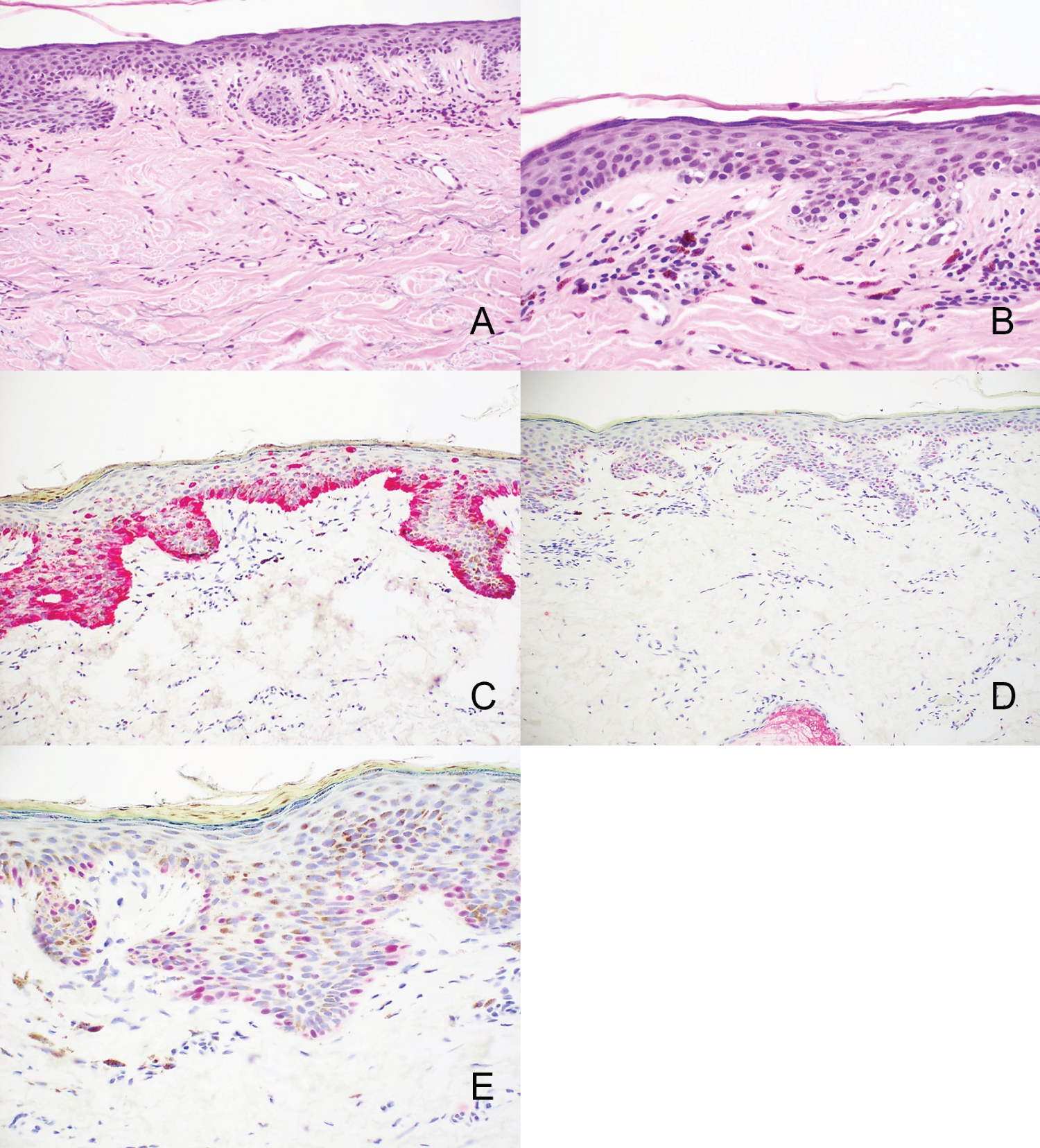 Figure 5: Melanoma in situ. A, B) Low and higher power views show a largely contiguous basal layer melanocyte proliferation with pagetoid spread. C) Melan A staining highlights all the melanocytes. D) Within this same lesion, there are areas of weaker intensity PRAME staining above a strongly staining internal positive control (sebaceous gland). E) Other areas of this lesion show more intense PRAME staining.
View Figure 5
Figure 5: Melanoma in situ. A, B) Low and higher power views show a largely contiguous basal layer melanocyte proliferation with pagetoid spread. C) Melan A staining highlights all the melanocytes. D) Within this same lesion, there are areas of weaker intensity PRAME staining above a strongly staining internal positive control (sebaceous gland). E) Other areas of this lesion show more intense PRAME staining.
View Figure 5
Paget's disease of the breast, extramammary Paget's disease, and pagetoid squamous cell carcinoma in situ share overlapping morphologic features with MIS. This frequently necessitates employment of immunohistochemical stains to confirm cell lineage for accurate diagnosis, including stains to evaluate for MIS. PRAME is one marker that can be used in the evaluation of melanocytic neoplasms, but its expression pattern and/or aptitude to discriminate between MIS and its mimickers has not yet been described.
Our results show that PRAME is not a reliable marker to differentiate between melanoma in situ and Paget's disease/extramammary Paget's disease. Although the majority of positivity in these cases was only focal and weak, one of these cases showed more diffuse positivity with moderate intensity. PRAME performs slightly better when discriminating pagetoid squamous cell carcinoma in situ with only 1 of 15 cases showing focal, weak staining and the remaining cases being negative. This is consistent with the previously reported pattern of PRAME expression in squamous cell carcinoma [19].
PRAME is not specific to a cell lineage and has been described in tumors of non-melanocytic origin including colon adenocarcinoma, cellular neurothekeoma, and some lymphomas [20-22]. Not surprisingly, expression of PRAME was identified in several of our non-melanocytic lesions. Such patterns of expression do not allow for using PRAME to reliably differentiate between MIS, Paget's disease of the breast, extramammary Paget's disease, and squamous cell carcinoma in situ, especially since PRAME may also show variable qualitative and quantitative expression in MIS.
PRAME is a relatively recently adopted component of the pathologist's armamentarium to use when examining melanocytic lesions. Expression of PRAME has not yet been evaluated in common mimics of MIS. Based on these results, PRAME staining in these entities is largely inconsequential given the small number of positive tumor nuclei observed. Therefore, PRAME positivity in the absence of diagnostic melanocyte staining using more traditional markers (Melan A/MART1, SOX-10, etc.) should be viewed as nonspecific staining or a proverbial "red herring." Moreover, although all but one case of pagetoid squamous cell carcinoma in situ were negative for PRAME, PRAME expression should not be used as a mechanism to differentiate between MIS and pagetoid squamous cell carcinoma in situ nor should it be used to help differentiate between pagetoid squamous cell carcinoma in situ and Paget's disease/extramammary Paget's disease.
Thank you to Kelvin Hayford, Regional Laboratory Manager at Epiphany Dermatology for completing the technical portion of these stains thereby enabling this publication.
None of the authors have a conflict of interest to report.