Normal intestinal endocrine cells are positive for chromogranin A (CgA) and synaptophysin (SPY) immunohistochemical staining, which are the most commonly used markers for neuroendocrine cells and their tumors. CgA is co-stored and co-secreted with catecholamine in carcinoids.
CgA and SPY immunostaining was performed with forty intestinal carcinoids, consisting of 4 duodenal, 14 ileal, 5 appendiceal and 17 colorectal carcinoids.
Duodenal and ileal carcinoids were strongly positive for CgA, with or without invasion through intestinal wall. Small appendiceal carcinoids were weakly immunostained for CgA and SPY. Colorectal carcinoids were low-grade tumors especially those of small polypoid tumors (< 1.0 cm) with CgA-negative staining while larger tumors (> 2.0 cm) infiltrating through wall were more aggressive with often CgA positive staining. CgA positive carcinoids of duodenum, small intestine and colorectum of larger than 2 cm are more aggressive than CgA negative colorectal tumors, majority of the latter was less than 2 cm. Midgut carcinoids of duodenal, ileal and ascending colon are strongly positive for CgA associated with high serum CgA levels while hindgut carcinoids of rectum, those of small polypoid tumors are negative for CgA but positive for SPY immunostaining.
Thus, CgA-positive immunostaining may be used as an independent marker for potential malignancy for gastroenteropancreatic neuroendocrine tumors regarding the location and sizes of tumors. Some colorectal carcinoids were negative for CgA but were all positive for SPY while some foregut carcinoids with positive CgA staining were negative for SPY immunostaining. Thus, CgA and SPY immunostaining provides significant information in carcinoids.
Carcinoid, Chromogranin A, Immunohistochemistry, Synaptophysin
Classic intestinal carcinoids are slow growing, indolent malignant tumors and the most common location is the small intestine (38%), followed by rectum (34%), colon (16%), stomach (11%) and unknown sites (1%) [1]. In small intestine, ileum is the most common site, followed by jejunum and distal duodenum and a 5-year survival is 85% if confined to intestinal wall versus 5% if invaded through serosa [2-4]. In large intestine, rectosigmoid (hindgut) is the most common site, followed by cecum and transverse colon (midgut) [3,5-7]. Colorectal carcinoids are usually of low grade, slow-growing tumor with a 5 year-survival of 90% [8]. The intestinal carcinoid derives from disseminated endocrine cells, which consist of less than 1% of the mucosa and are normally distributed at the surface or base of glandular epithelial cells, especially at the crypts of the small intestine and colorectum and contain secretary granules, from which colorectal carcinoids release various peptide hormones without presenting hormone specific clinical symptoms [9,10]. For immunohistochemical staining, there is positive staining for both epithelial and neuroendocrine markers [10]. For epithelial markers, CEA is positive at the apical or luminal location [10] and CK7 is positive in almost 10% of the tumors and CK20 is positive in 25% [11]. The positive neuroendocrine markers include chromogranin A (CgA), synaptophysin (SPY), neuron specific enolase, Leu 7, PGP 9.5 and others [12,13]. At a typical pathology laboratory, CgA and SPY are routinely used for carcinoids since CgA and SPY and are the two most widely used immunohistochemical markers for neuroendocrine cells and their tumors [14,15]. The granin family consists of eight granin peptides, chromogranin A, B, C, secretogranin III, IV, V, VI and VEGF [15]. Under immunoelectron microscopy, CgA was confined to the secretary granules, especially in the periphery of the granules [16], while SPY immunostaining was diffusely in the cytoplasm [16]. CgA is widely positive for neuroendocrine cells including those of intestines, thyroid C-cells, parathyroid chief cells, anterior pituitary cells, pancreatic endocrine cells and others [12]. In the endocrine pancreas, β-islet cells are weakly to moderately positive for CgA while non-β cells, such as α-, δ- and pancreatic polypeptide (PP) cells are strongly positive for CgA [14]. CgA and SPY are co-localized in the endocrine cell but the former occurs more basically in the cytoplasm throughout gastrointestinal tract endocrine cells, corresponding to the location of neurosecretory granules while SPY occurs more diffusely outside the secretary granules throughout the endocrine cell cytoplasm, corresponding to the diffuse distribution of synaptic vesicles (SVs) in the cytoplasm [16]. SPY belongs to a family of related vesicle proteins present in small SVs, which includes Synatopagmin (p65), SNAP-25, SNAP-receptor (SNARE), Sytataxin, Rab3A and others [14,16]. SPY was one of the first synaptic proteins identified, yet its structure -function relationship has remained elusive since ascribing a clear role in the SV cycle is missing [17,18] but SPY may play a role in the SV cycle [16-18]. Despite that both CgA and SPY have been widely used as neuroendocrine markers in neuroendocrine tumors, including intestinal carcinoids, no detail comparative immunohistochemical study has been reported for intestinal carcinoids, and this report deals with the comparative immunohistochemical staining in the intestinal carcinoids regarding the location, sizes of tumors and immunohistochemical staining for CgA and SPY in the carcinoid tumor cells. All gastrointestinal carcinoids are reportedly mostly positively stained for SPY while CgA immunostaining has been variable depending on the tumor locations in the intestines: foregut tumors including stomach, first portion of duodenum and pancreas are 88% positive and midgut tumors including small intestines, appendix, ascending colon and proximal transverse colon are 100% positive and hindgut tumors consisting of distal transverse colon, descending colon and rectum are 60% positive for CgA including typical and atypical carcinoids while SPY was reportedly positively immunostained 100% of gastrointestinal and pulmonary carcinoids [19,20]. More recently, however, Portla-Gomes, et al. reported that hindgut carcinoids were weakly immunostained for SPY but no staining for CgA [21].
All cases of intestinal carcinoids were from the University of Kansas Medical Center, collected between 1975 and 2001. A total of 40 cases were included in this study consisting of 4 duodenal carcinoids presenting with Zollinger-Ellison syndrome, 14 ileal carcinoids, 5 appendiceal carcinoids, 11 colonic carcinoids (5 ascending colonic, 1 transverse colonic, 1 descending colonic and 4 sigmoid colonic) and 6 rectal carcinoids. All the tumor tissues with adjacent normal tissues were fixed in buffered formalin and embedded in paraffin. The archival paraffin blocks were freshly sectioned and the paraffin sections were immunostained within two weeks after sectioning. The deparaffinized sections were treated with antigen retrieval procedure using citrate buffer pH 6.2 and sections were immunostained for CgA using monoclonal anti-CgA (Dako, clone DAK-A3, Santa Clara, CA) and for SPY using rabbit polyclonal anti-SPY (Cell Marque, Cat. 336-76, Rocklin, CA) at 1: 100 dilution. The immunostaining was performed with 20 sections each batch of staining to obtain a good comparative staining intensity to compare. The adjacent normal intestinal mucosa was used as the internal controls for immunostaining set for +++ for CgA and +++ for SPY immunostaining and the less immunostaining was listed as ++ and + and - as negative staining. The SPY immunostaining was relatively weaker than that of CgA immunostaining in our hands. All the tumors were well-differentiated neuroendocrine tumors using the WHO classification [22]. All carcinoid cases were positively immunostained for at least one of the two CgA and SPY markers. Histopathological endocrine patterns were classified into tubular, trabecular, lobular, globular and solid pattern.
Majority of intestinal carcinoids were trabecular-lobular histologic pattern in smaller tumors (< 2.0 cm) and a mixture of solid pattern in the mid-portion and trabecular and/or lobular at the periphery of tumors in larger tumors (> 2.0 cm) (Table 1 and Table 2). The duodenal carcinoids were all gastrinomas, which clinically presented with severe peptic ulcer, Zollinger-Ellison syndrome and the tumors were located submucosally in the distal duodenum, all less than 2 cm in size and were strongly positive to CgA and SPY (Figure 1A and Figure 1B, Table 1). The majority were trabecular pattern admixed with solid pattern (Table 1). Among 12 primary small intestinal carcinoids, 9 cases were smaller than 2 cm (Table 1). Small intestinal carcinoids from ileum were all strongly positive for CgA and weaker or negative for SPY in 10 cases (Figure 1C and Figure 1D). One metastatic liver lesion, Case 4 was strongly positive for CgA and SPY while invading individual tumor cells in the sinusoids were strongly positive for CgA but negative for SPY (Figure 1E and Figure 1F). Thus, SPY immunostaining did not immunostain invading individual tumor cells in the sinusoids (Figure 1E and Figure 1F, Table 1). Appendiceal carcinoids were incidentally found in the microscopic sections of the appendectomy specimen at the tip of the appendiceal submucosa and smooth muscle, measuring 0.2 - 1.0 cm and were mostly tubular pattern and the tumor cells were focally positive for CgA and weakly positive for SPY (Figure 1G and Figure 1H, Table 1). Among 17 cases of colorectal carcinoids, 5 cases were from ascending colon, one case from transverse colon, 4 cases from sigmoid colon and 6 cases were from rectum (Table 2). Among 16 primary colorectal carcinoids, nine cases were ≤ 0.5 cm, consisting of six trabecular/lobular pattern and two solid pattern while in seven larger cases (≥ 1 cm), two cases were solid pattern and five cases were lobular pattern admixed with solid or trabecular pattern (Table 2). Among three cases larger than 2 cm, one case was strongly positive for CgA and two cases were negative for CgA (Table 2). Among 5 cases of ascending colon and one case each of transverse and descending colon, all seven cases were weakly to strongly positive for CgA and were weakly to strongly positive for SPY (Table 2). Among 4 cases of sigmodal carcinoids, 2 cases were strongly positive for CgA and two small tumors were negative for CgA, and three cases were moderately to strongly positive for SPY (Table 2). Case 10, 0.2 cm in size was the smallest tumor and was negative for CgA but moderately positive for SPY (Figure 2A and Figure 2B). Case 12, 0.4 cm in size was negative for CgA and weakly positive for SPY (Figure 2C and Figure 2D). Case 3 of polypoid carcinoid (1.5 × 1.6 cm) from ascending colon was strongly positive for CgA and SPY (Figure 2E and Figure 2H, Table 2) while all four small, rectal tumors, Cases 12, 13, 16 and 17 (≤ 1.0 cm) were negative for CgA and positive for SPY (Table 2). Case 6 of transverse colon infiltrated through the colonic wall was strongly positive for CgA in < 1% of tumor cells and strongly and diffusely positive for SPY (Figure 3A and Figure 3B, Table 2). This tumor invaded deeper in the colonic muscle and were < 5% of the tumor cells strongly positive for CgA and strongly positive for SPY (Figure 3E and Figure 3F, Table 2). Among 6 cases of rectal carcinoids, all six cases were negative for CgA including a large tumor, Case 13 (4.8 × 2.3 × 2.3 cm) and all cases were weakly to strongly positive for SPY.
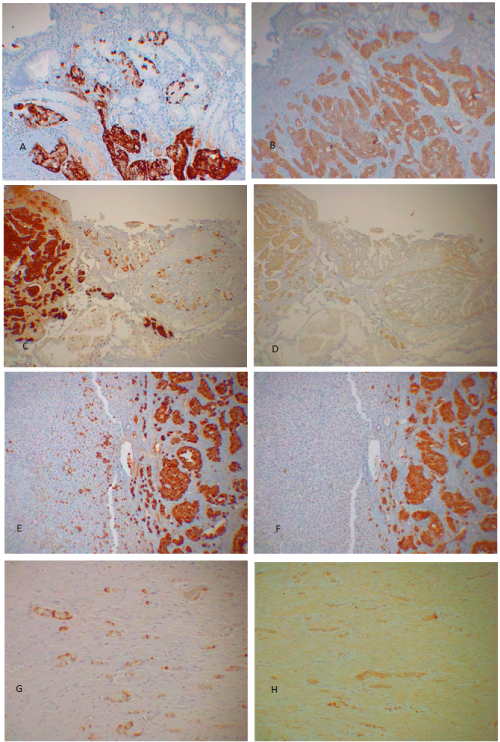 Figure 1: Duodenal gastrinoma, Case 2 (A and B), Ileal carcinoid, Case 8 (C and D), metastatic ileal carcinoid to the liver, Case 4 (E and F) and appendiceal carcinoid, Case 4 (G and H). Gastrinoma, Case 2 invaded into both surface mucosa and deep into the muscle layer in mixed trabecular-solid pattern and were strongly positive for both CgA and SPY (A and B). Ileal carcinoid, Case 8 was solid pattern, which invaded into the intestinal wall and were strongly positive for CgA and weakly positive for SPY (C and D). Another ileal carcinoid, Case 4 was a metastatic tumor to the liver, which was strongly positive for CgA including individual invading tumor cells in the sinusoid while these invading cells were not positively immunostained for SPY despite strong positive staining in the main metastatic tumor (E and F). Appendiceal carcinoid, Case 4 was mixed tubular-globular pattern and was partially positive for CgA in the tumor cell cytoplasm and weakly and diffusely positive for SPY (G and H). A, C, E and G: Immunostained for CgA; B, D, F and H: Immunostained for SPY. View Figure 1
Figure 1: Duodenal gastrinoma, Case 2 (A and B), Ileal carcinoid, Case 8 (C and D), metastatic ileal carcinoid to the liver, Case 4 (E and F) and appendiceal carcinoid, Case 4 (G and H). Gastrinoma, Case 2 invaded into both surface mucosa and deep into the muscle layer in mixed trabecular-solid pattern and were strongly positive for both CgA and SPY (A and B). Ileal carcinoid, Case 8 was solid pattern, which invaded into the intestinal wall and were strongly positive for CgA and weakly positive for SPY (C and D). Another ileal carcinoid, Case 4 was a metastatic tumor to the liver, which was strongly positive for CgA including individual invading tumor cells in the sinusoid while these invading cells were not positively immunostained for SPY despite strong positive staining in the main metastatic tumor (E and F). Appendiceal carcinoid, Case 4 was mixed tubular-globular pattern and was partially positive for CgA in the tumor cell cytoplasm and weakly and diffusely positive for SPY (G and H). A, C, E and G: Immunostained for CgA; B, D, F and H: Immunostained for SPY. View Figure 1
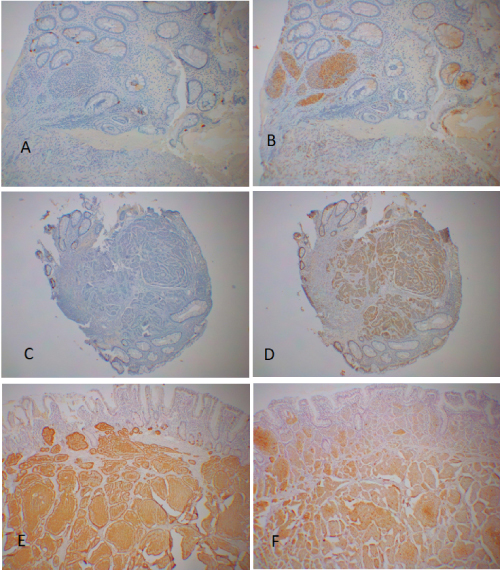 Figure 2: Colorectal carcinoids, small sizes. Case 10, 0.2 cm in size from sigmoid colon, was solid pattern and was negative for CgA (A) and was strongly positive for SPY (B). Case 12, 0.4 cm in size from rectum, was trabecular pattern, which was negative for CgA (C) in the presence of CgA-positive cells in the normal mucosa and weakly positive for SPY (D). Case 3, 1.5 × 1.0 cm in size from ascending colon, was lobular-solid pattern, which was strongly positive for both CgA (E) and SPY (F). A, C and E: Immunostained for CgA; B, D and E: Immunostained for SPY. View Figure 2
Figure 2: Colorectal carcinoids, small sizes. Case 10, 0.2 cm in size from sigmoid colon, was solid pattern and was negative for CgA (A) and was strongly positive for SPY (B). Case 12, 0.4 cm in size from rectum, was trabecular pattern, which was negative for CgA (C) in the presence of CgA-positive cells in the normal mucosa and weakly positive for SPY (D). Case 3, 1.5 × 1.0 cm in size from ascending colon, was lobular-solid pattern, which was strongly positive for both CgA (E) and SPY (F). A, C and E: Immunostained for CgA; B, D and E: Immunostained for SPY. View Figure 2
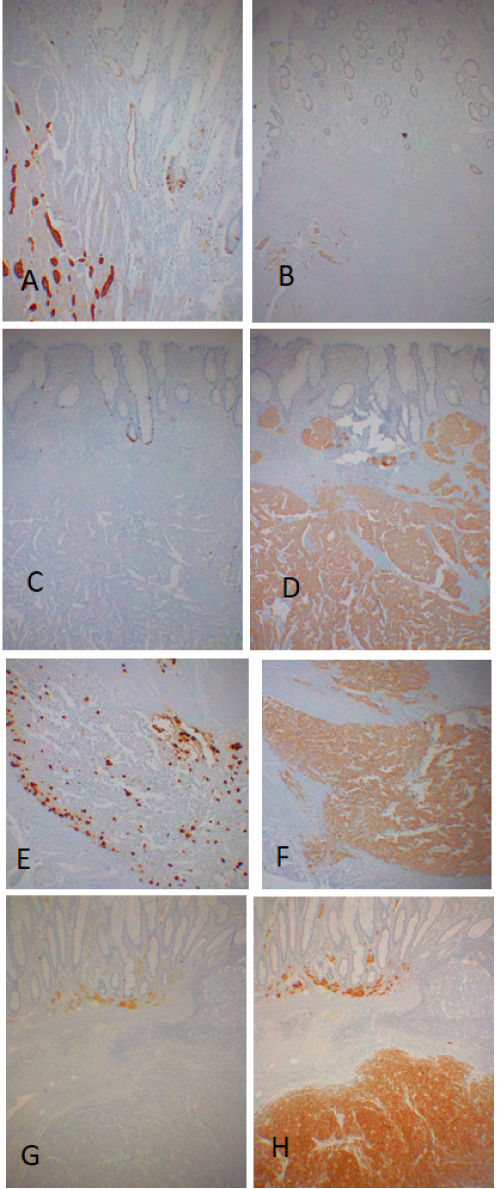 Figure 3: Colorectal carcinoids, large sizes. Case 6 from transverse colon was lobular pattern and was strongly positive for CgA (A) and weakly positive for SPY (B). Case 9 from sigmoid colon was mixed lobular-solid pattern and was positive for CgA in 1% of the tumor cells (C) which was strongly and diffusely positive for SPY (D). The invading tumor cells in the muscular wall from the same case was > 10% positive for CgA (E) and strongly positive for SPY (F). Case 14 from rectum was solid pattern and was negative for CgA (G) but strongly positive for SPY (H). A, C, E and G: Immunostained for CgA; B, D, F and H: Immunostained for SPY. View Figure 3
Figure 3: Colorectal carcinoids, large sizes. Case 6 from transverse colon was lobular pattern and was strongly positive for CgA (A) and weakly positive for SPY (B). Case 9 from sigmoid colon was mixed lobular-solid pattern and was positive for CgA in 1% of the tumor cells (C) which was strongly and diffusely positive for SPY (D). The invading tumor cells in the muscular wall from the same case was > 10% positive for CgA (E) and strongly positive for SPY (F). Case 14 from rectum was solid pattern and was negative for CgA (G) but strongly positive for SPY (H). A, C, E and G: Immunostained for CgA; B, D, F and H: Immunostained for SPY. View Figure 3
Table 1: Carcinoids of duodenum, small intestine and appendix. Chromogranin A and synaptophysin Immunohistochemical Staining. View Table 1
Table 2: Carcinoids of colon and rectum. Chromogranin A and synaptophysin immunohistochemical staining. View Table 2
Midgut carcinoids usually present clinically with the carcinoid syndrome including flushes, diarrhea, bronchial constriction and right heart failure due to serotonins, tacykinins, bradykinins and prostaglandin secretion [19]. Appendiceal carcinoids are considered as benign if the tumors are less than 2 cm including five cases here, which do not present clinical symptoms [15]. Hindgut carcinoids are often non-functioning, slow-growing tumors, although they may produce PYY, HCG-α and -β without presenting hormone-related clinical symptoms [23,24]. Hindgut carcinoids have a longer than 80 months of doubling time compared to estimated 109 days of doubling time for colorectal adenocarcinomas [25,26]. As compared to colonic adenocarcinomas, colorectal carcinoids appear to be generally smaller than adenocarcinomas at the time of diagnosis, thus they follow better prognosis thanks to early detection by colonoscopy. Eighteen cases of mid-gut carcinoids consisting of 4 duodenal gastrinomas and 14 ileal carcinoids were strongly positive for CgA (Table 1). All four duodenal carcinoids were strongly positive for both CgA and SPY (Table 1) where gastrin is co-secreted with CgA in the blood [27]. All 14 Ileal carcinoids were strongly positive for CgA while 7 cases were weakly to strongly positive for SPY, and 7 cases were negative for SPY (Table 1). Thus, all small intestinal carcinoids were strongly positive for CgA while 2 cases were strongly positive for SPY (Table 1). This strong CgA immunostaining for ileal carcinoids corresponds to co-secretion of CgA and catecholamines by the carcinoids [28].
These small intestinal carcinoids were aggressive and were positive for CgA, supporting that CgA positive staining may be used as a marker of possible biological malignancy. All five appendiceal carcinoids were incidentally found in the microscopic section of the appendectomy specimen within the submucosa and smooth muscle layer without extension to serosa and were all focally positive for CgA and weakly positive for SPY in tubular histology, supporting its non-aggressive nature (Table 1). Among 16 cases of primary colorectal carcinoids, only two cases were larger than 2 cm and six of nine small tumors (≤ 0.5 cm) were negative for CgA but six cases were positive for SPY (Table 2). For clinicopathological aspects, colonic carcinoids may be divided into 1) midgut carcinoids from ascending and proximal transverse colon and 2) hindgut carcinoids from sigmoid colon and rectum. All five midgut colonic carcinoids were positive for CgA including Case 6 tumor, 2.0 x 1.5 x 1.0 cm (Table 2). Regarding hindgut carcinoids, two of four sigmoid colon tumors were positive for CgA, but other two cases were negative for CgA (Table 2). One transverse colonic carcinoid, Case 6, revealed > 1% CgA -positive at the surface while invading, intramuscular tumor cells were > 10% positive for CgA, suggesting the latter being aggressive tumor (Figure 2, Table 2). A large rectal carcinoid, Case 13, was negative for CgA as an exception supporting both CgA-positive and -negative tumors are potentially malignant (Table 2). There was consistently strong CgA immunostaining for the midgut carcinoids including gastrinomas of the distal duodenum and ileal carcinoids (Table 1). Compared between the midgut colonic carcinoids from ascending and transverse colon and the hindgut colonic carcinoids from sigmoid colon and rectum, midgut colonic carcinoids were more CgA staining than smaller hindgut colonic carcinoids perhaps due to a difference between larger and smaller carcinoid sizes and a study with larger samples of different sizes of colonic carcinoids is warranted. About the sizes of colorectal carcinoids, smaller tumors less than 1 cm tended to be negative for CgA while colonic carcinoids larger than 2 cm tended to be mostly positive for CgA (Table 2) corresponding to the biological malignancy of the tumors. Regardless of the sizes of the tumors, all colorectal carcinoids were positive for SPY (Table 2). In addition to immunohistochemical CgA staining, there is a good correlation between serum CgA levels and prognosis in carcinoid-harboring patients. In midgut carcinoids, which are strongly positive for CgA, the median survival of patients with serum CgA > 5 µg/ml was 33 months compared to 57 months in patients with serum CgA level < 5 µg/ml [29]. Patients harboring midgut carcinoid with elevated serum CgA and liver metastasis were associated with significantly shorter survival when treated with a long-acting somatostatin analog while there was no correlation between survival and concentration of urinal 5-hydroxyindoleacetic acid (5-HIAA), the break down product of serotonin [15,29,30]. The highest serum CgA levels were reported in patients with small intestinal carcinoid, up to 200 times above the normal, correlating with the strong immunostaining for CgA in these carcinoids [15]. An elevated serum CgA level can be an early sign of carcinoids of the foregut and midgut and appears to be a better marker than the assessment of urinal 5-HIAA and/or circulating serotonin concentrations [25]. There is a strong correlation between the serum CgA levels compared to 5-HIAA and general condition and well-being of the patient, which makes serum CgA as a first-line marker for small intestinal carcinoids [23]. The highest serum CgA levels were reported in small intestinal carcinoids, particularly in cases presenting clinical carcinoid syndrome [15,28,30,31]. Significantly higher serum CgA levels were associated with disseminated carcinoids rather than the limited cases [15]. An exception was gastrinoma, in which serum CgA was high in the absence of liver metastasis as also supported by strong CgA immunostaining in the tumor in this study as well [24-26,30,32]. Thus, the serum CgA level is an independent marker for patients with carcinoids yet it is not specific since high serum levels are recorded in gastric, renal and heart conditions [15]. Nevertheless, elevated serum CgA levels were reported in 100% of gastrinomas, 89% of pheochromocytomas, 80% of small intestinal carcinoids, 69% of non-functioning pancreatic endocrine tumors (PETs) and 50% in medullary thyroid carcinomas [15] corresponding to all these neuroendocrine tumors positive for CgA immunostaining. There has not been reported correlative studies of tissue levels of CgA by directly measuring CgA with the tumor tissue extracts as compared to the serum CgA levels and a correlative study of CgA concentration of tumor tissue and serum levels is warranted. The significance of CgA in carcinoids is that CgA is the major soluble protein co-stored and co-secreted with catecholamines by exocytosis from CgA positive, catecholamines producing tissues like carcinoids [28].
The SVs, spheres of 40 nm in diameter, that store and release classic neurotransmitters such as acetylcholine, norepinephrine, serotonin, GABA, glycine, histamine and glutamate and do not contain usual secretory granules [16-19]. Although function of SPY is elusive, but it is involved in trafficking VAP2 back to SV during endocytosis and appears to play SV cycle [18]. CgA immunostaining is stable even in the acutely infarcted pancreatic endocrine tumor lesion by preserving secretory granules while SPY immunostaining is unstable being negative in the acutely infarcted lesion. It was also shown in this study that infiltrating individual tumor cells were positive for CgA immunostaining in the hepatic sinusoid, which were negative for SPY immunostaining (Figure 1E and Figure 1F). Thus, CgA immunostaining for secretory granules appear to be much more stable and reliable than that of SPY immunostaining. Using recently available more specific anti-SPY, there are some intestinal carcinoids which are negative for SPY as shown in this study (Table 1 and Table 2). Acutely infarcted tumors and invading individual tumor cells in the hepatic sinusoids were negatively immunostained for SPY (Figure 1F) perhaps due to infarction and ischemia of tumor tissue and in other conditions as well. Using recently available more specific anti-SPY antibody, there are some intestinal carcinoids which are negative for SPY as shown in this study (Table 1 and Table 2) as compared to the previous study using less specific anti-SPY antibody, showing positive immunostaining in the granny photos from all intestinal and pulmonary carcinoids [19]. The SVs of the readily releasable pool in the synapses are docked to the cell membrane and release neurotransmitters from SVs through endocytosis on stimulation in a similar mode of secretory granules secretion [5,10]. It has been suggested that neuroendocrine cells including pancreatic islet cells may secrete pancreatic peptide hormones through mostly exocytosis of secretory granules fusing with the cell membrane, which represents the second phase of insulin secretion while the early spike of insulin secretion may be secreted through SVs endocytosis by sympathetic nerve since neuroendocrine cells are equipped with both secretory granules for exostosis in a typical peptide hormone secretory mechanism and also with SVs through endocytosis, the latter are the main secretory system for neurotransmitter, which takes place instantaneously in a matter of split-seconds [5].
I want to thank the Department of Pathology, University of Kansas Medical Center for allowing me to use tissues from the Medical Center collected during my tenure there from 1975 to 2002.
There is no conflicts of interest.