Lymphomas of the gastrointestinal tract are very rare. Primary and secondary oesophageal lymphoma account for less than 1% of all gastrointestinal lymphomas. Secondary oesophageal involvement is by contiguous spread from the stomach or surrounding lymph nodes. Patients with oesophageal lymphoma have varying presentations and complications which include perforation with a tracheo-oesophageal fistula, mass and haemorrhage.
We present a case of recurrence of Hodgkin lymphoma involving the oesophagus and the surrounding lymph nodes. A 75-year-old male presented with dysphagia. He was diagnosed three years ago with Hodgkin lymphoma stage 2A and was managed by chemotherapy. Barium swallow was performed for the present complaint which showed a 12 mm lesion situated 5 cm above the cardio-oesophageal junction. Urgent gastroscopy was done, and the lesion was biopsied. The PET scan revealed FDG avid oesophageal malignancy with probable FDG avid nodal disease around the proximal lesser curvature of the stomach.
Gastrointestinal lymphoma, Oesophageal lymphoma, Hodgkin Lymphoma
Most of the oesophageal malignancies are of epithelial origin [1]. Lymphoma accounts for less than 0.5% of oesophageal malignancies [2]. The gastrointestinal tract (GIT) is the most commonly involved extranodal site [2]. The gastrointestinal involvement can be either primary or secondary. Gastrointestinal involvement by lymphomas occurs in 20% of patients and it involves mainly the stomach, small and large bowel but only 1% involve the oesophagus [1]. Most of the lymphomas involving the GIT are Non-Hodgkin lymphoma (NHL) and it's very rare for Hodgkin lymphoma to involve the GIT [3,4]. Oesophageal lymphomas are usually secondary to adjacent lymph node invasion or contiguous spread from gastric lymphoma [1]. Primary oesophageal lymphoma is defined by oesophageal involvement without evidence of lymphoma elsewhere [5]. The endoscopic appearance of oesophageal lymphoma is indistinguishable from oesophageal carcinomas [5]. Hodgkin lymphoma usually arises from the proximal or mid-oesophagus while non-Hodgkin's lymphoma usually arises at the distal oesophagus [5-9]. The differential diagnosis of oesophageal symptoms in a patient being treated for lymphoma includes infection (fungal and viral), reflux oesophagitis, therapy-related mucositis and recurrence [10]. Oesophageal involvement by lymphoma could manifest in different ways including polypoid mass, ulceration, stenosis, spontaneous perforation and malignant trachea-oesophageal fistula.
A 75-year-old male presented with dysphagia for the last 4 months for which he was managing himself with fluid only diet. Over the last two months he developed severe odynophagia, for which he took Tramadol/Endone before swallowing. He also reported loss of weight.
He had a gastroscopy done in 1992 for peptic ulcers and reflux for which he was on proton pump inhibitors for years. The study reported a normal oesophagus.
Patient has background of Hodgkin lymphoma stage 2A diagnosed three years ago and was managed by chemotherapy. His treatment was complicated by febrile neutropenia and peripheral neuropathy, the former necessitating the need to change his chemotherapy regime from ABVD to ChIVPP. He was in complete remission for the past 15 months.
Barium swallow was undertaken for the present complaint which showed a 12 mm lesion situated 5 cm above the cardio-oesophageal junction. The PET scan revealed FDG avid oesophageal malignancy with probable FDG avid nodal disease around the proximal lesser curvature of the stomach, large subcarinal node, superior mediastinal and bilateral supraclavicular lymph nodes. Urgent gastroscopy was done, and the lesion was biopsied.
The specimen received in the Anatomical Pathology laboratory consisted of multiple fragments of tissue measuring 10 × 10 × 5 mm. Histologically, all the biopsy fragments consisted of necrotic debris with a small amount of benign squamous mucosa. Within the necrotic debris there were scattered single large atypical cells with enlarged hyperchromatic nuclei, increased nucleo-cytoplasmic ratio, prominent nucleoli and moderately abundant eosinophilic cytoplasm (Figure 1). Some of these cells were multinucleated. No classic Hodgkin or Reed-Sternberg cells were seen.
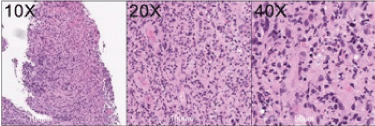 Figure 1: Haematoxylin and eosin staining demonstrating large atypical cells in a polymorphous background. View Figure 1
Figure 1: Haematoxylin and eosin staining demonstrating large atypical cells in a polymorphous background. View Figure 1
Because of the patient's previous history of classical Hodgkin lymphoma (nodular sclerosing), immunoperoxidase stains for Hodgkin lymphoma were performed. The large atypical cells stained for CD30, PAX5, CD15 and MUM-1. The tumour cells were negative for epithelial marker AE 1/3, CK7, CK20. No staining for both squamous and adenocarcinoma markers were present including P63, P40, CD15 and BER-EP4 (Figure 2, Figure 3 and Figure 4).
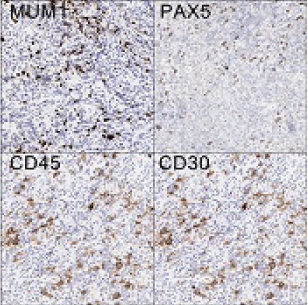 Figure 2: Immunohistochemistry demonstrating large atypical cells staining for CD30, CD45, PAX5 and MUM-1. View Figure 2
Figure 2: Immunohistochemistry demonstrating large atypical cells staining for CD30, CD45, PAX5 and MUM-1. View Figure 2
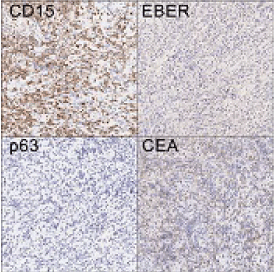 Figure 3: Immunohistochemistry demonstrating positive staining with CD15 and negative for p63, CEA and EBER. View Figure 3
Figure 3: Immunohistochemistry demonstrating positive staining with CD15 and negative for p63, CEA and EBER. View Figure 3
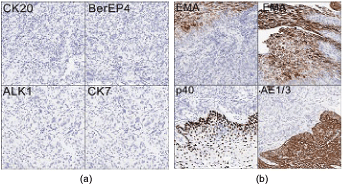 Figure 4: Immunohistochemistry demonstrating negative staining for epithelial marker AE 1/3, CK7, CK20, P63, p40, EMA, Alk1 and BerEp4. View Figure 4
Figure 4: Immunohistochemistry demonstrating negative staining for epithelial marker AE 1/3, CK7, CK20, P63, p40, EMA, Alk1 and BerEp4. View Figure 4
To the best of our knowledge, Hodgkin lymphoma of the oesophagus is extremely rare, and it could be primary or secondary. Primary Hodgkin lymphoma of the oesophagus is vanishingly rare. However, recurrent Hodgkin lymphoma in the oesophagus has been reported and it should be considered in the differential diagnoses of prolonged oesophageal symptoms in lymphoma patients [2,11].
In conclusion, we reported a case of Hodgkin lymphoma of the oesophagus in a patient with background of nodular sclerosing Hodgkin lymphoma in remission after completion of chemotherapy.
Data used in this research were obtained with the assistance of the Hunter Cancer Biobank, Hunter Cancer Research Alliance, University of Newcastle, NSW, Australia.