Hyperglycemia and type 2 diabetes mellitus have been shown to have a negative impact on cognitive performance in older adults. In order to assess the relevance of this effect in memory clinic patients, we examined the relationship between hyperglycemia and cognition in an unselected out-patient sample.
In a cross-sectional study in memory clinic patients, glycosylated hemoglobin (HbA1c) was determined and related to cognitive performance, subjective memory impairment, and self-perceived cognitive blackouts.
The frequency of mild cognitive impairment or mild dementia of the Alzheimer type was increased 1.7-fold in subjects with hyperglycemia. The HbA1c value was negatively correlated with global cognitive performance as well as with orientation, short-term memory and visuospatial capabilities. The frequency of cognitive blackouts was positively correlated with HbA1c levels and it was increased in hyperglycemic subjects.
Hyperglycemia could be confirmed as a major risk factor for cognitive impairment. This could be shown even in a non-representative cross-sectional memory clinic sample. Thus, the HbA1c determination may be recommended for a routine diagnostic memory clinic work-up.
Diabetes mellitus, Hyperglycemia, HbA1c, Mild cognitive impairment, Dementia, Alzheimer, Subjective memory impairment, Cognitive blackouts, Memory clinic
Patients with type 2 diabetes mellitus have an increased risk for cognitive decline [1], for mild cognitive impairment [2], and for vascular as well as for Alzheimer's dementia [3-6]. A relationship between blood glucose levels and risk of dementia has been established [7]. Even prediabetes and insulin resistance are associated with cognitive decline and brain atrophy [8].
Diabetes may compromise cognitive function by various mechanisms, such as brain vascular lesions, insulin resistance, oxidative stress, the accumulation of advanced glycilation end products, inflammation or a competition of insulin and β-amyloid on the insulin-degrading enzyme [9].
This may also apply to subjects with prediabetes as reflected by a moderately increased HbA1c level. Accordingly, it has been shown that the HbA1c value correlates with the rate of brain atrophy [10] and that it predicts conversion to dementia and mild cognitive impairment [11]. Glycosilated hemoglobin (HbA1c) is a measure of blood glucose concentration over the past two to three months and is used both for the diagnosis of diabetes and the assessment of diabetes management [12].
We studied the effects of diabetes and hyperglycemia as determined by elevated HbA1c values on cognitive performance, subjective memory impairment and self-perceived cognitive blackouts in a group of patients of our memory clinic.
The analyses were carried out in 113 patients consecutively presenting for the first time for memory assessment at our memory clinic. They were 70 women (61.9%) and 43 men (38.1%) at ages between 50 and 87 years (mean ± SD: 61.2 ± 9.1 years). Subjects with a history of neurological disorder, with an actual neurological or psychiatric disorder, or with a medication that may compromise cognitive performance were not included. Medical history was taken with special reference to a diagnosis of type 2 diabetes mellitus and the respective medication. HbA1c levels were determined by means of an enzymatic immunoassay.
Assessment of cognitive performance and assignment to the diagnostic groups of "No Cognitive Impairment" (NCI), "Mild Cognitive Impairment of the amnestic type" (MCIa) and "Mild Dementia of the Alzheimer Type" (mDAT) was performed by means of the Structured Interview for the diagnosis of dementia of the Alzheimer type, multi-infarct dementia and dementias of other aetiology according to ICD-10 and DSM-III-R (SIDAM) [13]. The neuropsychological test battery of the SIDAM includes the Mini-Mental State Examination (MMSE) [14]. If appropriate, further clinical, laboratory or imaging investigations were initiated.
The presence of subjective memory impairment was examined by asking the patients: (1) Do you feel like your memory is becoming worse? (2) If so, does its worry you? (3) Do you think that your memory is poorer than that of other persons of your age?
The frequency of self-perceived cognitive blackouts was assessed by means of the Checklist for Cognitive Blackouts (CCB) [15]. The CCB is made up of five items describing frequent self-observations of persons with beginning Alzheimer's dementia (Table 1).
Table 1: Items of the Checklist for Cognitive Blackouts (CCB). View Table 1
For items 2 to 5, the frequency of occurrence of the respective observations is graded as "0" (for "never"), "1" (for "rarely" or "less than once a week"), "2" (for "frequently" or "once or several times a week"), and "3" (for "permanently" or "once or several times a day"). Item 1, the frequency of which had been observed to be inversely related to cognitive impairment [15], is used to compensate for underreporting and is graded in the reverse sense. For each of the items 0 to 3 points are issued, leading to CCB scores between 0 and 15.
A diagnosis of diabetes had been established in 13 subjects (11.5%), of whom 5 were exclusively under dietary treatment, 8 were treated with metformin, one additionally with insulin. The HbA1c values ranged between 4.6 and 8.6 (mean ± SD: 5.4 ± 0.6), with values indicating hyperglycemia (≥ 5.8) in 26 subjects (23.0%).
The questions for subjective memory impairment were affirmatively answered for question 1 (memory decrease) by 75 subjects (66.4%), for question 2 (concerns) by 58 subjects (51.3%), and for question 3 (comparatively poorer memory) by 41 subjects (36.3%). With respect to self-perceived cognitive blackouts, the CCB scores ranged between 2 and 12 (mean ± SD: 5.8 ± 2.3), with critical CCB values (≥ 7) in 37 subjects (32.7%).
The SIDAM revealed No Cognitive Impairment (NCI) in 60 subjects (53.1%), a Mild Cognitive Impairment of the amnestic type (MCIa) in 38 (33.6%), and a mild Dementia of the Alzheimer Type (mDAT) in 15 (13.3%). The MMSE score ranged between 16 and 30 (mean ± SD: 27.6 ± 2.7).
When examining the relationships between diabetes or hyperglycemia and cognitive impairment (MCIa or mDAT) by means of chi-square tests, we found no effect of a diagnosis of diabetes (chi2 = 0.284). However, 69.2% of the hyperglycemic subjects (HbA1c ≥ 5.8) were found to be cognitively impaired, compared to 40.2% of the normoglycemic subjects (chi2 = 6.760; P = 0.009). There were no effects of hyperglycemia on subjective memory impairment, but with respect to self-perceived cognitive blackouts, the CCB score was significantly increased in the hyperglycemic subjects (6.8 ± 3.0 vs. 5.5 ± 1.9; T = 2.617; P = 0.010).
The HbA1c value was found to be inversely correlated with various measures of cognitive performance. After correction for age, there were significant negative correlations with the MMSE score (R = -0.287; P = 0.008), with the SIDAM score (SISCO) (R = -0.358; P < 0.001), and with the SIDAM syndromes "orientation" (R = -0.344; P < 0.001), "short-term memory" (R = -0.294; P = 0.002) and "visuospatial capabilities" (R = -0.259; P = 0.006). We also found a positive partial correlation of the HbA1c value with the frequency of self-reported cognitive blackouts as reflected by the CCB score (R = 0.258; P = 0.006). In Figure 1, cognitive performance (MMSE scores) and self-perceived cognitive blackouts (CCB scores) are plotted over the HbA1c values.
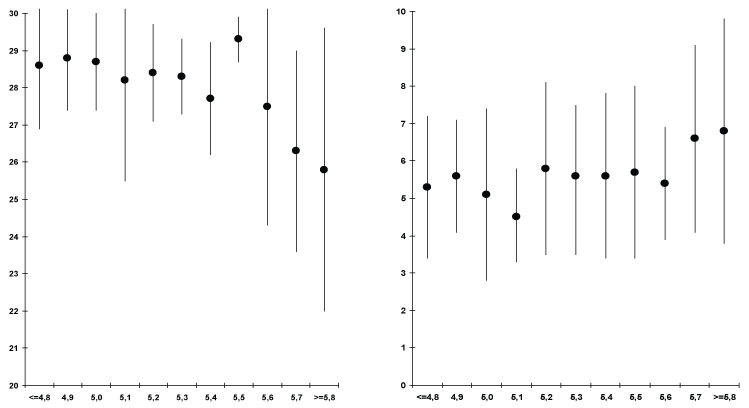 Figure 1: Relationship of cognitive performance and self-perceived cognitive blackouts to the HbA1c value.
Figure 1: Relationship of cognitive performance and self-perceived cognitive blackouts to the HbA1c value.
Cognitive performance (MMSE scores, left) and self-perceived cognitive blackouts (CCB scores, right) are plotted as means + SD over the HbA1c values [%]. View Figure 1
With increasing HbA1c values, the MMSE score decreases and the CCB score increases, particularly at HbA1c values above 5.6.
We found a 1.7-fold increased frequency of cognitive impairment in subjects with hyperglycemia. The HbA1c value was negatively correlated with the global scores for cognitive performance (MMSE and SISCO) and with the SIDAM syndromes for cognitive domains typically affected in incipient dementia, namely orientation, short-term memory and visuospatial capabilities. Furthermore, in subjects with hyperglycemia the frequency of self-perceived cognitive blackouts was increased and it was positively correlated with the HbA1c value. On a quantitative level, mainly HbA1c values above 5.6% contributed to these correlations.
These observations confirm the importance of hyperglycemia as a risk factor for late-life cognitive impairment [9]. They go well along with the longitudinal data of the large U.S. Health and Retirement Study [16]. The strength of the effect of hyperglycemia may be deduced from the fact that the significant relationships of HbA1c to cognitive performance could be even found in a cross-sectional examination of a moderately large non-representative sample of memory clinic patients. The quantitative increase of the risk for cognitive impairment is in good agreement with the relative risk of type 2 diabetic individuals for the development of dementia in longitudinal studies [17,18].
This also applies for the effect of hyperglycemia on self-perceived cognitive blackouts as assessed by means of the CCB score, which has been found to be closer related to cognitive impairment than subjective memory impairment, at least the way the latter is usually assessed [15].
It may be surprising that we found no relationship between a diagnosis of diabetes and cognitive impairment. However, apart from the small number of diagnosed diabetic patients in our sample, this may be attributed to factors, which come into effect once a diagnosis of diabetes has been established. Therapeutic interventions, such as dietary and drug treatments, may have various effects on HbA1c level and cognitive performance and may influence the relationships between them [19,20].
Hyperglycemia as reflected by the HbA1c values seems to have a considerable effect on cognitive performance in older adults. In only about half of the subjects with hyperglycemia a diagnosis of diabetes had been established. Given the frequency of hyperglycemia, it may be worth considering the inclusion of the HbA1c value in the routine diagnostic memory clinic work-up.
The support and assistance of Yvonne Lembach is gratefully acknowledged.
None of the authors has any conflicts of interest to declare.