The development of point of care diagnostic protease sensors applied to wound healing has received increased interest in chronic wound treatment as an interface for chronic wound dressings. Biosensor technology and the use of nanomaterials have grown exponentially in recent years. A biosensor is fundamentally a biomolecule (functioning as a transducer) attached to a transducer surface, which is activated by a biochemical property that prompts a detection signal specific to a target biomarker. For instance, nanomaterials are often derivatized with a biomolecule that provides selectivity and sensitivity for diagnostic biomarkers. Nanocellulose can be prepared as a transducer surface from an assortment of sources like wood and cotton in a variety of physical forms such as nanocrystals, nanocellulose composites, and nanocellulose aerogels that possess ideal properties including biodegradability, biocompatibility, functionality, and a high specific surface area. Interfacing nanocellulosic biosensors with a wound dressing having protease-lowering properties allows in situ sensor detection selectivity and sensitivity to monitor the effectiveness of the dressing and titer of protease removed from the wound. Here we discuss chronic wound dressing design and mechanism with an emphasis on protease-lowering dressings and chronic wound modalities and a discussion of a number of different types of nanocellulosic materials as interface materials for potential sensor-dressing application. As a specific model, we focus on nanocellulosic systems conjugated to the elastase substrate n-succinyl-Alanine-Alanine-Proline-Valine-7-amino-4-methylcoumarin and n-succinyl-Alanine-Proline-Alanine-7-amido-4-methyl-coumarin, and discuss comparative properties and molecular design, crystal structure, protease binding kinetics, specific surface area, permeability, surface charge, and sensitivity to proteases as relates to biosensor positioning in a dressing design.
Advances in biosensor technology hold promise to revolutionize healthcare and diagnosis through assessment of health status, disease onset, and progression using non-invasive methods [1-4]. This is especially the case in the area of sensor and imaging for wound healing [5], which is critical for the management of wounds stalled in the inflammatory state i.e., a chronic wound [6].
Numerous inflammatory-based disease states are noted for secreting neutrophil and bacterial enzymes including, myeloperoxidase and gelatinases, and neutrophilic proteases. Two proteases that have been associated with chronic wound pathology are Matrix Metalloproteases (MMPs) [7] and Human Neutrophil Elastase (HNE) which have prolonged deleterious effects, from excessive degradation of extracellular matrix proteins [8-10], fibronectin [11,12], and growth factors [13]. It is also noteworthy that HNE and MMPs have been shown to work in a collaborative way to accelerate degradation [14].
Chronic wounds arise from an arrestment in the inflammatory stage of the natural flow of the wound healing phases of hemostasis, inflammation, proliferation, fibroplasia, and remodeling and are often found to have prolonged high titers of neutrophils which lead to high protease concentrations [15]. Although neutrophils play important roles in host defense and debridement of damaged tissue they contain free radical generating enzymes and proteases implicated in chronic inflammatory diseases. Thus, the stalled inflammatory state of chronic wounds [16,17] is often associated with the wound fluid containing high levels of harmful proteases (HNE and MMP) that have become increasingly important biomarkers for point-of-care diagnosis which is also important in appropriate wound dressing selection [18-20]. Thus, an increased interest in protease biosensors which may be designed to detect proteases of clinical interest has arisen giving rise to what has been referred to as a ‘test and treat” strategy [21].
A biosensor's basic design may be defined as the interface of a molecular or receptor-recognition property with a cellular or biochemical activity, triggering a "bio-molecular switch" that in turn is connected with a detector signal [3,22]. Nanomaterials with high surface area and biocompatible properties make ideal transducer surfaces for biosensors. Nanocellulose, which we address in this paper, provides noteworthy advantages as a transducer surface, but has received less attention than other nanomaterials for sensor applications [18,23].
Chronic wound dressings used in the treatment of non-healing wounds may be organized into four groups including: passive, interactive, bioactive, and intelligent [24,25]. Passive dressings protect the wound bed from trauma and bacterial infection [24,26-28]. Interactive dressings maintain a moist wound and facilitate fibroblast proliferation and epithelialization [29,30] by way of a added functional property conferred by a material component of the dressing i.e. biomaterials like honey, collagen, hyaluronic acid, and chitosan have been associated with enabling mechanisms of wound healing. Current advancements in commercially available semiocclusive dressings include improvement on hydrocolloids, hydrogels, and semi permeable foam dressings that are designed to modulate fluid balance by retaining moisture or reducing exudate volume, and acting as a bacterial barrier while improving the range of their use i.e. to reduce pressure, friction, and shear for the patient for example [31].
Bioactive wound dressings, which include delivery systems, skin substitutes, and biological dressings, are being employed for their clinical efficacy to promote wound healing in challenging wounds by activating cellular roles in the proliferative or inflammatory stages of wound healing [24,32,33]. Bioactive dressings have been employed as a combination of keratinocytes, fibroblasts, and other cellular constituents as well as biopolymer composites designed to facilitate epidermal growth as are required in burn and full thickness wound patients [34,35]. It is noteworthy that a recent review by Dreifke, et al. has discussed FDA approved wound healing dressings based on collagen, silicon, chitosan, and hyaluronic acid in light of wound environment sensors for early detection of complications [36]. It is also important to note in the context of bioactive analogs the development of therapeutic growth factor and neuropeptide analogs, Advances in the understanding of wound healing pathophysiology have provided targets for bioactive topical therapies with an emphasis on peptide and growth factor therapies which have appeared promising for modulation of the proliferative phase of wound healing [37-39].
An intelligent dressing may perform a specific function, for example protease sequestration or low-level hydrogen peroxide generation. It may also have sensor capabilities. Sensor functions like color, pH, and temperature change, have been explored in dressing design where there is an underlying activation due to biochemical changes and cellular responses [31,40,41] in the wound. For example, biofilm detection and treatment is a clinically challenging area in 60 percent of chronic wounds; and Thet, et al. recently reported a prototype hydrogel wound dressing that is described as emitting a glow in response to bacterial biofilms present in a wound [42].
Wound fluids from chronic and acute wounds have different characteristic microenvironments and the composition of the fluid and the wound bed microenvironment undergoes basic changes in moisture, temperature, pH and oxygen. Thus, theranostic markers like pH useful in predicting whether the wound will heal may reflect the general condition of a wound in lieu of giving a more specific indication as a biomarker does [43-46]. In this regard it is notable that Rahimi, et al. evaluated a flexible pH (4-10) sensor fabricated on a polymer coated palette and paper substrate, consisting of cyto-compatible materials, which exhibited a sensitivity of -50 mV/pH unit in wound fluid i.e. a recent approach to point of care theranostic assessment [47]. The pH assessment is a beneficial indicator of wound healing considering acute and normal wounds have a slightly acidic pH (5.5-6.5) whereas chronic wounds tend to exhibit a higher pH ( > 7.4) due to alkaline byproducts from proliferating bacterial colonies [47]. The use of flexible polymer and paper substrates including silicone elastomers, parylene, polyimide, and cellulose paper as pH, oxygen, moisture, and temperature sensors also enable detection of changes in the wound microenvironment using materials that have conformability to the wound bed [45].
Some commercially available dressings now have the added functionality of indicating wound exudate saturation to signal dressing change [48]. However, current state of the art sensor nanoscience and wound dressing technology are poised to yield improved multifaceted sensor/dressings that will provide point of care diagnostics and theranostics by increasing sensitivity to detectable biomarkers while providing an improved opportunity to treat chronic wounds based on cellular and molecular specific pathologies.
Clearly, advanced dressings integrate more progressive characteristics and may overlap in their categorization. However, the combination of a bioactive, semiocclusive, or intelligent dressing with a point of care diagnostic or theranostic sensor is both an emerging paradigm and a reasonable extrapolation of the current technologies available for chronic wound care.
The amelioration of harmful levels of proteases in chronic wounds has been a topic of increasing interest and focus in the design and preparation of non-healing wound dressings over the last two decades. A protease sequestrant also termed protease-lowering or protease attenuation) dressing's design may be couched in a number of molecular motifs based on the structural features of the protease or target enzyme, which interferes with the healing process. The molecular features of the dressing's material may be targeted to the protease size, charge, active site, or conformation to enhance selective binding of the protein to the dressing material and remove proteases from the wound bed. Thus, the mechanism of action may also be through other cellular or biochemical events that modulate protease levels. Previously reported active wound dressings designed to redress the biochemical imbalance of the chronic wound in this manner are composed of collagen and oxidized regenerated cellulose [49], nanocrystalline silver-coated high density polyethylene [50], which also serves as an antimicrobial, deferioxamine-linked cellulose [51], electrophilic and ionically derivatized cotton [52], peptide [53] or carbohydrate-conjugates [54], and sulfonated ion exchange derivatives of hydrogel polymers [55]. Electrostatic uptake mechanisms have also been demonstrated with in situ fluorescence hydrogels that both detect and trap proteases [56]. In addition, phosphorylated cotton was shown to sequester both human neutrophil elastase and collagenase [57], and developed as an FDA approved dressing [58]. From this work it was shown that the best formulation for development was a treatment of sodium hexameta phosphate in urea that is attached through low cost conventional textile finishing approaches. More recently the role of polyphosphates when incorporated into a foam matrix have been shown to sequester metalloproteases (MMPs) derived from Pseudmonas aeruginosa [59]. Interestingly, when combined with silver in a hydrogel base polyphosphate was also found to give rise to anti-biofilm activity [60]. Also noteworthy is work reported by several authors that addresses the activity and mechanism of action of protease-lowering activity of phosphorylated analogs in terms of dressing functionalized active site inhibitors of elastase and matrix metalloprotease [61-63].
A veritable arsenal of different protease inhibitors has been developed for inflammatory diseases, and a variety of non-toxic protease inhibitors have been proposed and studied for controlled release into chronic wounds. Among these oleic acid, which is the principle component of olive oil has been demonstrated to be an elastase inhibitor [64], and more recently proposed as an inhibitor of matrix metalloprotease and plasmin [65] as well. Oleic acid has been demonstrated as an albumin-mediated elastase inhibitor when released from different dressing [62,66]. Interestingly olive oil has been studied for a healing-oriented mechanism of action [67] in chronic wounds as well as the subject of a recent clinical trial for chronic wound treatment [68]. Oleic acid has been shown to accelerate healing of acute wounds [69]. It is noteworthy that the judicious use of protease inhibitors should be considered for formulations where growth factor and peptide therapeutics are being applied to wounds to prevent the inactivation of proteinacesous drugs by inhibiting protein degradation in the high protease-containing wound environment. Although a number of groups have studied this approach and developed patents claiming formulations with growth factors and protease inhibitors, no products have yet been marketed using this approach [70].
The progress toward clinically based MMP sensors for point of care diagnostics has been reviewed in recent years [7]. Biosensors of MMP have been reported based on fluorescence resonance energy transfer (FRET) and include fluorescent tripeptide substrates of proteases with the mechanism being linkage of a highly sensitive fluorescent response between donor and acceptor molecules [71]; quantum dots, gold nanoparticles or organic dyes have also been explored with a similar approach [72,73]. Graphene which has a very large specific surface area has also been employed as a transducer surface for an oxide-based sensor, a fluoresce in labeled peptide based on electrostatic interactions and as a fluoresce in isothiocyanate-labled peptide conjugate [74,75].
A recent review of HNE biosensors has listed a variety of designs proposed and utilized with the corresponding protease sensitivity ranges [18]. Briefly, biosensor-based detectors of elastase have included a microchip (Henares, et al. 2006) [76], microdialysis probe [77], fluorometric supramolecular pore sensor [78], immobilization of peptides on quantum dots [79], immobilization of HNE on a biosensor chip for surface plasma resonance [80], ethoxyacrylate resin [81], using a colorimetric detection and fluorometric peptides coupled to paper [79] as well as cotton cellulose nanocrystals [18,82]. Schyrr, et al. recently reported fluorescent sensor film made from cellulose nanocrystals and polyvinyl alcohol has also been demonstrated as a protease sensor [83] with added functional porosity.
A variety of imaging spectroscopy technologies are beginning to replace traditional visual cutaneous assessment of burn, diabetic and traumatic wounds [84], and recently an approach utilizing wound blotting of necrotic tissue for distribution analysis of marker proteins with nitrocellulose has been proposed as an approach to the prediction of healing progress in pressure ulcers [85]. Point-of-care protease detection has received increased attention and has been applied clinically throughout the world to enable chronic wound treatment decisions [19,86,87]. The pros and cons of a ‘test and treat' strategy approach for elevated protease activity in venous ulcers was recently reviewed [21]. Moreover, peptide-based protease sensors are widely used in monitoring enzyme activity. They have a rich structure/function literature that can be utilized to design enzyme sensors and are central to a number of protease-based diseases and their therapy [88]. The clinical application of peptide-based biosensor approaches to protease biomarker disease assessment in chronic wounds is growing [19]. On the other hand, although there have been a few reports of prototypes, the combination of detection and treatment in a chronic wound dressing has not been adopted on a widespread clinical basis [18]. Thus, the ability to combine measurable protease detection with a dressing motif that removes proteases is an emerging research and development goal of wound healing biomaterial design [18,21,89].
There are many considerations in clinically evaluating a combined dressing-sensor system for protease detection. The use of an in situ detector should withstand scrutiny as to whether it be applied as an indicator to change the dressing and accurately detect the wound protease titer. It is also important to note that not all chronic wounds have abnormally high protease levels, and normal protease levels are important to wound remodeling [17]. In light of variations in wound protease levels a ‘dipstick' approach for assessment of protease levels in chronic wounds [19,86] has been suggested before treatment options and dressing selection is made.
Although the sophistication and sensitivity of detectors underlie much of the progress in the rapidly growing field of biosensors, the composition of the transducer surface material and its environment can also complicate and dampen the sensitivity of detection. Thus, biosensor materials should ideally be compatible with the complexity of the bio-system by being resistant to nonselective adsorption by lipids, proteins, polysaccharides, cellular debris, and breakdown of the transducer surface. In this regard, nanocellulose is both hydrophilic and structurally resilient, rendering it advantageous as a biomaterial.
The use of cellulose nanocrystals and nanofibrillated cellulose in biomaterials has been shown to have potential advantages in optics [90], nanocomposite materials [91], and has continued to show promise as an effective material in semiocclusive dressing design for chronic wound applications [92-94]. Nanocellulose is a crystalline biopolymer with hydrophilic properties and high surface area [91]; it possesses reactive hydroxyls that can be derivatized to covalently append a wide range of biologically active molecules. Its hydrophilic surface of nanocellulose improves wettability and abrogates binding by proteins and lipids.
The high specific surface area of the nanocellulose systems enables enhanced sensitivity of a peptide-based protease sensor by accommodating a higher peptide titer on the crystalline surface, and subsequently performing as a sensor more efficiently by way of increased binding of the protease on the sensor surface. For example, print cloth and filter paper, which are largely cellulosic when compared with and wood cellulose nanocrystals have a specific surface area of 0.016-0.020 m2g-1 and 261.2 m2g-1, respectively [95]. Upon attachment of peptides to these types of materials, it was found that the approximate number of peptides per 200 anhydroglucose cellulosic residues are print cloth (1), filter paper (2), and cellulose nanocrystals (12), and these ratios of peptide to cellulosic units correspond with a relative order of increasing specific surface area [95]. The relative activities also paralleled specific surface area of the cellulosic versus nanocellulosic materials and demonstrate the contrast in efficiency of cellulosic versus nanocellulosic sensors.
As introduced above elastase sensors provide a model for point of care biomarkers in inflammatory diseases and chronic wounds. It is understood that broad spectrum protease assessment would be beneficial in clinical point of care diagnosis. However, here we discuss elastase sensors as a model for broad spectrum protease sensors as a model for this approach. Here we evaluate fluorescent protease biosensors on nanocrystals, nanocellulose composites, and nanocellulose aerogels immobilized with the tetrapeptide substrate n-succinyl-Alanine-Alanine-Proline-Valine-7-amido-4-methyl-coumarin (Suc-Ala-Ala-Pro-Val-AMC) and the tripeptide substrate n-succinyl-Alanine-Proline-Alanine-7-amido-4-methyl-coumarin (Suc-Ala-Pro-Ala-AMC).
Standard fluorenylmethyloxycarbonyl (Fmoc) chemistry was used for esterification and immobilization of the peptide onto the transducer materials [96,97]. The esterification and peptide immobilization of the transducers is previously outlined by Edwards, et al. [82,95]. Briefly, the nanocellulosic transducers cotton cellulose nanocrystals (cCNC), wood nanocellulose composites 66/33 and 50/50 (wNCC), and nanocellulosic aerogel (NA) were esterified with Fmoc-Gly-OH with the respective coupling agents (Ethylcyanoglyoxylate-2-oxime (Oxyma Pure), Diisopropylcarbodiimide (DIC), and 4-dimethylaminopyridine (DMAP) in N,N-dimethylformamide (DMF)) and sonicated. The materials were washed thrice by centrifuging or filtration DMF, dichloromethane (DCM), and in select cases allowed to air dry. (Note: the NA-Gly-Fmoc was allowed to remain in DMF for storage and was not air dried). The transducers cCNC-Gly-Fmoc, wNCC-Gly-Fmoc, and NA-Gly-Fmoc were deprotected or stored at ~4-8°C.
A deblock solution (piperidine/DMF) was used to deprotect the Fmoc protecting group from the glycine esterified transducers. (Note: the NA-Gly-Fmoc was washed with DMF prior to soaking in the deblock solution). The materials were washed thrice by centrifuging or filtration with DMF, DCM, and in select cases allowed to air dry. (Note: the NA-Gly was washed thrice with DMF followed by preparation for peptide coupling or stored in DMF for further use).
Peptide immobilization of cCNC-Gly, wNCC-Gly, and NA-Gly were achieved by activating the tripeptide (Suc-Ala-Pro-Ala-AMC) or tetrapeptide (Suc-Ala-Ala-Pro-Val-AMC) substrates with DMAP in DMF for twenty minutes, to which Oxyma Pure and DIC were added. The solutions were sonicated for 3 hours, placed in the refrigerator overnight, and purified by centrifuging or filtering thrice with DMF, DCM or methanol (MeOH), and in select cases allowed to air dry. (Note: the NA-Pep was washed thrice with DMF, thrice with PBS, and stored in PBS). The biosensors were stored at ~4-8°C until further use. Note "biosensor" will be defined as the glycine esterified transducer immobilized with the tripeptide or tetrapeptide substrate.
Upon the completion of drying, the peptide was cleaved from each biosensor by adding a mixture of TFA/water/triisopropylsilane to the biosensor for three hours. The solution for each biosensor was diluted (water/acetonitrile) and submitted for Electrospray ionization liquid chromatography mass spectrometry (ESI-LC/MS), which confirmed the intact sequence of the peptide component of the biosensor via its molecular weight.
The specific surface area (SSA), identifies the total surface area of a solid material per the unit mass of the material. The calculation of the cellulosic nanocrystals and the BET specific surface area of the wNCCs were previously outlined in by Edwards, et al. [95]. The Brunauer-Emmett-Teller (BET) specific surface area of the NA was determined using a Micrometrics ASAP 2020 instrument [98] with a nitrogen sorption source at 77 K detector. The NA (0.033 g) was degassed at 100°C for 4 hours prior to the analysis followed by N2 adsorption at 77.4 K.
The sensitivity assay evaluates the limit of detection of which the biosensors are able to detect human neutrophil elastase (HNE). The sensitivity assay was previously outlined by Fontenot, et al. [99]. Briefly, duplicates of ~2 mg of each biosensor were placed into a 96 well plate and 100 μL of a phosphate buffer solution (PBS) was added. To start the reaction, 50 μL human neutrophil elastase (HNE) ranging from 2-0.0156 U/mL was added to the standard curve and to the biosensors to provide a total volume of 150 μL. Fluorescent measurements at 37°C were monitored for 1 hour at 1-minute intervals using a Biotech Synergy HT with tungsten halogen lamp and photomultiplier detection. The 96 well plates were shaken before each measurement for 3 seconds and the measurements were acquired at 360 nm excitation and 460 nm emission. The wound like fluid herein comprises of PBS and HNE.
The zeta potential (ζ) determines the surface charge properties of the peptide free transducers (cCNC, wNCC, NA), which are immersed in a conducting solvent and measured using a Malvern Zetasizer nano ZS90 equipped with a laser doppler and detected with a phase analysis light scattering. The wNCC and NA were ground into a powder using a Wig L Bug Ball Mill and pulsated for ~1.5 minutes. Stock solutions (5 mg/0.25 mL) of the ground transducers were prepared in DI H2O or PBS from which 1 mg/mL (cCNC and NA) and 0.5 mg/mL (wNCC) solutions were prepared. The respective solutions were sonicated for 2 hours prior to obtaining triplicate zeta potential measurements.
The MVTR measures the rate at which moisture can permeate solid porous materials such as the wNCCs. Commercial Testing Company performed the water vapor transmission rate according to the ASTM E96-95 method with procedure B - water method at 23°C for the wNCC.
Cotton cellulose nanocrystals (cCNC) and wood cellulose nanocrystals (wCNC) have a high surface area, mechanical robustness, a geometrical rod-like or whisker shape nanocrystal, and a negatively charged surface [18,100]. The cellulose nanocrystals have a high SSA of 186.2 m2/g-1 that allows for a greater number of peptide substrates to be anchored onto the surface. The fluorescence sensitivity of the nanocrystalline peptide biosensors was as low as 0.05 U/mL, which correlates to a higher concentration of peptide substrate loading and to a higher concentration of AMC-released fluorophore. Furthermore, as shown in table 1 the cellulose nanocrystals posses a negatively charged surface of -41 mV [82,101] that enable binding of elastase. The surface charge influences the interaction between the wound fluid and the wound dressing, which promotes uptake of positively charged proteases by negatively charged materials consistent with phosphorylation, sulfonation, and oxidation of cellulose. This electrostatic mechanism promotes increased binding to HNE by targeting positively charged amino acids in the proteins sequence [52,102]. Therefore, negatively charged cellulose nanocrystals bind the HNE proteases present both in the wound fluid and the dressing, which has been shown to accelerate the healing rate of the wound [52,102,103].
Table 1: List of each nanocellulosic biosensor with its respective peptide substrate, SSA, sensitivity, and surface charge values. View Table 1
Figure 1 shows a crystallite-tethered fluorescent tripeptide elastase substrate linked via the cellulose primary hydroxyls to a cotton cellulose nanocrystal surface. This depiction is representative of the molecular surface of a nanocellulose transducer with sensor molecules and constitutes the design components of a point-of-care protease sensor. Recently analogous structures have been discussed [99]. Modeling of the sensor molecule on the crystalline surface when accompanied with enzyme kinetic analysis is helpful in predicting the interaction of the protease with the sensor in terms of enzyme/substrate binding efficiency and crystal surface versus enzyme spatial considerations [104] i.e., of the enzyme on the transducer surface as it relates to peptide protease binding and kinetics. A peptide-nanocellulose analog characterized with a similar crystallite surface was shown to undergo a five-fold more efficient interaction with HNE than was observed of the analogous enzyme substrate in solution i.e. a higher Kcat/Km of the peptide-nanocrystalline material versus peptide in solution [104]. This improvement in the kcat/km is attributable in part to enhanced binding of the cationic serine protease to an anionic crystallite surface.
 Figure 1: Model of the cotton cellulose nanocrystals biosensor with the tripeptide based on the cellulose I diffraction patterns with a width of 58.8Ǻ (109 chains and 15 layers) [82]. The tripeptide is substituted on the crystallite structure with a D.S. level of 0.044 and a SSA of 186.2 m2g-1. View Figure 1
Figure 1: Model of the cotton cellulose nanocrystals biosensor with the tripeptide based on the cellulose I diffraction patterns with a width of 58.8Ǻ (109 chains and 15 layers) [82]. The tripeptide is substituted on the crystallite structure with a D.S. level of 0.044 and a SSA of 186.2 m2g-1. View Figure 1
Over the last ten years a variety of colorimetric protease sensors directed to point of care models have been reported. Edwards, et al. first reported a colorimetric approach for elastase detection with ethoxyacrylate resin as a transducer surface and demonstrated the relevant enzyme-substrate binding properties [105]. The incorporation of colorimetric detectors i.e. remazol brilliant blue into peptidoglycan and polymer motifs, with a goal of making them amendable to the wound environment, was reported with a variety of designs as a diagnostic device paradigm for wound infection relevant proteases [89,106]. Previously we proposed a colorimetric protease detection with dressings using cotton cellulose nanocrystals derivatized with both a tetrapeptide protease substrate (n-succinyl-Alanine-Alanine-Proline-Valine-p p-nitroanilide, Suc-AAPV-pNA) or tripeptide protease substrate (succinyl-Ala-Ala-Val-p-nitroanilide, Suc-AAV-pNA), which employs the visual amplification of a chromophore with a dye to enhance sensitivity [18] (Figure 2).
 Figure 2: Shows the cleavage pathway of A) pNA with NEDH amplification; B) AMC from the tetrapeptide substrate with HNE [110]. View Figure 2
Figure 2: Shows the cleavage pathway of A) pNA with NEDH amplification; B) AMC from the tetrapeptide substrate with HNE [110]. View Figure 2
The colorimetric sensor-based dressing diagram shown in figure 3 comprises a matrix of layers functioning to: 1) absorb wound exudate and allow entry of the protease onto the surface of the sensor through a semipermeable absorptive layer and allow, 2) protease reaction with the protease sensor substrate where upon, 3) release of the chromophore onto a 10 kDa dialysis membrane permeated with amplifying dye, and 4) a barrier layer to prevent backwash of the released dye into the wound. The cellulose nanocrystal tripeptide conjugate with colorimetric amplifying agents was capable of detecting HNE at 0.05 U/mL [18]. The free particle nature of the cellulose nanocrystals allows efficient use of the nanoparticles properties as a sensor, which includes high SSA, versatility of application, and improved sensitivity to the protease biomarker. The benefit of interfacing a protease sensor with a dressing may be construed in its value as an indicator for dressing saturation of proteases, a detector to monitor the efficacy of the dressing and its interaction with the wound environment.
 Figure 3: Diagram of the cotton cellulose nanocrystals conjugated to a chromophore derivatized (tripeptide) that in turn is interfaced with a semi permeable wound dressing. The contacting layer absorbs wound exudate and allows passage of wound exudate-containing proteases; the barrier layer prevents the para-nitroanilide (pNA) chromophore from entering the wound bed. The cellulose nanocrystal peptide conjugate is activated upon contact with protease when the dressing is saturated with protease. The pNA migrates through a 10 kD dialysis cellulose membrane to the color amplifying reagent layer where a color appears signaling the presence of proteases in the dressing. The semi permeable top layer protects the dressing. View Figure 3
Figure 3: Diagram of the cotton cellulose nanocrystals conjugated to a chromophore derivatized (tripeptide) that in turn is interfaced with a semi permeable wound dressing. The contacting layer absorbs wound exudate and allows passage of wound exudate-containing proteases; the barrier layer prevents the para-nitroanilide (pNA) chromophore from entering the wound bed. The cellulose nanocrystal peptide conjugate is activated upon contact with protease when the dressing is saturated with protease. The pNA migrates through a 10 kD dialysis cellulose membrane to the color amplifying reagent layer where a color appears signaling the presence of proteases in the dressing. The semi permeable top layer protects the dressing. View Figure 3
Fluorescent imaging on the other hand has higher sensitivity limits toward visualization of molecular biomarkers at nano- or picomole levels of protease, but requires a fluorometric device for imaging. However it is notable that fluorescence imaging of wounds has shown potential to directly visualize the skin's natural fluorophores (collagen and elastin for example), and this type of approach has been demonstrated with nicotinamide adenine dinucleotide (NADH), which is associated with oxidative phosphorylation as a maker for cutaneous healing [107]. Direct exogenous fluorescent imaging, for dressing application, has also been applied thru the use of fluorescent dyes injected into the systemic system, and the dyes are visualized in the vasculature of burn wounds to assess wound depth [108]. Also noteworthy is an elegant approach to incorporation of fluorgenic esterase sensors demonstrating potential as an in situ sensor functionality that addresses challenging signal and diffusion issues and can be incorporated into various cellulosic dressing and substrate designs using a chemo-enzymatic approach [109].
A point of care diagnostic approach for chronic wound dressing selection is sensitive detection of the biomarker of interest [40]. Protease biomarkers that correlate to outcomes in wound healing include both MMPs and HNE as discussed above. Figure 4A, shows the interface of a fluorescent protease biosensor motif with a dressing. The dressing-sensor design directs uptake of wound exudate through a semipermeable contact layer onto the sensor surface where proteases react with the peptide substrate giving deposition of proteolytic reaction by-products on a barrier surface of the dressing tangential to the biosensor where it does not mix with wound fluid. Figure 4B portrays a series of images of the nanocrystals, nanocellulose composites, and nanocellulose aerogels, which offer a visual image of the disposition of properties benefitting sensor interface with a dressing as outlined below.
 Figure 4: Illustration of an intelligent semiocclusive protease dressing designed with fluorescent sensor. Image 3A shows a contact layer that absorbs wound exudate secretion and allows passage of wound fluid. The surrounding layers allow protease capture and fluorophore release and entrapment. Each layer of the bandage contributes to maintaining moisture via its open semi permeable structure. Image 3B shows the images of the nanocellulose-based sensors used as a biosensor layer for interface with a semiocclusive dressing (the images were acquired using a Sony Full HD 1080 mega pixels). Each nanocellulosic material has a device specific advantage: 1) the cellulosic nanocrystals can be affixed to the barrier layer, 2) the nanocellulose composite film can be layered between the top layer and barrier layer, and 3) the nanocellulosic aerogel can be placed between the barrier layer and top layer or applied directly to the top layer of a dressing as a separate sensor. View Figure 4
Figure 4: Illustration of an intelligent semiocclusive protease dressing designed with fluorescent sensor. Image 3A shows a contact layer that absorbs wound exudate secretion and allows passage of wound fluid. The surrounding layers allow protease capture and fluorophore release and entrapment. Each layer of the bandage contributes to maintaining moisture via its open semi permeable structure. Image 3B shows the images of the nanocellulose-based sensors used as a biosensor layer for interface with a semiocclusive dressing (the images were acquired using a Sony Full HD 1080 mega pixels). Each nanocellulosic material has a device specific advantage: 1) the cellulosic nanocrystals can be affixed to the barrier layer, 2) the nanocellulose composite film can be layered between the top layer and barrier layer, and 3) the nanocellulosic aerogel can be placed between the barrier layer and top layer or applied directly to the top layer of a dressing as a separate sensor. View Figure 4
The colorimetric and fluorescent tetrapeptide elastase substrates function as sensors through a respective chromophore (pNA) and fluorophore (AMC), which have different mechanism pathways. The para-nitroanilide (pNA) chromophore upon release absorbs at 405 nm, and when coupled with amplification by way of N-(1-Naphthyl) ethylenediamine (NEDH) elicits a colorimetric signal that gives enhanced sensitivity of detection, but notably slightly less than is emitted by the fluorescent signal found with AMC. On the other hand, AMC upon hydrolysis fluoresces by way of excitation at 360 and emission at 460 nm. Figure 2 shows the pathway of A) hydrolytic cleavage of pNA from the peptide substrate followed by reaction with NEDH amplification and B) hydrolysis to yield AMC from a tetrapeptide substrate with elastase. Comparatively, the AMC signal is more sensitive than the colorimetric signal when the transducer surface is cellulose nanocrystals. As noted previously [18] the sensitivity of the flurophore with the tetrapeptide substrate is approximately two-fold greater than the colorimetric response.
Figure 2A portrays the molecular sequence of events underlying colorimetric detection from hydrolytic cleavage of pNA from the tetrapeptide substrate via a two-step reaction. The sensor signal is prompted by the following: HNE hydrolyzes of the amide bond between valine and the COOH-terminal pNA whereupon an azide bond forms between pNA and NEDH. The detection signal of the pNA chromophore (405 nm) is amplified with formation of the azide bond between pNA and NEDH (545 nm), which results in a red shift in absorbance and increases the colorimetric signal [18,110].
Figure 2B demonstrates the one-step reaction that occurs upon HNE hydrolysis of the amide bond between valine and AMC subsequently resulting in a strong and sensitive fluorescence signal. It is notable that the one-step approach for the fluorescence signal as a detection method for a biosensor layer in a multilayered wound dressing is depicted in figure 5.
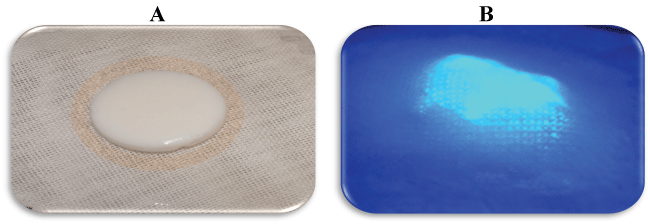 Figure 5: Depiction of nanocellulose aerogel peptide conjugates biosensor (diameter of 2.1 cm) with fluorescent protease activation interfaced in a multilayer bandage prototype with and without ultraviolet illumination. Image A demonstrates a gauze contact layer, barrier layer, and biosensor layer without ultraviolet activation. Image B depicts a barrier layer and gauze top layer above the biosensor layer with ultraviolet illumination. View Figure 5
Figure 5: Depiction of nanocellulose aerogel peptide conjugates biosensor (diameter of 2.1 cm) with fluorescent protease activation interfaced in a multilayer bandage prototype with and without ultraviolet illumination. Image A demonstrates a gauze contact layer, barrier layer, and biosensor layer without ultraviolet activation. Image B depicts a barrier layer and gauze top layer above the biosensor layer with ultraviolet illumination. View Figure 5
Nanocellulosic composites (NCC) may be considered appropriate for wound dressings and are porous paper-like films that have good gas barrier and mechanical properties [111]. Generating nanocellulose composites from wood cellulose nanocrystals and microfibrillated cellulose into thin film nanocellulose composites in ratios of 66/33 and 5050, respectively [95] allowed for improved dressing construction while maintaining a porous structure that permits air, moisture, and gas permeation [111,112]. As shown in figure 6 the porous nature of the nanocomposites resulted in favorable water vapor permeability of ~553 gm-2d-2 and is commensurate with levels associated with gas exchange at the wound bed. Thin films typically have a low permeability MVTR of 400-800 gm-2d-2, a moderate permeability MVTR of 800-3000 gm-2d-2, and a high permeability MVTR ranging from 3000 gm-2d-2 or even higher [113]. The composition of the materials influences the physical structure including the porosity, which alters the MVTR values [114,115]. The MVTR properties of the commercial gauze (677 gm-2day-1), wNCC (553 gm-2day-1), and commercial films (394 gm-2day-1) [116], which shows the wNCCs are in range with the planar commercial gauze and films for wound dressings. The wNCCs MVTR results are promising for use as a second layer detector interface to a chronic wound dressing.
 Figure 6: A bar graph depiction of the relative moisture vapor transmission rates (MVTR) for a nanocellulose composite film i.e. wNCC 50/50 (wood cellulose nanocrystals and microfibrillated cellulose) thickness ~300 micrometers, commercial gauze dressing material (36" wide folded to 4 ply (8"), 24/20 thread count, DeRoyal Textiles, Camden S.C., commercial film (Bioclusive, Johnson & Johnson, New Brunswick, N.J.) 0.077 mm thick. View Figure 6
Figure 6: A bar graph depiction of the relative moisture vapor transmission rates (MVTR) for a nanocellulose composite film i.e. wNCC 50/50 (wood cellulose nanocrystals and microfibrillated cellulose) thickness ~300 micrometers, commercial gauze dressing material (36" wide folded to 4 ply (8"), 24/20 thread count, DeRoyal Textiles, Camden S.C., commercial film (Bioclusive, Johnson & Johnson, New Brunswick, N.J.) 0.077 mm thick. View Figure 6
It was observed that protease sensors made from the films had lower protease detection sensitivity of 0.25 and 0.125 U/mL, respectively when compared with the cellulose nanocrystal-based sensors [95]. The lower sensitivity is thought to be due to aggregation of the nanocellulose during the preparation process, which yields a lower specific surface area of 0.021-0.035 m2g-1. As shown in table 1 the incorporation of cellulose nanocrystals into the nanocomposite imparts a negatively charged surface [101] that functions to sequester excess positively charged proteases.
Nanocellulosic aerogels (NA) are lightweight solid materials with low densities, high SSA, and an interconnected open porous structure [117,118], which make them attractive for use in semiocclusive dressings. Nanocellulosic aerogels may be prepared from a variety of cellulose fiber sources including wood and cotton. The interconnected open porous structure enables gas exchange between the wound bed and environment [24], membrane permeability [119,33], and prevents wound dehydration by way of promoting a high water vapor transmission rate [24]. Furthermore, the wettability and swelling properties of the NA promotes a hydrophilic and absorbent structure conducive to absorbing wound fluid. In addition as shown in table 1, the NA is a potential sequestrant of HNE due to the NAs negative surface charge of ~-24 mV [52,102,103]. The high SSA, 162.9 m2/g-1, of the nanocellulosic aerogels enables a higher loading of the sensor molecules (elastase tripeptide substrate) and increases sensitivity of the sensor by increasing the transducer surface area for protease binding. Figure 5 shows the NA biosensor interfaced with a semiocclusive wound dressing. The sensitivity studies of the NA peptide conjugate show the biosensor detects HNE as low as 0.125 U/mL.
The nanocellulosic biosensors discussed here and depicted in figure 4B are viewed as advantageous to the dressing interface based on the relative structure, SSA, and sensitivity as a biosensor component of a multilayered dressing. The nanocrystals exist in a powder form that is not readily adaptable in a wound dressing prototype as are the planar nanocellulose composites and nanocellulose aerogel. On the other hand the free particle nature of the nanocrystals maximizes the interaction with the human neutrophil elastase enzyme by virtue of the higher SSA thereby yielding a higher sensitivity to detect HNE present in chronic wound fluid. On the other hand, the incorporation of the nanocrystals into the wNCCs to create films affords a planar structure but a 5000-8000 fold reduction in the SSA occurs due to the aggregation of the nanocrystals blended with microfibrillated cellulose. The reduction in the SSA parallels the lower level of sensitivity; however, the planar but porous structure of wNCCs permits the permeation of moisture vapor and gaseous molecules, making it suitable for dressing interface as a biosensor layer. Nevertheless, the NA structure is also a compatible component of a multilayered dressing and offers a SSA with only a 1.14 fold reduction compared to the nanocrystals. Contrary to the SSA value, a higher sensitivity of 0.125 U/mL is observed versus the lower 0.05 U/mL compared to the cCNC, which may potentially be attributed to the pore sizes restricting the HNE enzymes penetration into the NA structure in order to interact with peptide substrate.
Even though the sensitivity of the nanocomposite and aerogel biosensors are at borderline levels for previously reported titers of elastase in chronic wounds [120] they are viewed as beneficial biosensors capable of detecting and sequestering HNE, and can be applicable as a dressing that can monitor wound saturation of HNE versus a preliminary way to detect the onset of a developing chronic wound. The benefit to both applications can provide information as to how the wound is healing when a new method of treatment is selected.
Biosensor development is undergoing exponential growth in the field of health care. In the area of point of care diagnostics, wound care developments with in situ detection that is compatible with wound dressing structure and function promises to change the face of wound care and help address the rapidly rising number of chronic wound patients worldwide. Here we have explored, in the context of the area of point of care diagnostic technology and dressing development, the comparative properties of nanocellulosic-based protease sensors for their potential interface properties with chronic wound dressings. Three nanocellulosic materials (nanocrystals, nanocellulose composites, and nanocellulose aerogels) were selected and evaluated for their ability to serve as a biosensor component for a multilayered chronic wound dressing. The nanocellulosic materials surveyed offer ideal properties including SSA, permeability, and surface charge that confer a compatible function to protease sequestrant semiocclusive wound dressings in the removal and detection of harmful proteases in the chronic wound.
A tetrapeptide (cCNC and wNCCs) and tripeptide (NA) substrate were immobilized onto transducer surfaces to generate the biosensors examined here. Each nanocellulosic biosensor was assessed for its ability to detect HNE and for its level of sensitivity in the presence of HNE. The relevant molecular and enzyme kinetic attributes suggest that negatively charged nanocellulose promotes efficient uptake of positively charges proteases. All of the biosensors were effective at detecting HNE at levels comparable to those found in chronic wound fluid or as may be expected to be taken up in a dressing. Furthermore, the anionic surface charge of the cellulosic materials provides an uptake mechanism for cationic human neutrophil elastase and is compatible with protease lowering dressings in that regard. Therefore, a single dressing motif based on different nanocellulosic materials could both sequester harmful proteases while serving to detect their presence as well as signaling dressing saturation.
Each of the biosensors, discussed here, has physical properties similar to commercially available thin films, hydrogels, and hydrocolloid dressings. Therefore, interfacing these types of nanocellulose-based biosensors with a semiocclusive dressing can be construed as compatible with detection of other markers of clinical interest as well, through substitution of the biomolecule needed for the desired point of care diagnostics or theranostic interest. Future work will look to fine tuning the structure function relationships of sensor and dressing interface.
The biosensors presented herein are promising in view of nanocellulosic-based materials with a high SSA, permeability, hydrophilicity, absorbent structure, and anionic surface charge. The combination of these features in a single sensor will increase the sensitivity threshold and enhance uptake of the biomarker.
The U.S. Department of Agriculture financed this project. We thank Dr. Alfred D. French for the X-Ray Diffraction studies that yielded the nanocrystallite model.