Introduction: Problematic skin conditions such as atopic dermatitis, acne vulgaris, and psoriasis are directly linked to microbial imbalance or 'dysbiosis'. Alterations of skin microbial communities along with other contributing factors, cause skin barrier dysfunction triggering immune and inflammatory responses. Acne is a prevalent problematic skin disorder characterized by unpleasant inflammatory lesions (e.g., pustules, papules), scars, and erythema which often cause psychological distress in acne patients. Common medications aim to reduce and control acne symptoms. However, most of these agents are associated with side effects and high cost. Therefore, alternative treatments are needed.
Methods: The aim of this pilot-study was to investigate the efficacy and safety of a novel product containing probiotics and hyaluronic acid, "InfiniteSkin™ Microbiome Serum", for the treatment of subjects with acne-prone skin or mild to moderate acne. The study involved twenty-nine healthy volunteers with such characteristics. Enrolled subjects were asked to apply "InfiniteSkin™ Microbiome Serum" nightly for 28 days. Clinical assessments to evaluate number of papules or pustules, extension of the facial area involved by acne, erythema and skin dryness were performed by a dermatologist. Skin hydration was measured with the MoistureMeterSC. Safety and patient's satisfaction were assessed by self-perception questionnaires.
Results: Topical application of "InfiniteSkin™ Microbiome Serum" reduced the typical manifestations of acne while improving skin hydration after 14 and 28 days of treatment. The product was well tolerated as no side effect was reported. According to the self-perception questionnaires outcome, patients were satisfied with the treatment.
Conclusions: Topical application of "InfiniteSkin™ Microbiome Serum" for 28 days resulted in reduced acne manifestations and improved skin hydration. The product was well tolerated as no adverse effects were reported up to 30 days from the end of treatment. Study participants expressed a favorable perception of the treatment effect.
Acne vulgaris, Hyaluronic acid, Lactobacillus, Probiotics, Skin, Topical application
Colony-Forming Units (CFU); Confidence Interval (CI); hyaluronic acid (HA); Least Significant Difference test (LSD)
Skin diseases are the 4th leading cause of non-fatal disease burden in humans. However, despite their visibility, their impact is often underestimated [1,2]. Acne (also known as acne vulgaris) is a problematic skin condition with an estimated global prevalence of 9.38% [3,4]. Often starting in preadolescence, and persisting into adulthood, acne affects about 85% of adolescents [5].
In general, the pathogenesis of acne is characterized by hyperproliferation and abnormal differentiation of the follicular epithelium, excess sebum production, inflammation, over-proliferation, and biofilm formation of Cutibacterium acnes (C. acnes, also known as Propionibacterium acnes) [6], the most prevalent bacterium in the pilosebaceous follicles of the skin [7]. Typical signs of acne disease are comedones, papules and pustules. Other symptoms include scars, erythema, and hyperpigmentation with or without soreness, itching, and pain [8,9]. An important effect of problematic skin is also related to poor quality of life. Indeed, acne can have a negative impact on social life, self-esteem and body image of individuals and it is often associated with depression, anxiety, embarrassment, and social inhibition [10]. Considering the prevalence and chronicity of the disease, the annual cost of acne treatment is quite high [8].
The main goal of acne treatment is to control and treat existing acne lesions, to prevent permanent scarring as far as possible, to limit the duration of the condition and to minimize psychological disorders [11]. Some topical lotions containing probiotics already demonstrated the capability of lactic acid bacteria to affect the skin microenvironment, impairing the C. acnes proliferation and reducing the inflammation caused by the bacteria, even if the underlying mechanisms are not fully understood [12,13]. Therefore, a variety of cosmetic products containing probiotics, are currently available on the market for the treatment of problematic skin. However, too many products fail to comply with the characteristics required to be called probiotic. Many false claims and rampant misuse of the term has resulted in mainstream consumer channels providing incorrect information to consumers [14].
Hyaluronic acid (HA), a key ingredient of many cosmetic products, has been widely used in skin care for its well-known hydrophilic and moisturizing proprieties [15]. Furthermore, HA has the capability to modulate and reduce the sebum production in oily skin subjects [16].
"InfiniteSkin™ Microbiome Serum", a new product developed by LAC2βiome S.r.l (Milan, Italy), for the first time combines the advantages of live probiotics, expected to reduce skin dysbiosis (i.e., acne extension), with the properties of HA, a natural skin moisturizer and modulator of sebum production [16]. In the present study, we report the first evidence regarding efficacy and safety of "InfiniteSkin™ Microbiome Serum" topical application in volunteers with acne-prone skin and with the presence of acne from mild to moderate entity.
Lactobacillus paracasei m.biome LiveSkin88TM (DSM 33788) was provided by LAC2βiome S.r.l. For administration, lyophilized Lactobacillus paracasei m.biome LiveSkin88TM at a dosage of no less than 8 billion colony-forming units (CFU) at time of activation was mixed with a solution of aqua propanediol, sodium hyaluronate, phenoxyethanol, maltodextrin, 1,2 hexanediol, caprylyl glycol, sodium anisate, hydrolyzed hyaluronic. Prior application, the product is activated as follows:
- tear off tamper evident tab,
- compress the plunger with the palm of the hand and shake the bottle for 5-10 seconds to completely disperse the powder in the liquid.
Product application is performed according to the following instructions:
- product is to be applied nightly
- product is to be applied as last step in the nightly routine
- shake the bottle for 2-3 seconds before each application
- take 2 pipettes of serum, place the serum in the palm of the hand, and apply it on the face and neck. Let it dry
- wash the face in the morning.
Volunteers were recommended to keep the product in the refrigerator (4 °C) after reactivation and after each application. Each bottle has 7 applications; each subject was supplied with the appropriate number of bottles to cover the 28-days treatment.
This study was conducted in compliance with the ethical principles originating in or derived from the Declaration of Helsinki and in compliance with Good Clinical Practice Guidelines. The study was carried out with the involvement of healthy volunteers, in a non-hospital setting and under the supervision of a dermatologist. All Subjects provided signed informed consent.
The volunteers participating in the study were selected from a panel of healthy Subjects at Neotron, Stradello Aggazzotti, 104, 41126 Modena - Italy. Inclusion criteria for the trial were determined as follows: age between 18 and 40 years old, acne-prone skin and with the presence of acne from mild to moderate entity, healthy without skin diseases or allergy/intolerance to cosmetics or drugs, agree to follow the study procedures and respect the control steps. Subjects under topical or systemic treatment with any drug that could influence the outcome of the efficacy study, or Subjects affected by skin diseases, pregnant or lactating women, and Subjects with intolerance to drugs and/or cosmetics were excluded.
Each volunteer signed an informed consent to enter the study and was given an explanatory sheet with the conditions for using the investigational product. According to the inclusion/exclusion criteria, 30 volunteers with acne were selected.
The aim of this 4 week-pilot clinical study was to evaluate the efficacy and safety of the product "InfiniteSkin™ Microbiome Serum" after a 14- and 28-days treatment through clinical evaluation, instrumental measurement of the skin hydration and self-perception questionnaires.
Before the beginning of the study, the Investigator delivered to the volunteers the investigational product and the informative form. Each Subject, read and countersigned the informed consent to take part to the study. Volunteers were asked to apply the product according to the product's instructions (see Methods). For the entire duration of the study, volunteers were asked not to apply on the analyzed skin area cosmetic products different than the one under study.
At time 0 and after 14 days, 28 days of treatment, the following evaluations were carried out:
- Photographic documentation with a digital camera.
- Clinical evaluation performed by dermatologist regarding the facial area affected by acne, the number of pimples, skin redness (erythema) and skin dryness.
- Instrumental measurements with the MoistureMeterSC (Delfin) to evaluate the surface hydration of the skin in a defined area of the face.
- After 14-days, 28 days of treatment and after 30 days from the end of the study, volunteers were asked to assess the treatment, in terms of perceived efficacy and side effects, answering a questionnaire.
This study was conducted in compliance with the ethical principles originating in or derived from the Declaration of Helsinki and in compliance with Good Clinical Practice Guidelines. Due to the cosmetic nature of the investigational product, ethics committee approval was not required. The study was carried out with the involvement of healthy volunteers, in a non-hospital setting and under the supervision of a dermatologist. All Subjects provided signed informed consent before any study procedure was performed.
Digital images were taken of each Subject (right side, left side, and center views) using the Canon IXUS 180. Prior to photography procedure, volunteers were asked to remove makeup and jewelry. Photographs were consistent in content, camera angle, distance of the camera (photographer distance; Subject distance from background), framing and exposure, and intensity of the illumination source.
The data of all the following evaluations were collected at baseline, after 14 days and after 28 days of treatment with the investigational product.
Clinical assessment of each volunteer was performed by a dermatologist to verify variations in terms of number of pimples/or pustules present, extension of the facial area involved by acne (clinical score from 0 to 3 where 0 = no area involved, 1 = < 25% of the face, 2 = between 25% and 50% of the face, 3 = >50% of the face), red areas/erythema (clinical score from 0 to 4 where 0 = no evidence of erythema, 0,5 = barely noticeable erythema, 1 = slight erythema, 2 = moderate redness, 3 = strong uniform redness, 4 = fiery redness), skin dryness (clinical score from 0 to 4 where 0 = absent dryness, 0,5 = very slight dryness, 1 = slight dryness, 2 = moderate dryness, 3 = severe dryness, 4 = severe dryness with visible peeling). During the clinical evaluation, the dermatologist checked there were no reactions in progress and asked the volunteers if they experienced any adverse reactions during the period of use. This information was only recorded in presence of reactions.
There were no adverse reactions during the study.
To evaluate the surface hydration of the skin in a defined area of the face, instrumental measurements were performed in a defined area of the face treated with the investigational product. For each volunteer, three repetitions were carried out at each step with MoistureMeterSC (Delfin Technologies Ltd), a commercial device that measures (in a range of 0-150 arbitrary units) the moisture level in the outer layer of the skin and the ability of the skin to retain water (by indirect measurement of electrical capacitance, which depends on the water content of the stratum corneum) [17,18]. The measuring frequency of MoistureMeterSC is 1.25 MHz.
All measurements were done under rested conditions of the Subjects and at temperature conditions between 20 °C and 25 °C and relative humidity between 40% and 70%.
For the self-perception questionnaire, volunteers were asked to assess the treatment, in terms of perceived efficacy and side effects, by answering a 7-item self-perception questionnaire. Four questions (overall liking, improvement in skin condition, reduction in pimples and their appearance, tested treatment soothes the redness/inflammation of the areas with pimples) were rated with a score from 1 to 5 (1 = not at all satisfied, 2 = slightly satisfied, 3 = so so satisfied, 4 = moderately satisfied, 5 = fully satisfied).
Safety was assessed by a) monitoring of adverse events during the study through clinical examination and b) self-perception questionnaires after 14 days, 28 days of treatment and after 30 days from the end of the study.
For the explorative nature of this pilot study, no hypothesis was tested and, therefore, no sample size calculation was performed. A sample size in the range of 20-30 volunteers was considered to be adequate for the purpose of this study [19]. The aim of this pilot study was to obtain sufficient preliminary data on efficacy and safety of the product, that will be useful to design a controlled study involving a larger cohort of patients.
Kolmogorov-Smirnov test was performed to check the normality of data distribution. For not normal distribution, Wilcoxon test for paired data was chosen for providing results regarding statistical significance on the data. For normal distribution, t-test for paired data was chosen for providing results regarding statistical significance on the data. The data were showed as percentage (%) of Subjects showing each potential assignable score.
For the self-perception, volunteers' evaluations were subjected to variance analysis and to the Least Significant Difference test (LSD). A two-tailed value of p < 0.05 was considered statistically significant.
For the number of papules and/or pustules, the Instrumental evaluation of skin hydration and the self-perception evaluations, the data were showed as mean value with a 95% Confidence Interval (CI).
Twenty-nine (n = 29) of the 30 healthy volunteers, aged 18-40 years old (mean age 24; 23 female and 6 male) with acne-prone skin and with the presence of acne from mild to moderate entity, participated in this pilot study. One Subject dropped out because of personal reasons (moved to another city).
The treatment efficacy after 14 and 28 days of application, supported by photographic documentation (Figure 1a), resulted in a statistically significant improvement in the number of papules and/or pustules if compared with baseline value (Figure 1b; Wilcoxon test for paired data: p < 0.0001).
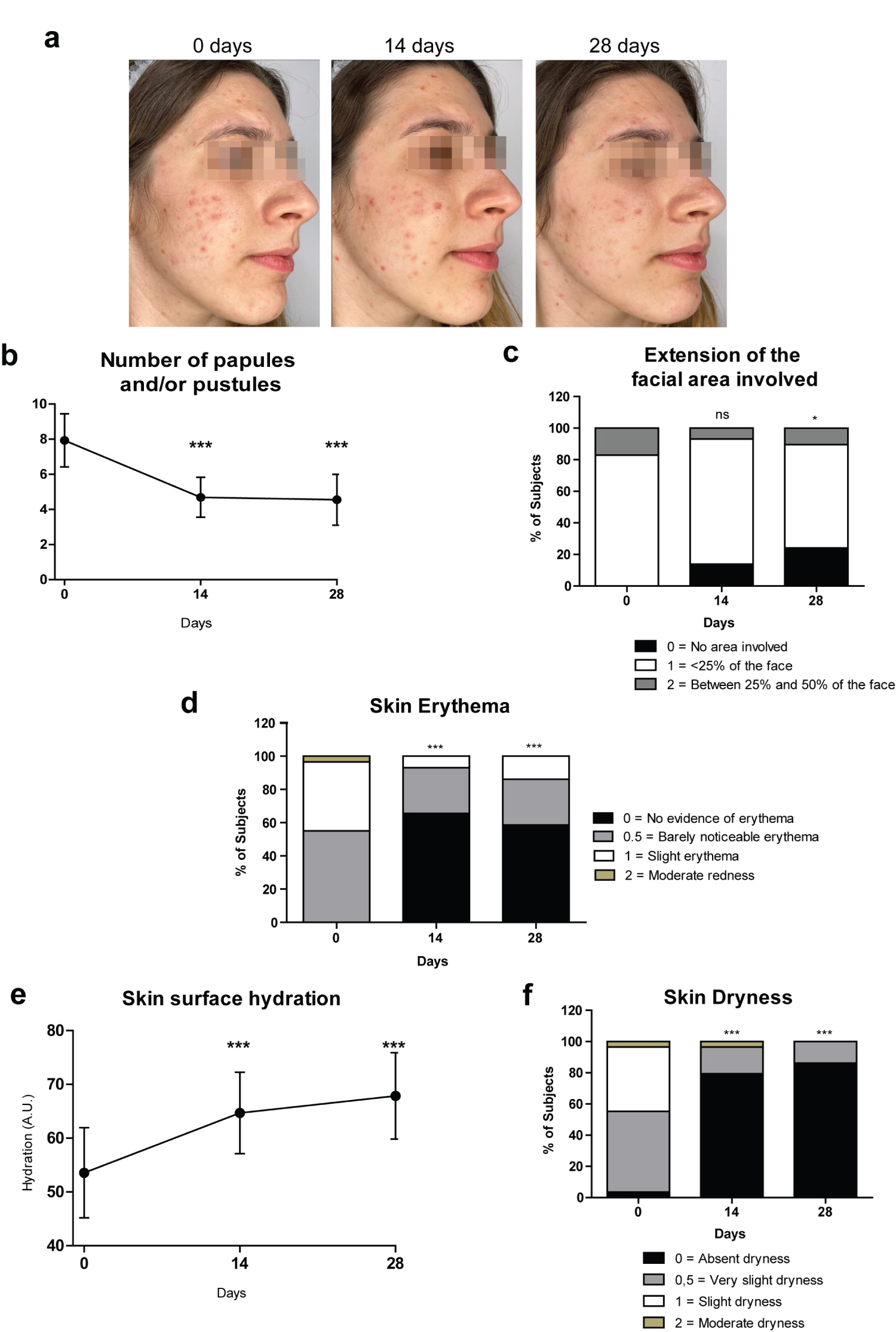 Figure 1: "InfiniteSkin™ Microbiome Serum" supports skin normalization.
Figure 1: "InfiniteSkin™ Microbiome Serum" supports skin normalization.
(a) Representative pictures of a female volunteer at baseline (0 Days) and after 14 Days and 28 Days treatment with "InfiniteSkin™ Microbiome Serum"; (b) Line graph showing the number of papules and/or pustules at the indicated time points. Results are shown as mean values with a 95% Confidence Interval (CI); (c) Bars graph showing the % of Subjects with the observed clinical scores used to measure the extension of the facial area involved by acne, at 0 Days, 14 Days and 28 Days (clinical score from 0 to 3 where 0 = no area involved, 1 = < 25% of the face, 2 = between 25% and 50% of the face, 3 = > 50% of the face; no one shown score 3); (d) Bars graph showing the % of Subjects with the observed clinical scores used to assess erythema at baseline and after 14 and 28 days of treatment (clinical score from 0 to 4 where 0 = no evidence of erythema, 0,5 = barely noticeable erythema, 1 = slight erythema, 2 = moderate redness, 3 = strong uniform redness, 4 = fiery redness; no one showed score 3 or 4); (e) Line graph of the delta hydration values (arbitrary units: A.U.) at 0 Days, 14 Days, 28 Days. Skin surface hydration measurements were obtained with the MoistureMeterSC device. Results are shown as mean values with a 95% Confidence Interval (CI); (f) Bars graph showing the % of Subjects with observed clinical scores used to evaluate skin dryness at the indicated time points (clinical score from 0 to 4 where 0 = absent dryness, 0,5 = very slight dryness, 1 = slight dryness, 2 = moderate dryness, 3 = severe dryness, 4 = severe dryness with visible peeling; no one showed score 3 or 4).
Significance levels in comparison with baseline values as tested by Wilcoxon signed-rank test for paired data (number of papules and/or pustules; extension of the facial area; erythema; skin dryness: *p < 0.05, ***p < 0.0001, ns = not significant) or by t-test (hydration: ***p < 0.0001) according to the normal distribution of the collected data by each evaluation.
View Figure 1
After 14 days, the mean number of papules and/or pustules was 4.7 ± 3.0 (mean value ± SD) compared to 7.9 ± 4.0 at day 0. The improvement observed at day 14 was maintained after 28 days of treatment (mean value ± SD: 4.5 ± 3.8).
The clinical evaluation of the extension of the facial area involved by acne indicated a significant improvement of the clinical score after 28 days of application. At day 0, none of the Subjects were recorded with clinical score of 0 (no area involved by acne), 24 (82.8%) Subjects were assessed with clinical score of 1 (< 25% of the face involved by acne), and 5 (17.2%) Subjects with clinical score of 2 (area of the face involved by acne between 25% and 50%). However, after 28 days of treatment, 7 (24.1%) Subjects were assessed with clinical score of 0, while 19 (65.5%) Subjects with clinical score of 1, and 3 (10.3%) Subjects with clinical score of 2. During the study, no Subject was recorded with clinical score of 3 (> 50% of the face involved by acne). The distribution of scores regarding the area of the face involved by acne at day 0 and day 28 was statistically different (Figure 1c; Wilcoxon test for paired data: p < 0.05). The distribution of scores regarding the area of the face involved by acne at day 0 and day 14 was not statistically significant (Figure 1c; Wilcoxon test for paired data: p = 0.0704).
Clinical evaluation of erythema indicated a modest, although significant, improvement after 14 and 28 days of treatment (Figure 1c and Figure 1d; Wilcoxon test for paired data: p < 0.05). At day 0, no Subject was assessed with clinical score of 0 (no evidence of erythema), 16 (55.1%) Subjects were recorded with clinical score of 0.5 (barely noticeable redness), 12 (41.4) Subjects with clinical score of 1 (slight erythema), and 1 (3.5%) Subject with moderate redness. However, after 14 days, there were 19 (65.5%) Subjects with clinical score of 0, and 8 (27.6%) Subjects with clinical score of 0.5. Only 2 (6.9%) Subjects reported slight erythema. The distribution of scores regarding erythema at day 0 and day 14 was statistically different (Figure 1d; Wilcoxon test for paired data: p < 0.0001). The positive results were confirmed and remained stable after 28 days of application. No Subject was recorded with strong uniform redness or fiery redness (clinical score of 3 or 4) during the entire period of the study.
Instrumental evaluation of skin hydration was performed with the MoistureMeterSC (Delfin Technologies Ltd). For each volunteer, the hydration values were recorded at baseline (0 Days), 14 Days and 28 Days post treatment. At day 0, the hydration value was 53.5 ± 22.0 (mean value ± SD). At day 14, the mean value increased to 64.7 ± 19.9. Similar results were obtained after 28 days of application. Differences between day 0 and day 14 or day 28, in terms of variation of skin surface hydration were highly statistically significant (Figure 1e; Paired t-test: p < 0.0001). The results were confirmed by improved skin dryness on a clinical score as assessed by a dermatologist (Fig. 1f; Wilcoxon test for paired data: p < 0.0001). After 14 days, 23 (79.3%) Subjects were recorded with clinical score of 0 (no evidence of dryness), 5 (17.2%) Subjects with clinical score of 0.5 (very slight dryness) and only 1 (3.5%) Subject with clinical score of 2 (moderate dryness). The distribution of scores regarding skin dryness at day 0 and day 14 was statistically different (Figure 1f, Wilcoxon test for paired data: p < 0.0001). The positive results obtained on day 14 were confirmed after 28 days, although with higher percentage of Subjects with absent dryness (86.2%). The distribution of scores regarding skin dryness at day 0 and day 28 was statistically different (p < 0.0001).
No Subject was recorded with severe dryness or fairly firm skin (clinical score of 3 or 4) during the entire period of the study.
According to the self-perception questionnaire, overall, the treatment was fairly appreciated by the volunteers both after 14 and 28 days of treatment. The volunteers quite agreed in perceiving an improvement in the skin regarding the redness/inflammation of the area with pimples. Slightly lower, although still discreet, the degree of agreement for the statements "I perceive the condition of my skin improved" and "I see a reduction in pimples and their appearance" (Figure 2; no statistically significant difference was found between the two time points).
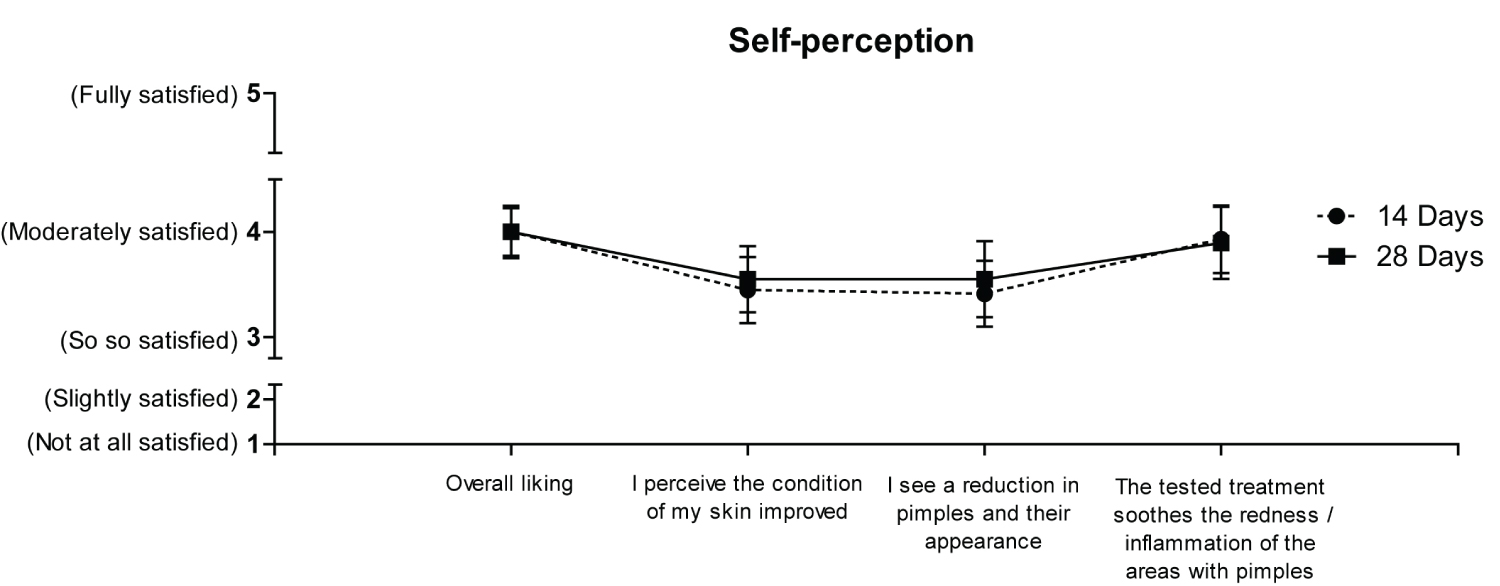 Figure 2. Self-perception outcome.
Figure 2. Self-perception outcome.
Graph of the self-perception items completed by enrolled volunteers (overall liking, improvement in skin condition, reduction in pimples and their appearance, tested treatment soothes the redness/inflammation of the areas with pimples) at 14 Days and 28 Days. For each question, the score was rated from 1 to 5 (1 = not at all satisfied, 2 = slightly satisfied, 3 = so so satisfied, 4 = moderately satisfied, 5 = fully satisfied). Results are shown as mean values for each question recorded at 14 Days (dotted line) and at 28 Days (solid line). Volunteers' evaluations were subjected to variance analysis and to the Least Significant Difference test (LSD). Results are shown as mean values with a 95% Confidence Interval (CI). No statistically significant difference was found between the two time points.
View Figure 2
The treatment was also well tolerated by all the Subjects participating in the study: no adverse skin reactions were monitored during the whole test period of 28 days and after 30 days from the end of the treatment.
Acne is a chronic skin inflammatory condition affecting adolescents and adults worldwide. It is characterized by visible manifestations of comedones, pustules, papules, scars, erythema, and hyperpigmentation [9,15]. As acne mostly affects the face, patients often suffer from psychological stress and decreased quality of life [10,20].
Multiple factors contribute to acne etiology including inflammatory process, altered sebum production, hyperproliferation, and follicular colonization by C. acnes which in turn contributes to acne inflammation [10,21,22]. First-line treatments for this dysbiosis state are topical antibiotics (e.g., clindamycin, erythromycin) used in combination with topical retinoids (e.g., tretinoin, adapalene and tazarotene) or benzoyl peroxide [10]. Systemic treatments are usually intended for patients unresponsive to topical agents [10,11].
Treatment with antibiotics is associated with skin dryness and pruritus. Furthermore, misuse of antimicrobial agents increases the risk of bacterial and alters the skin microbiome. Topical or oral retinoids are often associated with skin irritation and itching; typical side effects of benzoyl peroxide are redness, desquamation and burning sensation. Such complications together with high medical costs contribute to insufficient treatment adherence and patient's dissatisfaction [23-25]. For these reasons, alternative treatments are needed.
Probiotics are "live microorganisms which, when administered in adequate amounts, confer a health benefit on the host" [26]. Probiotic supplements have been widely used to treat gut disorders associated with dysbiosis. Reduction of the luminal pH and competition for nutritional sources are key mechanisms by which probiotics restore microbial balance thus alleviating intestinal symptoms [27].
Supplementation and topical application of probiotics have beneficial effects in the management of problematic skin conditions associated with dysbiosis. Indeed, probiotics have been proposed for the treatment and prevention of atopic dermatitis and to alleviate psoriasis symptoms [28-30]. More recently, reports from clinical studies showed that topical application of Lactobacillus has positive effects on acne by improving skin appearance [31], decreasing sebum secretion [32], and reducing comedones count [33]. The positive effects of probiotics have been mainly attributed to the inhibitory activities towards skin pathogens (e.g., Staphylococcus aureus), and commensal bacteria (e.g., C. acnes) [34-36].
Hyaluronic acid (HA) is a natural occurring polymer belonging to a group of heteropolysaccharides named glycosaminoglycans. HA is characterized by excellent ability to retain water molecules, high biocompatibility, and unique rheological properties [37,38]. HA is highly abundant in the skin where it acts as natural moisturizer. Indeed, almost all cosmetic products and dermal fillers that aim to improve skin elasticity, hydration, turgor, and aging signs, contain HA as functional ingredient [15,39,40]. HA-based formulations have been also proposed for the treatment of inflammatory skin diseases. Indeed, topical application of HA gels improves erythema and pruritus in patients with facial seborrheic dermatitis [41] and reduces sebum excretion in subjects with oily skin [16]. Invasive applications of HA (injections) are mostly used for the treatment of acne scars [42].
"InfiniteSkin™ Microbiome Serum" is an innovative product which combines the positive effects of both probiotics and HA. The results of this pilot study provide the first preliminary clinical evidence of its beneficial effects: to reduce typical acne signs and to improve skin hydration. A 14-days application of the product was sufficient to observe a significant reduction of the of papules and/or pustules (Figure 1b), a significant improvement of the skin erythema (Figure 1d) and skin hydration (Figure 1e and Figure 1f). These effects were maintained after 28 days of treatment. However, at least 28 days of application were needed to appreciate a significant reduction of the facial area involved by acne (Figure 1c). In the future, it will be interesting to evaluate the efficacy of "InfiniteSkin™ Microbiome Serum" after longer treatment periods. Importantly, no side effects were observed during the 28-days treatment period and most volunteers were satisfied with the product (Figure 2). Overall, these outcomes are in line with the current knowledge about the potential benefits and safety of probiotics and HA. The data obtained within this pilot-study will support the design of a controlled clinical investigation involving a large cohort of patients. In this way, we hope to deal with the current study limitations regarding the small sample size, and the single-arm design.
"InfiniteSkin™ Microbiome Serum" emerges as a novel product to control acne symptoms and to improve skin hydration. It would be interesting to evaluate "InfiniteSkin™ Microbiome Serum" contribution in reducing antibiotics use in acne patients, its effect in combination with other therapeutic agents, and its potential application for the management of other problematic skin conditions. It is worth noting that, due to the COVID-19 situation, volunteers enrolled in the trial used to wear face masks. It has been shown that wearing surgical masks is associated with dry skin and erythema [43,44]. However, even in this challenging context, the application of "InfiniteSkin™ Microbiome Serum" not only improved acne manifestations, but also skin hydration.
A 28-days topical treatment with "InfiniteSkin™ Microbiome Serum" resulted in a statistically significant improvement of acne signs in Subjects with acne-prone skin or presenting with mild to moderate acne. The product was also well tolerated and appreciated by study participants.
We thank the volunteers involved in the study.
The authors are grateful to Pier Andrea Nicolosi and Maria Giulia Manzione from 1MED for their constant assistance in preparing the manuscript
The pilot study and the journal's Rapid Service fee were funded by LAC2βiome S.r.l.
All named authors meet the International Committee of Medical Journal Editors (ICMJE) criteria for authorship for this article, take responsibility for the integrity of the work, and have given their approval for this version to be published.
All authors contributed to the study conception and design under the direction of Walter Fiore. LAC2βiome provided the study product. Alice Casari, Rima Al Sibai, Michela Mediani, Monica Molinari executed the clinical study, performed data collection and analysis.
All authors read and reviewed the manuscript. All authors approved the final manuscript.
Medical writing support under the guidance of the authors was provided by Pier Andrea Nicolosi, PhD, and Maria Giulia Manzione, PhD, at 1MED SA, Agno, Switzerland.
Walter Fiore is an employee of SOFAR S.p.A. and has worked as a consultant to LAC2βiome for this study. Alice Casari is employee at Skin Center. Rima Al Sibai, Michela Mediani, and Monica Molinari are employees at NEOTRON. This study was financially supported by LAC2βiome.
Informed consent was obtained from all Subjects involved in the study. This study was conducted in compliance with the ethical principles originating in or derived from the Declaration of Helsinki and in compliance with Good Clinical Practice Guidelines. All Subjects provided signed informed consent.
The raw data used and/or analyzed are available from the corresponding author on reasonable request.