Extramammary Paget Disease (EMPD) is a relatively rare condition with a reported incidence of 0.1 to 2.4 patients per 1,000,000 person-years [1]. It most commonly presents in the vulvar region of Caucasian females between 50 and 80 years of age [1]. EMPD clinically presents as an erythematous eczematoid rash which mimics inflammatory cutaneous disorders clinically and may result in delayed diagnosis [2]. EMPD can be classified into primary and secondary, with the latter representing spread of noncutaneous internal carcinoma by direct extension or by epidermotropic metastasis. The most common carcinomas causing secondary EMPD are anorectal and urothelial carcinomas [2]. We report a case of secondary EMPD of the peristomal skin in a patient with a distant history of cystectomy with ileal conduit for urothelial carcinoma who developed extensive peristomal eczematoid rash over the years.
An 87-year-old man with history of invasive transitional cell urothelial carcinoma of the bladder and left distal ureter status-post radical cystoprostatectomy and left distal uretectomy with uretero-ureterostomy and ileal conduit urinary diversion 14 years prior presented to our dermatology clinic with complaint of peristomal rash for many years. The patient reported the rash had been present for at least 5 years and was getting larger. It was described as pruritic and tender. The rash was initially attributed to urine leakage and had been treated as eczema, a fungal infection, and with wound and stoma care unsuccessfully for years before presentation. Patient had been using wound cleanser spray, normal saline, nystatin powder, and miconazole. The dermatologic exam revealed a large erythematous plaque with areas of maceration around the stoma involving most of the right abdomen extending to the pubis (Figure 1a and Figure 1b). A shave biopsy was performed from around the peristomal lesion for histopathologic diagnosis. The biopsy showed infiltration of the epidermis with large, atypical epithelioid cells with enlarged hyperchromatic nuclei and pale cytoplasm in solitary units and nests (Figure 2a and Figure 2b). These cells showed diffuse and strong expression of CK7 (Figure 2c) and GATA 3 (Figure 2d), and focal expression of CK20, CDX2, and BerEp4. The cells were negative for P63, CEA, Sox10, PSA, and TTF-1. No intracytoplasmic mucin was seen on mucicarmine stain and periodic acid-schiff stain was negative for fungal organisms. The morphologic and immunohistochemical features are consistent with secondary extramammary Paget Disease, consistent with involvement by the patient's known urothelial carcinoma of the bladder. These findings could represent local extension from recurrent cancer in the stoma itself or metastasis. A biopsy of the stoma and urothelial tract, as well as CT chest, abdomen, and pelvis with and without contrast were planned to assess for metastatic disease, however, soon after diagnosis was made the patient stated he did not want any invasive treatment or procedure. He was offered treatment with topical imiquimod which he denied. He subsequently passed away years later from other systemic complications.
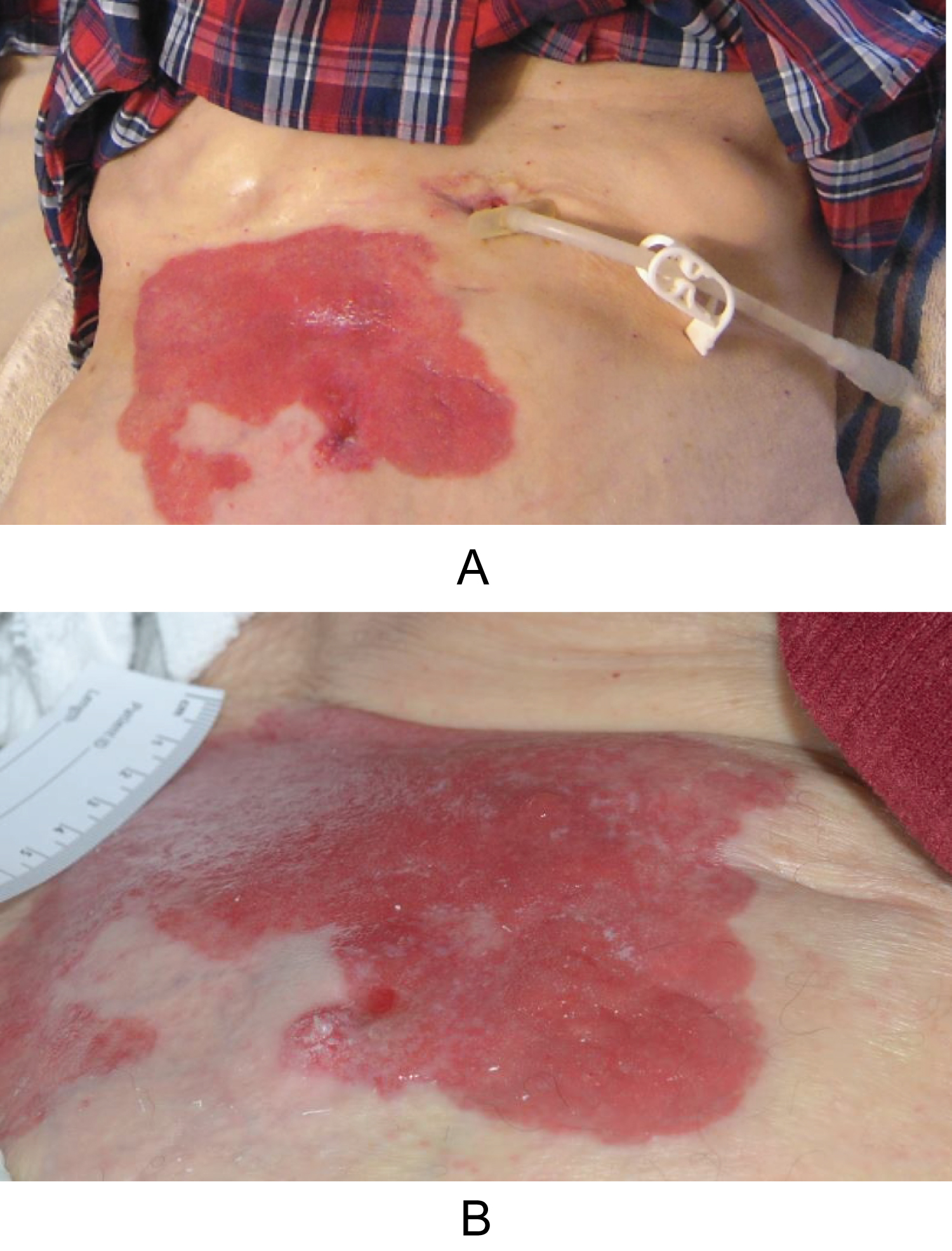 Figure 1: (a) Peristomal macerated, bright red plaque involving the right abdomen, adjacent to an uninvolved gastrostomy; (b) Peristomal macerated erythematous plaque involving the right abdomen.
View Figure 1
Figure 1: (a) Peristomal macerated, bright red plaque involving the right abdomen, adjacent to an uninvolved gastrostomy; (b) Peristomal macerated erythematous plaque involving the right abdomen.
View Figure 1
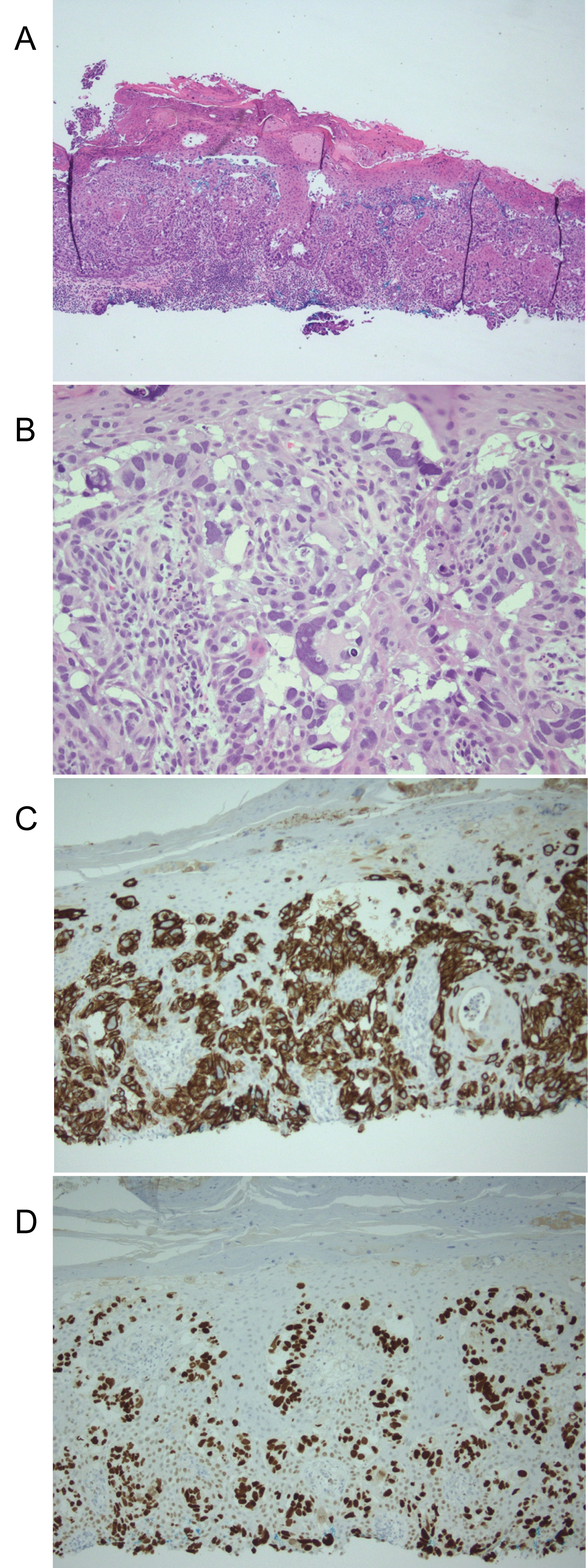 Figure 2: (a) Epidermis displaying infiltration by large, atypical epithelioid cells at low magnification (H&E, original magnification x40); (b) High magnification (H&E, original magnification x400) view of lesion displaying cells in solitary units and nests with enlarged, hyperchromatic nuclei and pale cytoplasm; (c) Diffuse, strong CK7 positivity (original magnification x200); (d) Diffuse, strong GATA 3 positivity (original magnification x200). View Figure 2
Figure 2: (a) Epidermis displaying infiltration by large, atypical epithelioid cells at low magnification (H&E, original magnification x40); (b) High magnification (H&E, original magnification x400) view of lesion displaying cells in solitary units and nests with enlarged, hyperchromatic nuclei and pale cytoplasm; (c) Diffuse, strong CK7 positivity (original magnification x200); (d) Diffuse, strong GATA 3 positivity (original magnification x200). View Figure 2
Currently, the exact mechanism underlying EMPD is unknown, but two widely known theories exist, namely the epidermotropic and transformation theories. The epidermotropic theory is more commonly accepted and suggests migration of adenocarcinoma cells into the epidermis, while the transformation theory postulates in situ malignant transformation of basal keratinocytes [3]. However, there remains a lack of evidence for either theory, and the pathogenesis of EMPD is considered controversial. There have been several studies which seek to identify biomarkers and signaling pathways in an effort to guide development of treatments. Specifically, the Msi1-mTOR pathway has been cited to promote Paget cell conversion of keratinocytes, suggesting the utility of Rapamycin as a novel therapy in patients expressing this phenotype [3]. Similarly, HER2 and downstream molecules in the RAS-RAF-MEK and PI3K-AKT-mTOR pathways have been identified as potential targets for treatment [4]. Though the current understanding of EMPD is improving, more research is needed to determine the implication of these findings on treatment options for patients with EMPD.
Extramammary Paget Disease largely affects body areas with a high apocrine gland concentration, most commonly the genitals, however, EMPD has rarely been reported to occur in body areas that are less populated with apocrine glands such as the trunk and cheeks [2]. Our literature review revealed one similar case of secondary extramammary Paget's disease around the cutaneous ureterostomy stoma after radical cystectomy in an 85-year-old man [5]. This patient presented by Kanda, et al. similarly developed a refractory dermatitis around his stoma on the abdominal skin. The time after radical cystectomy and cutaneous ureterostomy to development of cutaneous manifestations was four years in this patient, which represents a similar time frame to that of our patient. The patient's cutaneous malignancy was cured by skin excision around the cutaneous stoma, however he ultimately died of metastatic urothelial carcinoma [5]. Other cases exist of EMPD secondary to urothelial carcinoma, however the cutaneous manifestations presented on the apocrine-rich genital skin in these cases [6,7].
Many different treatment modalities exist for EMPD, depending on the presence or absence of metastatic disease and surgical-candidacy of the patient. In patients with primary EMPD, treatment typically consists of surgery with wide-local excision or Mohs Micrographic Surgery combined with neoadjuvant or adjuvant Imiquimod8. In patients with secondary metastatic EMPD, such as the one presented here, systemic chemotherapy is typically used, however the most effective regimen for metastatic disease has not yet been established. Secondary EMPD is difficult to treat and is associated with poorer outcomes. Proposed chemotherapeutic regimens include antimetabolites such as 5-Fluorouracil, alkylating antineoplastics like Cisplatin or Carboplatin, and alkaloids including Paclitaxel or Docetaxel [8]. Due to our patient's poor surgical candidacy and DNR status, he was offered topical Imiquimod therapy for palliation only which he denied.
The dermatologic exam of EMPD most commonly demonstrates a well-demarcated, erythematous plaque with or without overlying secondary skin changes. Due to the variation in clinical appearance, the initial differential for EMPD may include many different infectious and non-infectious entities, including dermatophytosis, irritant or allergic contact dermatitis, and inverse psoriasis. Adding to the diagnostic difficulty is the fact that patients may be asymptomatic or present with non-specific symptoms such as pruritus, tenderness, and burning [1]. Given its ability to mimic several inflammatory conditions, the diagnosis of EMPD is often delayed by many years. Early recognition of EMPD is paramount given its tendency to demonstrate multifocal and discontinuous subclinical extension, therefore it is recommended that all patients with pruritic eczematous lesions of apocrine gland-bearing skin who fail to respond to standard topical treatment after 4-6 weeks receive skin biopsy of the affected area for diagnosis [1,2].
In summary, it is important for the urologist and stoma care team to consider secondary cutaneous involvement by malignancy regardless of the time passed postoperatively. Dermatology referral should be considered for most dermatological conditions that fail to improve or resolve as expected despite treatment, and this is particularly true of treated cancer patients with persistent peristomal rashes.
We confirm that this work is original and has not been published elsewhere. All authors have approved the manuscript and agree with its submission. We have no conflicts of interest to disclose.