Kaposi sarcoma (KS) is a tumor originating from endothelial cells, both vascular and lymphatic, and is associated with human herpesvirus 8 (HHV-8). Four distinct KS subtypes have been defined: Classic, endemic, iatrogenic, and epidemic or acquired immunodeficiency syndrome (AIDS)-related KS. A fifth subtype has more recently been recognized in men who have sex with men (MSM) in the absence HIV infection. We report here on three cases of KS in HIV-negative MSM and review the literature. Two patients were treated with topical propranolol, resulting in a partial response in one and no response in the other. The third patient was treated with pegylated liposomal doxorubicin (PLD), which led to regression/flattening of multiple lesions as well as symptom relief. To the best of our knowledge, this is the first case series that reports on the use of topical propranolol and PLD in this KS subtype. Further research into this fifth subtype of KS is warranted as the optimal management strategy remains unclear.
Kaposi sarcoma (KS), Men who have sex with men (MSM), Topical propranolol, Pegylated liposomal doxorubicin (PLD)
Kaposi sarcoma (KS) is a tumor originating from endothelial cells, both vascular and lymphatic, and is associated with human herpesvirus 8 (HHV-8); also known as KS-associated herpesvirus (KSHV)). KS typically presents as asymptomatic purple, red or brown macules, plaques or nodules, with a multifocal origin. However, numerous morphologic variants have been reported [1]. Extra-cutaneous lesions can develop, sometimes in the absence of cutaneous lesions, and can affect the oral mucosa, lymph nodes and viscera.
Four distinct KS subtypes have been defined (Table 1), each with a different clinical presentation and prognosis: Classic, endemic, iatrogenic, and epidemic or acquired immunodeficiency syndrome (AIDS)-related [1-3]. A fifth subtype of KS has more recently been recognized. KS in HIV-negative immune competent men who have sex with men (MSM) is also called the MSM-related or nonepidemic KS. Although its exact pathogenesis is unclear, new insights on risk factors gradually become clearer. We report here on three cases of MSM-related KS seen at Ghent University Hospital and review the literature.
Table 1: Subtypes of Kaposi Sarcoma. View Table 1
All patients were referred to the department of dermatology with a histologically confirmed KS (Figure 1). The patients repeatedly tested negative for HIV antibody/antigen and are compliant with the sexually transmitted infection (STI) screening every 3 months. Main characteristics of the patients are shown in Table 2.
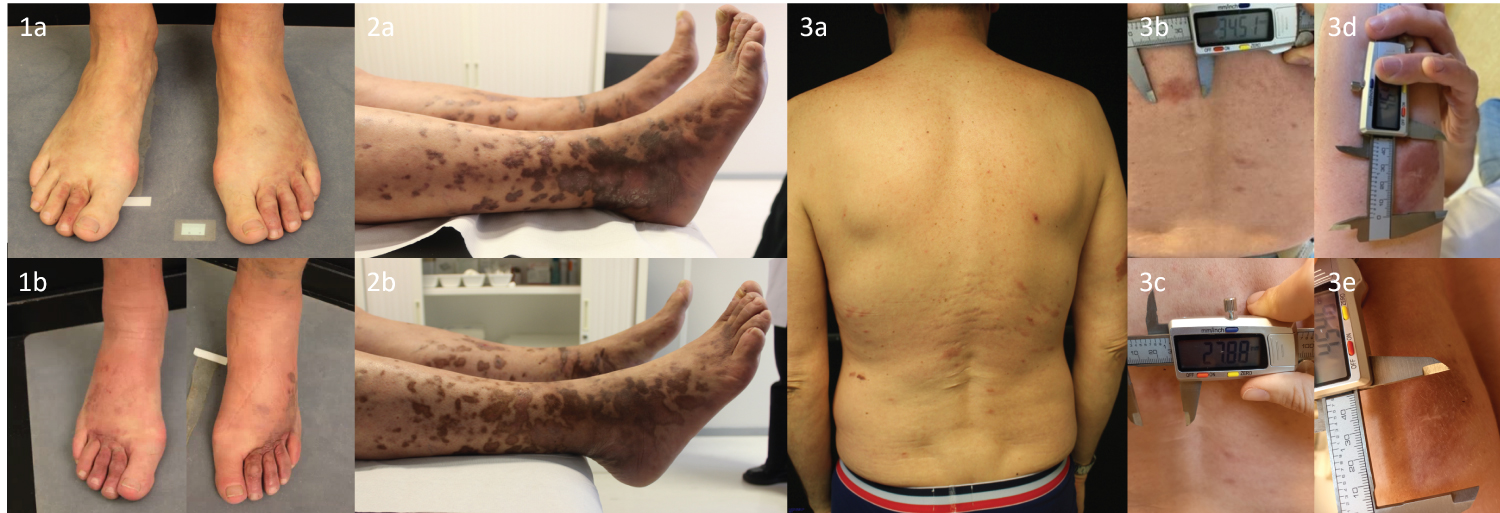 Figure 1: Clinical presentation of the cases.
Figure 1: Clinical presentation of the cases.
Case 1 at presentation (1a) and after 4 months of topical propranolol (1b). No response was seen. Case 2 at presentation (2a) and after 4 cycles of pegylated liposomal doxorubicin (2b). There was clinical improvement with regression/flattening of multiple lesions and the discomfort in the legs had resolved. Case 3 at presentation (3a overview, 3b back and 3d right upper arm) and after 3 months of topical propranolol (3c back and 3e right upper arm). The lesions showed some improvement.
View Figure 1
Table 2: Patients characteristics. View Table 2
A 65-year-old Caucasian man, who is taking tenofovir disoproxil/emtricitabine (TDF/FTC) as event driven pre-exposure prophylaxis (PrEP), presented with multiple red-brownish infiltrated lesions on the abdomen and extremities, primarily on the lower limbs. The lesions appeared shortly after he received systemic steroids for pruritus 3-4 months before. Additional blood examinations showed no signs of underlying immune deficiencies.
18F-fluorodeoxyglucose (FDG) positron emission tomography/computed tomography (PET/CT) showed no internal lesions. Due to the extent of the lesions, there was opted for a treatment with interferon alfa-2b. However, this was not available anymore. A trial with a timolol gel 0.1% was initiated on a lesion on the upper arm with partial regression after 4 weeks. Because of this partial response a treatment with propranolol 4% cream twice a day was started, however, no response was seen after 4 months.
This 64-year-old Caucasian male presented with multiple purple plaques and nodules on the extremities, causing a burning sensation. The lesions on the feet expanded after bilateral varicectomy. Laboratory work-up found low B-lymphocytes and a FDG-PET/CT showed no additional lesions. Due to the rapidly expanding lesions, systemic treatment with pegylated liposomal doxorubicin (PLD) was initiated, at a dose of 20 mg/m 2 every 2 weeks. After 4 cycles there was clinical improvement with regression/flattening of multiple lesions and the discomfort in the legs had resolved.
This 50-year-old Caucasian man presented with cutaneous lesions on his back, legs and right upper arm. Laboratory work-up found a CD4/CD8 ratio under the lower reference limit. Further investigations found no evidence for a primary immunodeficiency. FDG-PET/CT showed a pulmonary infiltrate which resolved spontaneously after several months. Further examinations, bronchoscopy and gastro- and coloscopy, showed no arguments for visceral disease or adenopathies. During his follow-up, his partner tested positive for HIV. Daily PrEP with TDF/FTC was initiated, after which both his genital herpes lesions and cutaneous Kaposi lesions improved according to the patient. Several months later there was slow progression of the lesions. Treatment with topical propranolol 2% cream twice a day was initiated. After 3 months the lesions showed some improvement. A large lesion, which started to ulcerate, on the right thigh was excised.
KS in HIV-negative immune competent men who have sex with other men has been recognized as a fifth subtype of KS: the MSM-related or nonepidemic KS [3-7]. This subtype usually occurs in young or middle-aged males who have sex with other men. It has a clinical presentation similar to classic KS with usually an indolent course and extension limited to the skin, although lymph node involvement has been reported in 5% of patients [6]. No immune deficiencies are present [2-6]. Our cases fulfill all of these criteria.
HHV-8 is required but not sufficient for the development of all types of KS as it is mainly modulated by the patient’s immune status and other still unknown factors. Although AIDS-related KS and iatrogenic KS are associated with conditions of decreased immunity, an impaired immune function is not well characterized in classic KS, endemic KS and MSM-related KS [2]. Studies have shown an increased seroprevalence of HHV-8 among MSM compared to heterosexuals [8]. In a recent Chinese study among 520 MSM, more male sex partners, lower income and being a student were associated with higher seropositivity for HHV-8. In addition, being coinfected with hepatitis C virus and syphilis resulted in higher risk for seropositivity [9]. This higher seropositivity has been attributed to the more frequent use of saliva as a lubricant in homosexual practices as saliva is the main harborer of HHV-8 [10-12]. It is therefore believed that the development of MSM-related KS may be secondary to the increased HHV-8 seroprevalence in this population [3,5].
Increasing anti-HHV-8 antibody titers have been reported as the disease progresses [13]. Little is known about the variation of antibody titers across the different stages of KS. However, detectable HHV-8 viremia is associated with disease progression and tumor burden [14]. Evaluation of plasma HHV-8 antibody titer and viral load might therefore be useful in the work-up of HIV-negative KS patients. Other factors that may play a role in developing MSM-related KS are aging and comorbidities, such as diabetes, as suggested by Lanternier, et al. as these are also thought to play a role in the development of classic KS [4]. In addition, systemic corticotherapy can induce the development of new KS lesions as illustrated by case 1 but may also increase KS related mortality [15]. One of our patients reported exacerbation of his lesions after a bilateral varicectomy.
An infection with HHV-8 is lifelong, similar to other herpes viruses, and there is currently no treatment for permanent eradication of the virus. At present, there are also no rigorously studied interventions which can prevent HHV-8 infection, mainly because the specific routes of transmission are not fully understood. MSM-related KS usually has an indolent course and treatment will depend on the extension and/or progression of the disease. As this subtype of KS is rare, most management options are obtained from case reports and small case series as large randomized clinical trials are not at hand, in contrast to AIDS-related KS. Treatment options previously used in MSM-related KS are listed in Table 3 [4,5,16-22]. To the best of our knowledge, the use of topical propranolol and PLD have not been reported before in this KS subtype. Beta-lytics, both systemic and topical, have been used in infantile hemangioma [23]. Systemic beta-lytics are also being explored as an adjuvant therapy for angiosarcoma [24]. Topical beta-lytics have been previously used in classic and AIDS-related KS with varying results. Beta-lytics could act via three different consecutive mechanisms: First they induce vasoconstriction, followed by inhibition of pro-angiogenic signals, and finally they promote endothelial cell apoptosis. In a study performed by Abdelmaksoud, et al. complete remission of KS lesions was obtained following application of 0.1% topical timolol gel [25]. However, Giorgio, et al. reported variable responses following propranolol 2% cream or timolol 0.5% application [26]. Furthermore, they demonstrated that topical propranolol was superior to timolol in treating KS lesions. No adverse events were reported in the prior studies nor in the presented cases. In one case described here there was only a partial response with topical propranolol in some cutaneous lesions. In the other patient no response was seen. Although differences in prescription composition and application frequency may account for different effects, these findings may also indicate that response to beta-lytics may differ between different KS subtypes. PLD has been predominantly used in AIDS-related KS with good results, even in advanced stages [27]. In our patient, PLD provided clinical improvement with regression/flattening of multiple lesions as well as symptom relief.
Table 3: Treatment options for MSM-related Kaposi sarcoma based on current literature. View Table 3
In conclusion, we report on three cases of MSM-related KS, considered as a fifth subtype of KS. To the best of our knowledge, this is the first case series that reports on the use of topical propranolol and PLD in this KS subtype. Further research into this fifth subtype of KS is warranted as it is currently unclear which factors play a role in its development and how to best manage it.
The patients in this manuscript have given written informed consent to publication of their case details.
No potential competing interests were reported by the authors.
This research received no external funding.
N.T. Hilhorst conducted the literature search and wrote the paper; B. Devocht conducted the literature search and wrote the paper; E. Verhaeghe proofread the paper and gave corrections; L. Lapeire collected the patients, performed the research, proofread the paper and gave corrections; L. Vandekerckhove proofread the paper and gave corrections; S. Desmet collected the patients, proofread the paper and gave corrections; MA. De Scheerder collected the patients, performed the research, proofread the paper and gave corrections; L. Brochez collected the patients, performed the research, proofread the paper and gave corrections.
All authors have read and approved the final manuscript.