Amelanotic melanoma is a type of cutaneous melanoma characterized by the absence of pigment. The rhabdoid features are an uncommon histological variant of malignant melanoma. Only a handful of primary rhabdoid melanoma cases have been reported in the literature. We describe here an additional case of a 60-year-old man with a primary pure dermal amelanotic melanoma with rhabdoid features and review the clinicopathological features of the previously reported cases of this rare entity. This work reviews and discusses newly acquired data and evidence on the clinical, pathological, immunohistochemistry, and prognosis of this unusual entity, in the hopes of assisting pathologists in correctly diagnosing it and avoiding confusion with other rhabdoid neoplasms.
Skin, Melanoma, Amelanotic, Rhabdoid phenotype
Amelanotic melanoma is a subtype of cutaneous melanoma characterized by the absence of pigment on clinical examination [1] and represents an atypical form that may not be easily recognized as malignant melanoma [2]. The histological variants of melanoma are numerous and among these rare variants, we find the rhabdoid phenotype. This variant has no prognostic significance but has importance because it may easily be mistaken for other tumors, with potentially serious consequences [3].
A 60-year-old Moroccan man with no medical history, presented with a 2-year history of an unpigmented reddish-white solitary tumor on the right cheek. On physical examination, the tumor measured 50 × 36 mm and was firm, reddish-white, and nodular with irregular contours and a multi-lobular surface (Figure 1A). The clinical differential diagnosis included squamous cell carcinoma, adnexal tumor, metastatic skin cancer, and soft tissue sarcoma. No palpable regional lymph node was identified. Two homo-lateral lateral cervical lymph nodes were enlarged to palpation with no other significant abnormalities at the somatic examination.
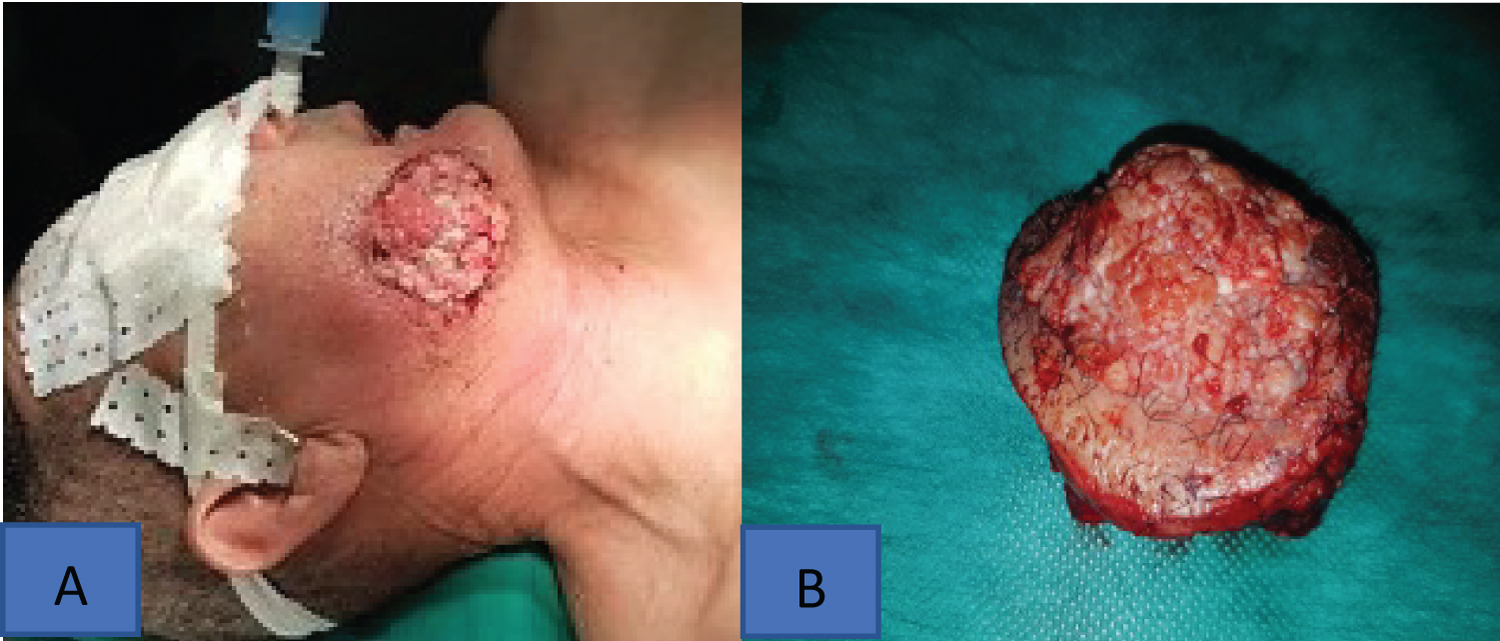 Figure 1: (A) Clinical picture (before surgery) of the tumor: A 5 cm firm, white-reddish nodular tumor with irregular contours and multi-lobular surface; (B) Gross pathological specimen of the tumor surrounded by a border of normal skin.
View Figure 1
Figure 1: (A) Clinical picture (before surgery) of the tumor: A 5 cm firm, white-reddish nodular tumor with irregular contours and multi-lobular surface; (B) Gross pathological specimen of the tumor surrounded by a border of normal skin.
View Figure 1
An incisional biopsy was performed, and the diagnosis favoured an undifferentiated mesenchymal malignant tumor with rhabdoid features. Immunohistochemistry was not performed because of the small size of the biopsy material. A computed tomography scan of the head was performed for preoperative evaluation and showed no obvious mandibular involvement by the lesion and detected two homo-lateral lateral cervical lymph nodes. No distant metastasis or other neoplasms were identified. The patient underwent wide excision of the tumor with clear margins extending to the internal oral mucosa and the homo-lateral cervical lymph nodes. Macroscopic examination revealed a firm exophytic tumor with irregular contours and a multi-lobular surface. The lateral and deep surgical margins were macroscopically negative. (Figure 1B). The cervical lymph node dissection noted 10 lymph nodes, the largest measuring 1.8 cm.
A histopathologic examination of the surgical excision revealed a nodular proliferation without ulceration composed entirely of proliferating sheets and nests of discohesive atypical rhabdoid and epithelioid cells with eccentric large, bizarre, and pleomorphic round to oval nuclei with anisocytosis, a single prominent nucleolus, and large hyaline cytoplasmic inclusions (Figures 2A and 2B). Mitotic figures were observed (> 10 per 10 high-power fields), and no evidence of melanin pigment or intra-epidermal component was identified in multiple sections. The tumor’s Breslow thickness measured 18 mm and its Clark level was 5. Pathological examination revealed no lymphovascular or perineural invasion. The surgical margins returned microscopically negative.
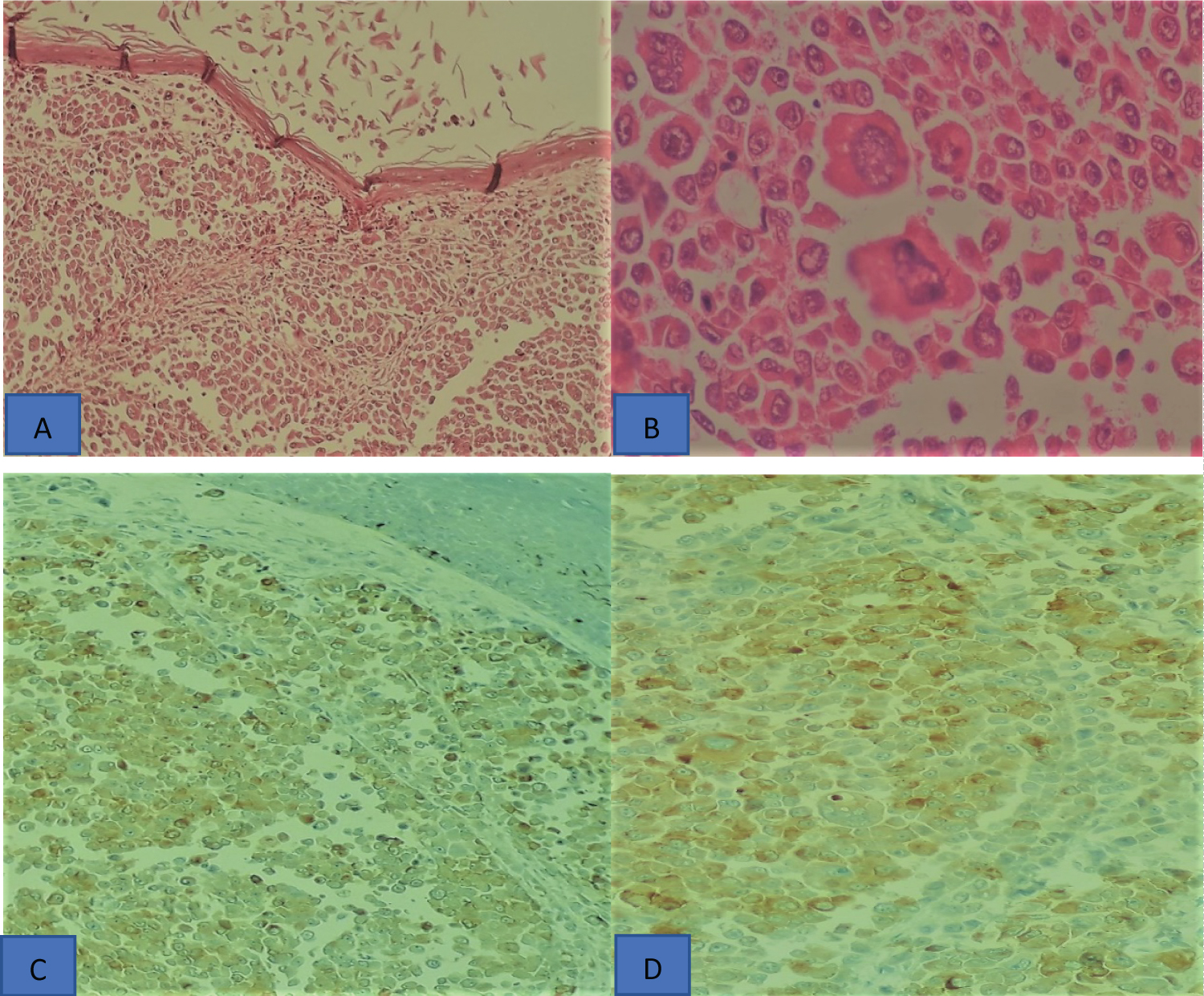 Figure 2: Histopathological aspects and immunohistochemical profile of rhabdoid melanoma (A) The tumor is composed by sheet and alveolar nests of large polygonal neoplastic cells with abundant eosinophilic cytoplasm without ulceration (Hematoxylin and eosin, ×100); (B) Tumor cells show enlarged, hyperchromatic, pleomorphic nuclei, with prominent nucleoli, mitoses, and abundant eosinophilic cytoplasm (Hematoxylin and eosin ×400); (C) Tumor epithelial cells are positive for s100 protein and (D) HMB45.
View Figure 2
Figure 2: Histopathological aspects and immunohistochemical profile of rhabdoid melanoma (A) The tumor is composed by sheet and alveolar nests of large polygonal neoplastic cells with abundant eosinophilic cytoplasm without ulceration (Hematoxylin and eosin, ×100); (B) Tumor cells show enlarged, hyperchromatic, pleomorphic nuclei, with prominent nucleoli, mitoses, and abundant eosinophilic cytoplasm (Hematoxylin and eosin ×400); (C) Tumor epithelial cells are positive for s100 protein and (D) HMB45.
View Figure 2
Among the 10 lymph nodes, histological examination revealed two metastatic nodes with capsular rupture. Immunohistochemical studies revealed focal positivity for S-100 protein (Figure 2C) and HMB-45 (Figure 2D). The neoplastic cells were negative for Melan-A, pancytokeratin (AE1/AE3), desmin, and smooth muscle actin. Based on these histological and immunohistochemical features, this tumor is diagnosed as primary cutaneous amelanotic rhabdoid melanoma. According to the eighth edition of the AJCC TNM staging manual for melanoma, the patient was classified as stage III C disease (pT4a, N2b, M0). After a multidisciplinary consultation meeting, immunotherapy was recommended, and the patient was referred to the oncology department where he underwent immunotherapy (pembrolizumab 200 mg/week). The patient is still alive 11 months after the diagnosis with neither local recurrence nor metastasis.
Melanoma is characterized by a capacity for a variety of cytomorphological and architectural features. It may be composed of balloon cells, small cells, signet-ring cells, or spindle, clear, plasmacytoid, or large pleomorphic cells [4]. Rhabdoid melanoma is a relatively rare malignant variant and in 1992, Bittesini described the first case of metastatic melanoma. [5]. Most cases have presented in a metastatic or recurrent form [6]. Clinically, this variant is often presented in the literature as a nodular amelanotic melanoma (as in our case) [7]. No predilection of age or sex was reported. The scalp, back, shoulder, and thigh are the most likely locations described in the literature, but the cheek is occasionally reported [8]. Histologically, the rhabdoid phenotype of melanoma typically comprises sheets of polygonal cells with round nuclei, vesicular chromatin, and abundant eosinophilic cytoplasm containing a large round eosinophilic hyaline inclusion [9].
The profile immunohistochemistry of rhabdoid melanoma is highly heterogenic. S-100 protein and HMB45 are expressed in many cases; however, this expression may be absent or weakly positive (as in our case) [10]. This heterogeneity may render the diagnosis difficult, particularly, when the melanin pigment is absent. Malignant extrarenal rhabdoid tumor, epithelioid sarcoma, rhabdomyosarcoma, myoepithelioma, myoepithelial carcinoma, and poorly differentiated carcinoma form the histological differential diagnosis of rhabdoid melanoma [11].
The prognosis and treatment of rhabdoid melanoma are similar to that of conventional melanoma [12].
we describe here an additional case of an amelanotic rhabdoid melanoma of the cheek. This entity is an uncommon tumor rarely reported in the literature. The clinical appearance is often misleading. The diagnosis of this entity is based on the morphological aspect and the immunohistochemical profile. The prognostic of the rhabdoid phenotype in melanoma is unknown. The rhabdoid phenotype of any malignant tumor, especially of the skin, should evoke this entity in the minds of pathologists even in the absence of melanin pigment as the prognosis and treatment differ compared to other tumors.