Diabetic retinopathy, Diabetic macular edema, Plant-based diet
Diabetic retinopathy (DR) is one of the major neurovascular complications of diabetes and is a leading cause of blindness in adults of working age group, 20-74 years [1]. From 1990-2010, DR ranked as the fifth most common cause of preventable blindness and fifth most common cause of moderate to severe visual impairment [2]. In 2018, the estimated prevalence of diabetes among adults in the United States was 10.2% (26.8 million cases) [3]. In 2019, the disease was responsible for 4.2 million deaths worldwide, as well as $760 billion in health care expenditure. Globally, 629 million persons are expected to have diabetes by 2045 [4].
In most patients, retinopathy develops 10 to 15 years after diabetes has been diagnosed. With increased prevalence of diabetes, more people are at risk for retinopathy, and greater resources are required to identify and treat these conditions. Given the disease burden, efficient and accurate diagnosis of diabetic retinopathy, risk assessment, treatment, and strategies for prevention are crucial.
The most clinically important risk factors for progression to vision loss include duration of diabetes, hyperglycemia, and hypertension. Besides these, hyperlipidemia and obesity are also found to be contributing risk factors in some studies [5,6].
The current practice in the management DR has been primarily aimed at its neovascular complications, and rests upon the use of laser photocoagulation, intravitreal anti-VGEF injections, and on occasion glucocorticoid therapy. However, these treatments are not curative and there is increasing evidence that anti-VEGF treatment is unlikely to improve retinal perfusion and may not prevent gradual progression of nonperfusion or loss of peripheral visual field associated with worsening diabetes-related eye disease [7]. Moreover, there are many short term and long-term complications associated with these treatments. Diabetic retinopathy is the result of the convergence of many metabolic abnormalities leading to retinal vasculature dysfunction, inflammation, and neurodegeneration [8]. DR has been considered a microcirculatory disease of the retina. However, there is emerging evidence to suggest that retinal neurodegeneration is an early event in the pathogenesis of DR [9].
Diet is a major contributing factor in developing retinopathy. The Rice Diet had been effective in reversing type 2 diabetes and retinopathy [10]. However, in clinical practice the implementation of plant-based diet is practically absent. Here is a case being presented where regression of diabetic macular edema (DME) is achieved by remission of diabetes with Plant-Based diet.
LL is a 61-year-old female presented to the hospital on 6/21/2015 due to syncope. She was found to be dehydrated. Her blood sugar was markedly elevated at 535 mg/dl, and HbA1c 10.8%. She was appropriately hydrated and started on medical treatment for newly diagnosed diabetes with Humalog insulin 3 units before each meal, Lantus insulin 12 units at bedtime. In the hospital, she received education on the management of diabetes and to follow an ADA diet. At the time of discharge from the hospital, she was on Lantus insulin 12 units sq at bedtime, Metformin 1000 mg po BID, and Glipizide 10 mg po BID. During the ensuing period, her HbA1c has stayed between 7.2% to 6.5%, and her antidiabetic medications were gradually reduced. An eye examination 6/29/2016 was found to be normal.
She has experienced blurry vison in the right eye greater than the left eye. An eye examination on 2/6/2019 revealed evidence of DME in the right eye and posterior vitreous detachment in the left eye. She was sent to a retina specialist and the above diagnosis were confirmed. She received Eylea (aflibercept) injection to the right eye. In the left eye, she was found to have mild non-proliferative diabetic retinopathy (NPDR), vitreous detachment without holes or tears; and these did not call for any treatment but close monitoring. In addition, she had bilateral hypertensive retinopathy and age-related cataracts. On 5/29/2019, she has required a second treatment of Elyea (aflibercept) injection to the right eye, and in addition, she received focal laser treatment in the same eye on 6/24/2019.
In the meantime, she has experienced symptoms of angina pectoris and was seen in consultation in our Cardiovascular Wellness Clinic on 4/10/2019 for evaluation and management. A stress/echo study was normal. She was recommended to adopt a Plant-Based Diet which is composed of vegetables, fruits, legumes, whole grains, seeds, and nuts. She was particularly recommended to avoid all animal-derived products, processed foods, dairy products, eggs, oils, and refined carbohydrates, to improve symptoms of angina, hypertension, hyperlipidemia, and the hope for achieving remission of diabetes [11].
In addition, she has participated in our Ornish Lifestyle Medicine (Intensive Cardiac Rehabilitation) program to reverse heart disease. This program consisted of an intensive education on Plant-Based diet, aerobic exercise, Yoga/meditation for relaxation, and group support. Each session consisted of 4 hours of supervised training by experienced staff for 18 sessions over 9 weeks (total of 72 hours). She made great progress with resolution of the symptoms of angina pectoris and de-escalation of antidiabetic medications with improved blood sugars [12].
Following the above lifestyle changes, she was able to go off most of the antidiabetic medications, except metformin. On 11/16/2019 her HbA1c has come down to 6.0%. Additionally, her blood pressure, blood cholesterol level, and weight had also reduced. Her current medications were - Alpha Lipoic Acid 1 capsule daily, ASA 81 mg daily, Biotin 1000 mg daily, Calcium 600 mg twice daily, Vit D3 1000 units daily, Vitamin B12 1000 mcg daily, Cozaar 25 mg daily, Metformin ER 1000 mg twice daily, and Crestor 40 mg daily. An eye examination on 12/11/2019, surprisingly showed resolution of DME in the right eye, and she has not required any further treatment for retinal disease (Table 1) (Figure 1 and Figure 2).
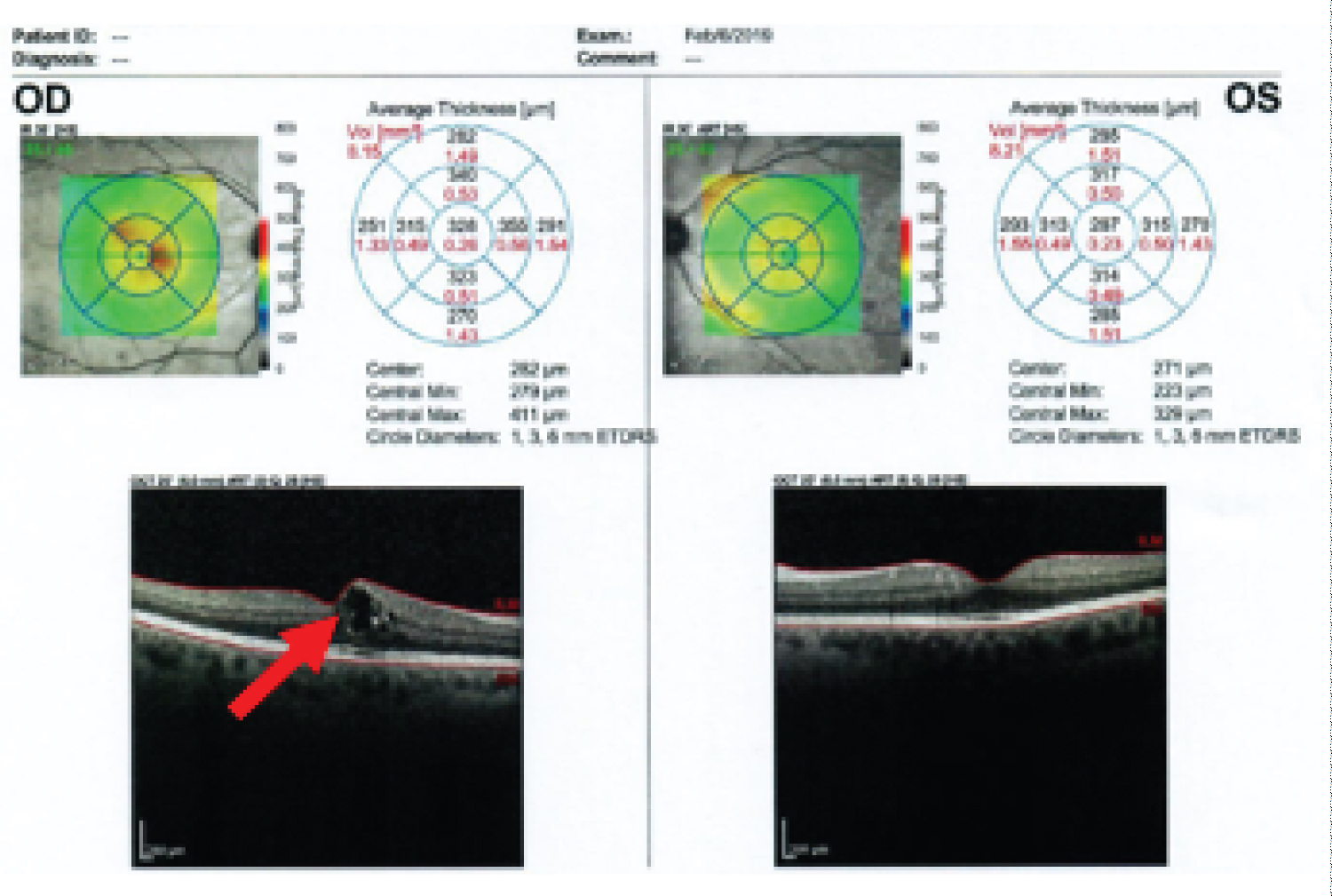 Figure 1: Diabetic retinopathy right eye-macular edema-red arrow (2/6/2019).
View Figure 1
Figure 1: Diabetic retinopathy right eye-macular edema-red arrow (2/6/2019).
View Figure 1
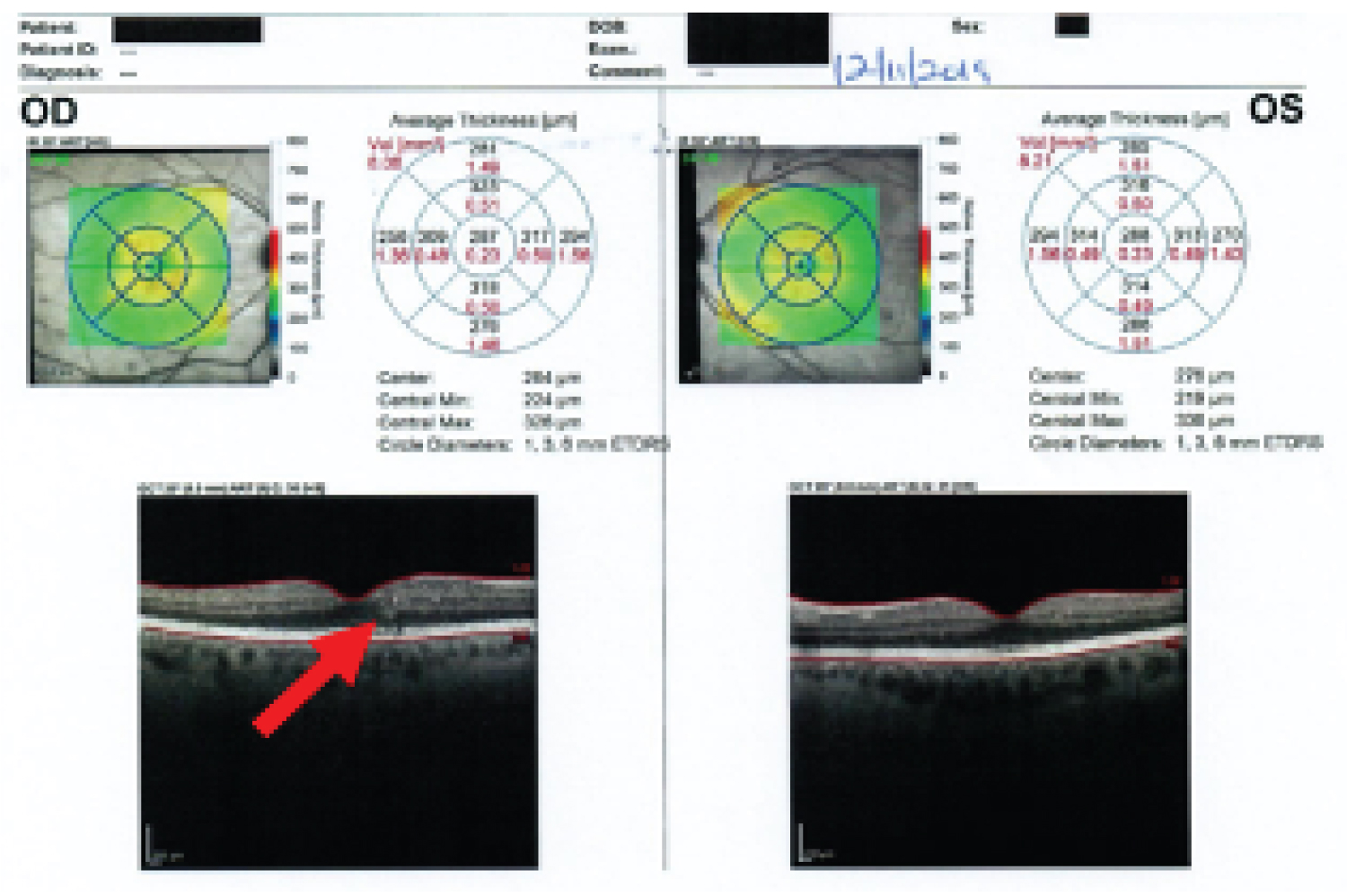 Figure 2: Diabetic retinopathy right eye-improved macular edema-red arrow (12/11/2019).
View Figure 2
Figure 2: Diabetic retinopathy right eye-improved macular edema-red arrow (12/11/2019).
View Figure 2
Table 1: Resolution of DME in the right eye. View Table 1
Glycogen storage has been found in various tissues, mostly in the liver and muscle and in the retina. Glucose is the only substrate that serves for the synthesis of glycogen. It has been found to be most concentrated in the Muller cells and fibers, but also found in the photoreceptors. In addition, glycogen is found more in the peripheral than central retinal tissue [13]. The tissue bearing the brunt of diabetic manifestations (lens, retina, kidney, blood vessels, and islet cells) are freely permeable to glucose and do not require insulin for glucose penetration as muscle and adipose tissue do, and hence are exposed to the ambient blood-glucose levels [14]. Mandarino and colleagues have demonstrated that insulin had no effect on glucose transport activity in the pericytes or the endothelial cells at the insulin concentrations of 0, 10, and 1,000 ng/ml respectively. It is also unlikely that GLUT isoforms other than GLUT1 is involved in glucose transport activity in pericytes and endothelial cells. In fact, pericytes had a markedly greater (about 5-fold) maximal rate of glucose transport than endothelial cells. They had speculated that pericytes likely contributed greater amount of glycoprotein and proteoglycans to the retinal capillary basement membrane than endothelial cells, and they might require more hexose [15].
DR is one of the most prevalent complications of diabetes, and while it is now well recognized that this disease involves perturbations of all the components of retinal tissue, its diagnosis still mostly relies on the detection of damages associated with vascular network. Based on those clinical features, diabetic retinopathy has been subdivided into two main stages: Non-proliferative diabetic retinopathy (NPDR), and proliferative diabetic retinopathy (PDR). In the primary stages of NPDR, microaneurysms or the swelling of small blood vessels, are the first detectable clinical signs of DR, and are associated with hypoperfusion of discrete regions of retina. While the clinical gradation is still made based on vascular pathology, recent studies have shown that ganglion cell sensitivity as well as dark adaptation is altered in patients with NPDR, thus confirming early alteration of neuronal function [16]. The progression of swelling can also be associated with leakage of fluid into the soft tissue of the eye causing inflammation, which can ultimately lead to swelling of the macula, the most sight-threatening stage of NPDR called DME. While not detectable on the fundus photographs, several studies have shown that DME was also characterized by dramatic loss of neuronal function and increased inflammation, confirming the progression of the non-vascular alterations at this stage of the disease [17]. In some cases, it is believed that progression of NPDR yields a severe ischemic state in specific areas of the retina, causing production of various factors leading to growth of new blood vessels in a process called neovascularization, making the transition to PDR [18,19]. These new vessels grow in a less controlled manner and disturb vision by developing in normally avascular regions, such as vitreous cavity, often leading to hemorrhage, or the macula leading to dramatic and persistent vision loss. Lastly scarring or gliosis associated with retinal neovascularization can cause traction between the vitreous and the retina, which can ultimately lead to detachment of the neural retina from the retinal pigment epithelium, a phenomenon called retinal detachment. When taking place in the macula, this detachment is a major cause of severe vision loss in DR patients [8].
Hyperglycemia plays a central role in the pathogenesis of diabetic retinopathy. Glucose travels across cell membrane on a on a transport protein in a way of insulin-dependent (GLUT 4) and insulin-independent (GLUT 1) manner. Glucose is the only nutrient that normally can be used by the brain, retina, and germinal epithelium of gonads [20]. In the retina, GLUT 1 is dispersed widely - in the nerve fiber layer, the ganglion cell layer, outer nuclear layer containing the photoreceptor bodies, the outer limiting membrane, and the retinal pigment epithelium (RPE]. In addition, GLUT 1 is expressed in the cell membrane of the Muller cells which are thought to serve as the principal nutritional supporting cells of the mammalian retina. Also, GLUT 1 transporter is expressed in the cells of blood-brain barrier (BBB) and brain-retinal barrier (BRB]. The BRB consists of two major components: The endothelial cells of the retinal capillaries (the inner BRB] and the cells of the retinal pigment epithelium, the barrier functions of which are conferred by tight junctions (zonule occludens). States of acute and chronic hyperglycemia give rise to pathologic processes in various structures of the eye [21,22]. This is described below.
The polyol pathway of glucose metabolism becomes active when intracellular glucose levels are elevated. Aldose reductase (AR) the first and rate-limiting enzyme in the pathway, reduces glucose to sorbitol using NADPH as a co-factor; sorbitol is metabolized to fructose by sorbitol dehydrogenase that uses NAD+. Sorbitol is hydrophilic and therefore does not diffuse readily through the cell membranes and accumulates intracellularly with possible osmotic consequences. The metabolites of fructose produced by the polyol pathway are powerful glycosylating agents that enter the in the formation of advanced glycation end-products (AGEs), which are inflammatory. The usage of NADPH by AR may result in less co-factor available for glutathione reductase, which is critical for the maintenance of the intracellular pool of reduced glutathione (GSH). This would lessen the capability of cells to respond to oxidative stress. Retinal ganglion cells, Muller glia, vascular pericytes, and the endothelial cells are endowed with AR. Hence these cell types are exposed to polyol pathway activation in diabetes. These are the cells that manifest the best-known changes or damage in diabetes. In primates, Muller cell density is over 5 times greater in the parafoveal than in peripheral regions of the retina. These characteristics make dysfunctional Muller cells relevant to the development of macular edema [14,23].
Mitochondrial dysfunction plays a significant role in accelerating the process of apoptosis in the retinal vascular cells. The retina is particularly susceptible to oxidative stress because of high energy demands and light exposure. In the development of DR, mitochondrial morphology and function are altered because of high metabolic demand, superoxide radicals are elevated, and oxygen consumption are impaired leading to progressive retinal damage [24]. Superoxide radicals, in addition to damaging protein and lipids, can also damage DNA by combining with guanine and forming oxidatively modified guanine base (8-OHdG) resulting in irreversible genotoxic alterations [25]. Diabetes is shown to damage mtDNA in the retina and its capillary cells. In addition to damage to mtDNA diabetes also compromises the DNA repair machinery in the retina further contributing to mitochondrial dysfunction [26].
Loss or dysfunction of the endothelial glycocalyx may also contribute to increased vascular permeability and inflammation. The glycocalyx is a network of membrane bound proteoglycans and glycoproteins in the luminal side of endothelium, which constitute the first permeability barrier for plasma proteins and adhering leucocytes. Under hyperglycemic conditions the glycocalyx is significantly reduced in thickness. This coincides with endothelial dysfunction and a general systemic increased vascular permeability in humans. This suggests that an intact glycocalyx is necessary for the normal function of the BRB [27].
Vasoregression is the primary response of the retinal micro vessels to chronic hyperglycemia [28]. The vasodegenerative phase of DR is characterized by inner blood-retinal barrier breakdown, capillary basement membrane thickening, pericyte dropout, microaneurysm formation and capillary closure. In advanced stages of non-proliferative DR severe endothelial damage occurs and the profiles of vasoactive factors synthesized by the endothelium become favorable to the vasoconstrictor agents. Therefore, there is a clear predominance of vasoconstriction, thus favoring hypoxic environment. As the disease progresses not only severe endothelial damage but also endothelial cell loss becomes a generalized phenomenon. Therefore, the capillaries are constituted only by tubes of thickened basement membrane, which are highly thrombogenic, and they could be occluded by classic thrombosis or even by leukocytes. This is crucial for the worsening of the ischemia and the consequence is proliferative DR. Sever hypoxia, acellular capillaries, and leukostasis (the inappropriate adherence of leukocytes to the retinal capillaries] are the three main conditions for initiation of PDR [29].
DR has been considered a microcirculatory disease of the retina. However, there is emerging evidence to suggest that retinal neurodegeneration is an early event in the pathogenesis of DR. Neural apoptosis and reactive gliosis, the hallmarks of retinal neurodegeneration, have already been observed in diabetic donors without microcirculatory abnormalities [9].
In recent years accumulating evidence suggests that inflammation plays a major role in the pathogenesis of DR [30]. Increased level of inflammatory mediators may lead to an early, persistent chronic inflammatory condition in the diabetic retina resulting leukocyte activation, adhesion to the vascular endothelium (leukostasis), and alteration of BRB [31]. Activated monocytes in the extravascular space differentiate into macrophages that secret cytokines and growth factors including VEGF, Ang-2, TNF-alpha, interleukins, and chemokines thus leading to increased vascular permeability and participating in the angiogenic process [32]. Among the proinflammatory cytokines/chemokines involved in the pathogenesis of DR the IL-1 beta, TNF-alpha, Monocyte Chemotactic protein-1 (MCP-1), stromal-cell derived factor-1 (SDF-1), and the adhesion molecules (ICAM-1, VCAM-1, sVAP-1) play a relevant role in the development of DR [33].
Microvascular complications are associated with endothelial and pericyte cell death, capillary degeneration, and obliteration. These lead to focal retinal ischemia. In response, vascular endothelial growth factor (VEGF) is liberated which is responsible for neoangiogenesis. In addition, cytokines are liberated which include tumor necrosis factor alfa (TNF-alfa), transforming growth factor beta (TGF beta), platelet derived growth factor (PDGF), which have been postulated to play important role in the development of diabetic retinopathy. However, VEGF is found to be a central player in the development of PDR and DME.
These new vessels grow in less controlled manner and disturb vision by spreading into relatively avascular areas, such as the vitreous cavity, often leading to hemorrhage, or the macula with dramatic loss of vision. Lastly scarring or gliosis associated with neovascularization can cause traction between the vitreous and the retina, which can ultimately lead to detachment of the neural retina from the retinal pigment epithelium, a phenomenon called tractional detachment. When this takes place in the macula, the detachment is a major cause of vision loss in diabetic retinopathy.
In 1953 Poulsen had published a case report on a patient with diabetic retinopathy who had experienced reversal of retinopathy after suffering from post-partum hemorrhage and loss of pituitary gland function (Simmonds' Disease). After reviewing several similar cases, he concluded that diabetic retinopathy is reversible and that retinopathy most likely is a consequence of metabolic disorder [34].
Before the early 1970s, pituitary ablation was the primary treatment for severe diabetic retinopathy, despite its limited efficacy and frequent, often sever sequalae, including death [7].
Before the use of anti-VEGF agents, intravitreal glucocorticoid therapy gained popularity among treating physicians. However, in 2008, the DRCR Retina Network showed that laser photocoagulation was superior to intravitreal triamcinolone injections for diabetic macular edema [35]. Glucocorticoids such as sustained release fluocinolone acetonide and dexamethasone implants also shown to reduce retinal thickening and to improve vision [36], and benefits for at least 3 years in patients with diabetic macular edema [37]. However, intravitreal treatment with glucocorticoids results in an increased risk of cataracts requiring surgery and was observed to induce increased intraocular pressure leading to glaucoma.
With the advent of panretinal laser photocoagulation in the 1970s, the risk of sever vision loss from PDR was reduced by more than 90%. Focal laser treatment of DME reduced the associated risk of moderate vision loss by 50% [7].
Elevated VEGF concentration in the posterior segment of the eye is found to be involved in the development of DR and DME. The permeability and angiogenic effects of VEGF led to the development of anti-VEGF agents. In 2010, the DRCR Retina Network showed that intravitreal injections of ranibizumab, an antibody to VEGF, with immediate or deferred laser treatment to the macula, if necessary were superior to the use of laser alone for the treatment of vision-impairing macular edema. With subsequent large and randomized studies, the efficacy of intravitreal injection of anti-VEGF agents over laser photocoagulation therapy has been confirmed. At present, this has replaced macular laser therapy as the initial standard treatment for eyes with visual acuity loss from DME [7].
Most eyes with DME respond to anti-VEGF therapy with some degree of anatomical improvement, visual improvement, or both. But in nearly 40% of eyes, complete resolution of macular edema is not achieved [7,38].
DR is a common complication of Type II diabetes mellitus and carries with it the threat of blindness. The UKPDS demonstrated that improvement in glycemic control improved microvascular disease and retinopathy outcomes. In the trial a difference of HbA1c between a median of 7.9% in the conventional therapy group and 7.0% in the intensive therapy group over the first ten years resulted in a risk reduction for progression retinopathy at 12 years by 21% assessed as a two-step change. Both baseline and updated mean data were concordant with escalated risk with higher HbA1c and the therapeutic trial demonstrated that intensive therapy improved outcomes [39].
Treatment of diabetic retinopathy starts with strict control of metabolic parameters of diabetes and blood pressure to help prevent vascular complications. The human retina has dual vascular supply-vascular supply to the photoreceptors via blood vessels from the choroid, whereas retinal neurons of the inner retina are supplied by branches of the retinal artery. The retina is a complex system of neural, glial, and vascular elements integrally tied together forming the neurovascular unit. The Muller cells, which envelope neural synapses and the blood vessels in the inner retina provide an important role in neurovascular coupling. In diabetics, activation of microglia via Angiotensin II and AT1-R (Angiotensin 1-Receptor), causes an increase of inflammatory cytokines/chemokines into the retina, which is pertinent to diseases such as diabetic retinopathy, known to involve increases in components of RAS [40]. Treatment with AT1 receptor blockers reduced the development of diabetic retinopathy in those free of retinal pathology. Tight blood pressure control in patients with hypertension and type 2 diabetes achieves a clinically important reduction in the risk of deaths related to diabetes, complications related to diabetes, progression of diabetic retinopathy, and deterioration of visual acuity [41].
Dyslipidemia, a major systemic disorder, is one of the most important risk factors for cardiovascular disease. LDLs modified by oxidation/glycation are implicated in the diabetic vascular complications. They cause cytotoxic effects in the retinal capillary pericytes and endothelial cells [42]. Patients with diabetes have an increased risk of suffering from dyslipidemia concurrently. In diabetic retinopathy with dyslipidemia, fenofibrate alone [43,44] and fenofibrate with simvastatin were found to retard the progression of retinopathy [45]. Lipid lowering drug atorvastatin as an adjunct in the management of DME have been found to retard the progression of retinopathy [46]. Intensive glycemic control and dyslipidemia has reduced the rate progression of DR [47].
As stated above intravitreal steroid injections are associated with cataract and increased intraocular pressure leading to glaucoma.
Laser-photocoagulation can cause (i) Vitreous hemorrhage, (ii) Exudative retinal detachment and choroidal detachment if too many laser shots are given in a single sitting, (ii) Reduction of contrast sensitivity, peripheral vision, and night vision after pan-retinal photocoagulation (PRP), (iv) Permanent scotomas in the visual field, (v) Worsening of preexisting macular edema or development of macular edema after laser/PRP, and (vi) Chances of worsening optic disc pallor.
Anti-VEGF therapy is associated with (i) Intraocular pressure spike, (ii) Cataract formation, (iii) Iatrogenic retina/posterior capsule tear, (iv) Vitreous hemorrhage, (v) Rhegmatogenous retinal detachment, and (vi) Worsening of traction over the macula if given in patients with tractional retinal detachment.
Walter Kempner had treated advanced cases of diabetic retinopathy with the rice diet. This diet was high in carbohydrate, low in protein, low in fat, low in sodium -containing 565 to 570 gm. of carbohydrate, 20 to 25 gm. of protein, less than 5 gm. of fat, and 70 to 120 mg. sodium per 2400 calories. There was improvement in diabetes, hypertension, and cholesterol levels. The effects of the rice diet on "specific" diabetic retinopathy (aneurysms; punctate, preretinal, vitreous hemorrhages; waxy exudates; retinitis proliferans) in 44 patients was as follows: progression of lesion in nine cases, improvement in one eye but progression lesion in the other eye in seven cases, no change in fifteen cases, and improvement in thirteen cases were seen. He concluded that the course of this disease can be favorably changed by intensive treatment with the rice diet [10].
Heritability has been estimated to be as high as 27% for DR and 52% for PDR. A large number of putative genes and genetic variants have been reported in the literature and some of them exhibit consistent association with DR (ALR2, VEGF and RAGE genes [48]. Diabetic environment disturbs metabolic homeostasis and alters various genes associated with oxidative stress, apoptosis, and inflammation. The major epigenetic mechanisms considered to regulate gene expression are DNA methylation, histone modifications, and noncoding RNA activity. Epigenetic modifications are influenced by environmental and dietary factors present in many natural foods. Polyphenols, including resveratrol, a natural compound found in the skin of red grapes and a constituent of red wine, are implicated in the regulation of histone deacetylase and Sirt1. Curcumin (diferuloylmethane), a natural compound commonly used in a curry spice, is shown to modulate a number of histone modifying enzymes and miRNAs. This has shown to ameliorate retinal metabolic abnormalities in the development of diabetic retinopathy. Thus, natural compounds can have potential benefits in inhibiting the development of retinopathy in diabetic patients via modulating both metabolic abnormalities and epigenetic modifications [49].
Diabetic retinopathy may be the most common microvascular complication of diabetes. It is responsible for about 10,000 new cases of blindness every year in the Unites States alone. Retinopathy may even begin to develop as early as 7 years before the diagnosis of diabetes in patients with type 2 diabetes [50]. The risk of developing diabetic retinopathy or other microvascular complications of diabetes depends on both the duration and severity of hyperglycemia. In the U.K. Prospective Diabetes Study (UKPDS) both hyperglycemia and hypertension were found to be related to the development of DR [41].
In DR, it is now well recognized that the disease involves perturbations of all the components of retinal tissue. But the diagnosis and treatment still center around the damages associated with the vascular outgrowth. The current treatments, though promising in arresting the progression of retinopathy, demand repeated treatments, are expensive, inconvenient for many patients, and are not without significant side effects, as described above. These make the role of prevention very appealing.
The role of Plant-Based diet, as illustrated in this case report, is promising. This diet consists of vegetables, fruits, legumes, whole grains, and small amount of seeds and nuts. Besides providing all the macro and micronutrients, they provide antioxidants, phytochemicals, and are rich in fiber. These are the nutrients those have been found to be essential in reversing diabetes, improving hypertension, and decreasing cholesterol levels which are the leading factors responsible for diabetic retinopathy [11,51]. In this patient, by making the lifestyle changes namely whole food plant-based diet, regular exercise complemented with adequate medications, she has achieved remission of diabetes by achieving HbA1c of 6.0% without anti-diabetic medications, except metformin. In addition, she has achieved lower blood pressure, decreased blood cholesterol level, and reduction in weight. This is accompanied with resolution of macular edema and there is no progression in retinopathy. Although, she had received laser and anti-VEGF treatments, but the magnitude of these treatments would not have made this degree of regression achieved without the contribution of intensive lifestyle changes.
A plant-based diet is promising in reversing and preventing the progression of diabetic retinopathy and diabetic macular edema by improving most of the common risk factors - diabetes, hypertension, and dyslipidemia. Moreover, the diet plan is simple, widely available, well tolerated, and is free of side effects. Their worldwide application will reduce the burden of diabetic retinopathy and its consequences, and not to mention the cost saving.