Chronic kidney disease (CKD) is associated with high incidence of morbidity, mortality, and is a costly burden to the healthcare system [1,2]. CKD is now the 8th leading cause of death in the United States and affects 14% of its people [3]. As CKD progresses, cardiovascular mortality rises, eventually becoming the most common cause of death among end-stage renal disease (ESRD) patients [4,5]. There is a growing interest in the dietary treatment of CKD, and a Plant-Based low-protein vegetarian diet has shown to be safe and effective in both treating and in slowing the progression of CKD [6].
A 72-year-old female has long standing history of coronary artery disease (CAD), hypertension, hyperlipidemia, and obesity. In 1987, at the age of 39 years, she had sustained an anterior wall myocardial infarction. She had cardiac catheterization and received a stent to the left anterior descending coronary artery (LAD), the infarct related artery. In 2007, she had sustained a second heart attack at the age of 59 years, and this time she was treated with simple balloon angioplasty to the LAD. In 2013, she presented with unstable angina pectoris and cardiac catheterization revealed critical two vessel CAD - 90% stenosis in the Right Coronary artery (RCA) and 90% stenosis in the LAD beyond the previous stent with normal left ventricular systolic function with ejection fraction (EF) of 60%. These were treated with a stent to RCA and another stent to LAD with excellent angiographic results, with resolution of symptoms.
At that time, her medications included Cozaar 50 mg po daily, Lopressor 50 mg po twice daily, Hydrochlorothiazide 25 mg po daily, Nitro-patch 2% topically daily, Zocor 40 mg po daily, Fenofibrate 134 mg po daily, Aspirin 81 mg po daily, Plavix 75 mg po daily, Neurontin 100 mg po daily.
On 3/13/2014, after discharge from hospital, she was seen in our Cardiovascular Wellness Clinic for continued care. She weighed 207 pounds (BMI 30.55 kg/m2), blood pressure was 140/80 mm of Hg, and heart rate was 64 beats per minute. The lipid profile revealed a total cholesterol of 254 mg/dl, triglyceride 123 mg/dl, HDL cholesterol 51 mg/dl, and LDL cholesterol were 179 mg/dl. Evaluation of renal function revealed blood urea nitrogen of 31 mg/dl and creatinine was 1.4 mg/dl, and eGFR of 44 ml/min/1.73 m2 of body surface area.
She was recommended to adopt a Plant-Based diet (PBD) to reverse heart disease and to prevent progression of renal disease. This diet consisted of vegetables, fruits, legumes, whole grains, and nuts and seeds. This diet provides about 10% of the calories coming from protein (plant), 10% coming from fat, and the remainder from complex carbohydrates; and there is no cholesterol [7]. At the same time, she was also counseled to avoid oils and animal-derived foods such as meat, fish, cheese, dairy, eggs, and processed foods.
But she followed the prescribed PBD only intermittently. She worked as an event planner which catered typical Western-style meals, and she helped herself with this diet some of the time. On 7/14/2014, her laboratory chemistries revealed a total cholesterol of 218 mg/dl, triglyceride 121 mg/dl, HDL cholesterol 47 mg/dl, and LDL cholesterol were 147 mg/dl. Her weight was 203 pounds (BMI 29.96 kg/m2), blood pressure was 140/80 mm of Hg, and heart rate was 64 beats per minute. The follow up blood works on 7/30/2015 were worse, the total cholesterol was 263 mg/dl, triglyceride 128 mg/dl, HDL cholesterol 43 mg/dl, and LDL cholesterol were 193 mg/dl. The BUN was 16 mg/dl, creatinine was 0.9 mg/dl and eGFR was > 60 ml/min/1.73 m2 of body surface area.
On 6/12/2017, she presented to the emergency room of the hospital in hypertensive crisis. Her blood pressure was 266/145 mm of Hg, heart rate 117/min, and respiratory rate of 18/min. She weighed 220 pounds (BMI 32.49 kg/m). Her total cholesterol was 296 mg/dl, triglyceride 203 mg/dl, HDL cholesterol was 39 mg/dl, and LDL cholesterol were 216 mg/dl. Her renal function revealed BUN of 26 mg/dl, creatinine of 1.6 mg/dl, and eGFR 47.5 ml/min/ 1.73 m2 of body surface area. She was admitted to the Intensive Care Unit (ICU) and her blood pressure was hard to control despite multiple medications, both oral and intravenous medications. Renal artery vascular study indicated bilateral critical (> 60% diameter reduction) renal artery stenosis. On 6/18/2017, the renal function has worsened reaching BUN of 45 mg/dl, creatinine 3.68 mg/dl, and eGFR of 14.4 ml/min/1.73 m2 of body surface area. Because of uncontrolled blood pressure and worsening renal function, she had renal artery angiogram with stent placement to the right renal artery, and intervention to the left renal artery failed because of critical stenosis not able to be crossed with a balloon. From 7/21/2017 trough 7/26/2017, renal function has deteriorated reaching a peak BUN of 38 to 43 mg/dl and creatinine reaching 5.7 mg/dl to 6.6 mg/dl, with the lowest eGFR of 9.1 ml/min/1.73 m2 of body surface area. A tunneled intraperitoneal catheter was inserted, and peritoneal dialysis was started on 7/26/2017.
On 2/22/2018, she returned to our Wellness Clinic for visit. On examination her weight was 189 pounds (85.7 kg) with BMI of 26.08 kg/m2, blood pressure 191/102 mm of Hg, heart rate 88 beats/minute. Her lipid profile revealed total cholesterol of 171 mg/dl, triglyceride 158 mg/dl, HDL 38 mg/dl and LDL 101 mg/dl. With dialysis her renal function has improved and the laboratory blood chemistries on 10/20/2018 showed a BUN of 26 mg/dl, creatinine of 1.6 mg/dl, and eGFR of 37.7 ml/min/1.73 m2 of body surface area. She was counseled to strictly follow a plant-based diet, and its importance on prevention of cardiovascular and renal disease was further emphasized.
She complied with the recommendation of following a PBD, and in about 18 months after initiation of peritoneal dialysis, her renal function had improved to the point she was able to go off dialysis by the end of December 2018. Her peritoneal dialysis catheter was removed on 1/3/2019.
During the office visit of 9/10/2020, she weighed 79.38 kg (175 pounds) with BMI of 25.4 kg/m2, her blood pressure was 121/77 mm of Hg, and heart rate was 77 beats/minute. Her lipid profile also has improved with total cholesterol of 161 mg/dl, triglyceride 90 mg/dl, HDL 52 mg/dl, and LDL 91 mg/dl. Her renal function has been stable with BUN of 28 mg/dl, creatinine of 1.5 mg/dl, and eGFR of 35.3 ml/min/1.73 m2 of body surface area. In the last clinical evaluation on 6/7/2021, she weighed 79.83 kg (176 pounds) with BMI of 26.09, her blood pressure was 155/58 mm of Hg, and heart rate 83 per minute. Her lipid profile on 5/18/2021 revealed total cholesterol of 155 mg/dl, triglyceride 134 mg/dl, HDL 45 mg/dl, and LDL 83 mg/dl. Her renal function has been stable with BUN 21 mg/dl, creatinine 1.6 mg/dl, and eGFR of 37.5 ml/1.73 m2 of body surface area (Figure 1).
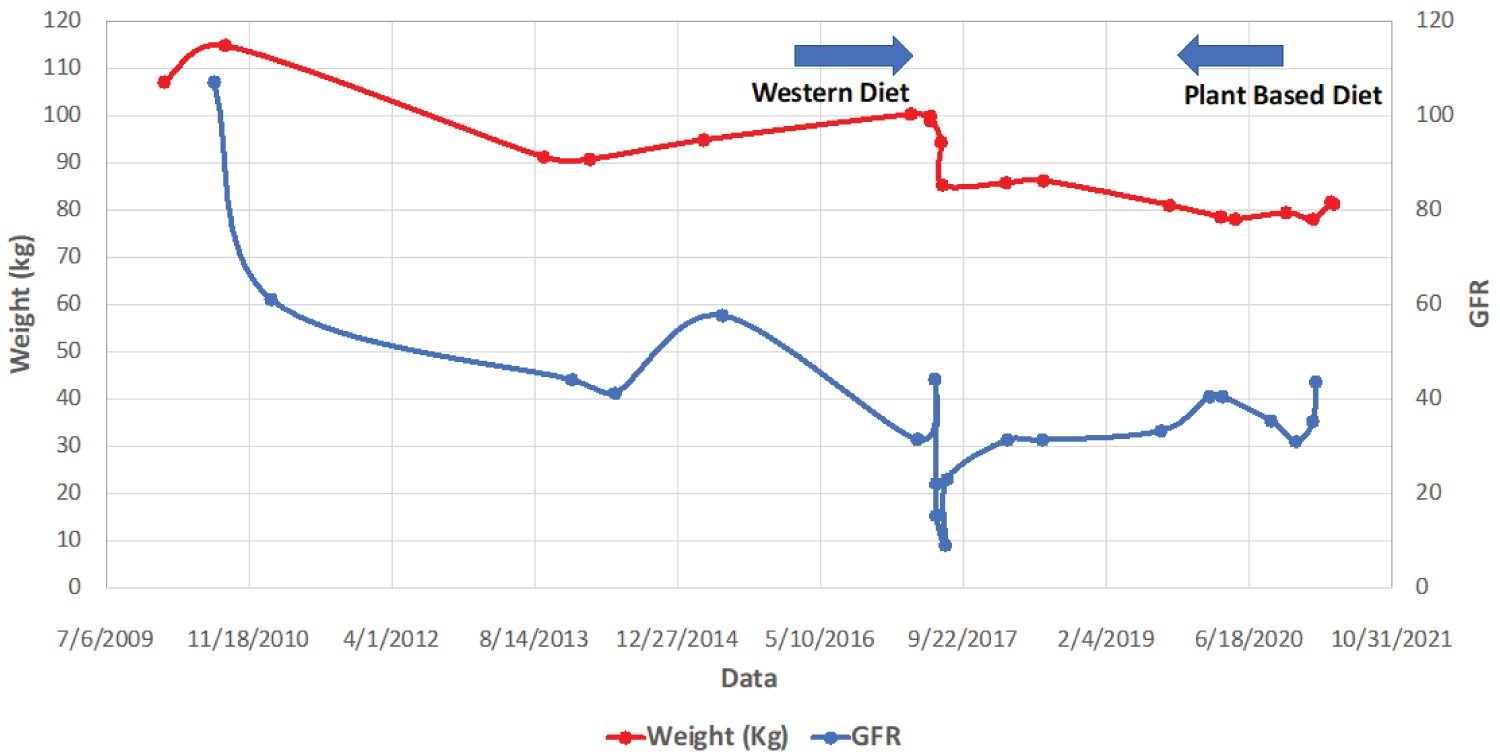 Figure 1: Relationship between weight and GFR with western diet vs. plant-based diet.
View Figure 1
Figure 1: Relationship between weight and GFR with western diet vs. plant-based diet.
View Figure 1
Her current medications are Coreg 25 mg po bid, Procardia XL 90 mg po daily in AM, Procardia XL 30 mg po daily in PM, ASA 81 mg po daily, Crestor 40 mg po daily, and sub-lingual nitroglycerine 0.4 mg for prn use.
Diet has a profound effect on kidney health. The Western-style diet consisting of refined cereals, high in animal protein, excess fat, dairy products, processed foods which are high in sugar, salt, and refined vegetable oils, have been shown to cause renal dysfunction [8].
Refined cereals and sugar lead to acute elevation of blood glucose concentration. This is accompanied with increased hormones secreted from the gut, and these in in combination with elevated blood glucose, stimulate insulin secretion from the pancreas causing an acute rise in blood insulin level. Chronic hyperinsulinemia contributes to the primary metabolic defect leading to obesity, hypertension, hyperlipidemia, diabetes, and atherosclerotic vascular disease, and are the risk factors those lead to chronic kidney disease (CKD) [8].
In this case, long standing history of obesity, hypertension, hyperlipidemia, coronary artery disease, and bilateral atherosclerotic renal artery stenosis, all directly and/or indirectly have contributed to chronic kidney disease (CKD). Presentation with hypertensive crisis precipitated acute kidney injury (AKI), on the top of existing CKD, and she urgently required peritoneal dialysis to deal with the complications related to acute renal failure such as hyperkalemia, acidosis, and volume overload.
Western-style diet is rich in saturated fat. The major sources of saturated fat in the US are from these common foods - meats, baked goods, cheese, milk, margarine, and butter. They lead to elevated blood levels of total and LDL cholesterol, the major risk factors for atherosclerotic vascular disease, including renal artery stenosis [8]. There is strong evidence that our current consumption of salt is the major factor increasing BP, CVD, and CKD. A reduction of salt from the current intake of 9-12 g/day to the recommended level of 5-6 g/day will have a major effect on BP, CVD, and CKD. In most developed countries approximately 80% of the salt comes from processed foods [9]. In the EPIC-Oxford study, the influence of diet of meat-eaters, fish-eaters, vegetarians, and vegans have been documented. The differences in macronutrient intakes accounted for about half the difference in mean BMI between vegans and meat-eaters. High protein and low fiber intakes were factors most strongly associated with increasing BMI [10].
Higher BMI has been found to be associated with an increased risk of kidney disease [11]. Obesity may also directly lead to CKD [12]. Pathologic studies have demonstrated that subjects with sever obesity develop proteinuria with podocyte hypertrophy, mesangial expansion, glomerular enlargement, and focal glomerular sclerosis, even in the absence of diabetes and hypertension [13]. In the obesity related glomerulopathy (ORG) many inflammatory factors have been implicated such as hypoadiponectinemia, hyperleptinemia, and hyperaldosteronism [14]. Obesity-induced adipose tissue expansion provides a plethora of intrinsic signals due to hypoxemia, mechanical stress, and adipocyte death, capable of initiating inflammatory response. Macrophages are the most abundant innate immune cells infiltrating and accumulating in the adipose tissue of obese individuals. Inflammatory markers such as CRP, TNF-alpha, and interleukin 6 (IL-6) are elevated leading to insulin resistance [15]. Obesity related glomerulopathy is an important complication of obesity and has a linear relationship [16]. Obesity is associated with glomerular hyperfiltration and increased urinary protein excretion, as well as structural and functional changes that lead to kidney disease and failure [17,18]. The gene expression profile from glomeruli derived directly from renal biopsy samples of patients with obesity-related glomerulopathy (ORG) impressively showed 256 genes were expressed, compared to the samples from donor kidneys. They were related to inflammatory cytokines such as TNF-alpha, its receptor Il-6 signal transducer, and interferon - gamma. The expression level of several key genes in lipid metabolism were increased over 2-fold, including low-density lipoprotein receptor, fatty acid binding protein-3, and sterol regulatory binding protein 1. As an indication of insulin resistance in the local glomerular cells, levels of glucose transporter 1 (GLU 1), leptin receptor, peroxisome proliferator activated receptor-gamma, and vascular endothelial growth factor are increased too [19].
The source of protein is equally important in its effect on GFR. In a study of healthy subjects, vegetable protein which constituted 100% of protein in vegans as compared to 64% vegetable protein in lactovegetarians, and 32% vegetable protein in omnivores, the mean GFR was significantly lower in the vegans (100 +/- 13 ml/min/1.73 m2) as compared to the omnivores (113 +/- 16 ml/min/m2, p less than 0.04), and in lactovegetarians it was intermediate (105 +/- 16 ml/min/1.73 m2) [20].
In another study of 6 volunteers with normal renal function, effect of protein-induced changes in renal function were evaluated. This revealed the rise in GFR and ERPF were lowest with soy protein and highest with beef (baseline GFR, 110+/-5; soy, 122+/-5; beef, 131+/-5 ml/min/1.73 m2; mean+/-SEM) [21].
In the Atherosclerosis Risk in Communities (ARIC) Study, red and processed meat consumption was associated with increased risk for CKD [22].
With accumulated experiences, high protein diet has been found to be associated with increased renal plasma flow (RPF), high GFR, and increased intraglomerular pressure, which leads to glomerular hyperfiltration [23,24]. These phenomena are found to be mediated by secretion of glucagon and prostaglandins [25]. Increased RPF, GFR, fractional clearance of albumin, and IgG were significantly higher with ingestion of animal than vegetable protein [26-29]. The recurrent episodes of these events lead to progressive glomerular damage causing glomerulosclerosis leading to chronic kidney disease (CKD) [30,31].
The abnormalities in plasma amino acid concentration representing a primary defect that precedes and perhaps facilitates the development of obesity is known since 1969 [32]. The Western diet rich in meat and dairy products contain high amount of branch-chain amino acids (BCAAs) - leucine, isoleucine, and valine. These have been found to over-stimulate the mammalian target of rapamycin complex 1(mTORC1) leading to obesity and insulin resistance. In contrast, the plant proteins have opposite metabolic effect [33,34]. In addition, BCAAs have been found to rapidly interfere with renal function, decreasing GFR and stimulating kidney fibrosis, thus increasing CKD progression [35].
The kidney plays an important role in the regulation of acid-base balance. Major determinants of net endogenous acid production are the generation of large amounts of hydrogen ions, mostly from animal-derived proteins. At an equal protein intake level, the net acid production is lower using plant foods [36]. With the progression of CKD its ability to excrete acid declines over time, leading to metabolic acidosis. On the other hand, a plant-based diet rich in fruits and vegetables are base-producing and has been found to counterbalance this acid-base abnormality. Vegetable proteins seem to induce renal changes comparable to those obtained by reducing the total amount of protein in the diet and prevent the vasodilating and proteinuric effects of meat. The nutritional treatment of chronic renal failure with a low-protein, low-phosphorus diet is found to be effective in reducing uremic intoxication, slowing the progression of renal failure and in preventing secondary parathyroidism [30].
Progressive renal failure results in higher urea concentration in the blood. This leads to increased secretion of urea to gastrointestinal (GI) tract. Exposure of intestinal bacteria to urea results in modifying the bacterial families containing urease which converts urea to ammonia. Gut derived uremic toxins, p-Cresyl sulfate, and indoxyl sulfate are derived from dietary protein have been found to cause renal tubular damage, coagulation abnormality and endothelial dysfunction [37].
Processed foods are high in salt [9], which is a major component of Western-style diet. Excess salt leads to hypertension, cardiovascular disease, and renal disease. The total salt intake in the typical US diet amounts to 9.6 g per day, which is about twice the normal recommended amount. Out of which 75% of the salt is in the processed foods, 15% are added during cooking and on the table, and 10% is present in the naturally occurring foods [8,9].
Advanced glycation end products (AGEs) from food have been shown to cause kidney disease. High AGE levels are found in processed meats, meat-substitutes, as well as in foods with high content of fat and protein - the products that are part of the Western-style diet [38]. In addition, cooking at high temperature, longer time, broiling, roasting, frying, and baking generates large quantity of AGEs. This shows that diet can be a significant environmental source of AGEs which may constitute a chronic risk factor for kidney disease and cardiovascular disease [39-42]. In contrast, carbohydrate-rich foods such as vegetables, fruits, and whole grains contain relatively few AGEs, even after cooking [43].
AGEs are proinflammatory and pro-oxidative compounds that play a role in the high prevalence of endothelial dysfunction and subsequent CKD. AGEs increase the level of ROS (Reactive Oxygen Species) through activation of NADPH oxidase and mitochondrial pathways in both receptor dependent (through RAGE) and a receptor-independent manner. AGES are shown to increase oxidative LDL. Glycosylated LDL are more susceptible to oxidation and are less effectively cleared from the circulation. AGEs have been shown to amplify inflammatory response in patients with CKD through RAGE, because AGE-RAGE interaction activates redox-sensitive transcription factor NF-kB, which leads to gene expression and release of pro-inflammatory molecules, such as IL-Ia, IL-6, and TNF-alpha [30].
The endothelium plays an important role in the pathophysiology of kidney injury. The endothelial layer is covered with a protective layer of gel called endothelial glycocalyx (eGCX), which comprises membrane-bound negatively charged proteoglycans, glycoprotein, glycolipids, and glycosaminoglycans [44]. It creates a negatively charged molecular sieve that prevents large molecules from passing through. It is the first line of defense to prevent endothelial injury. This delicate protective layer is easily damaged by hypertension, hyperglycemia, hyperlipidemia, oxidized lipoproteins, inflammatory molecules, and oxidative stress [45,46].
In addition, asymmetric dimethylarginine (ADMA) derived from methylation of arginine residues in proteins is partly (about 20%) excreted by the kidneys. With renal insufficiency excretion of ADMA declines and this is associated with increased blood levels of ADMA leading to further progression of CKD [47,48]. Also, ADMA inhibit endogenous nitric oxide synthesis by inhibiting nitric oxide synthase, playing a role in endothelial dysfunction [42,49].
Injury to endothelial cells results in enhanced leukocyte adherence, platelet aggregation, vasoconstriction, reduced blood flow to the nephron, and impairment of the permeability barrier of the glomerulus. Loss of peritubular capillaries is a recognized consequence of AKI which leads to chronic kidney.
There is increasing evidence that weight loss not only helps to reduce glomerular hyperfiltration and proteinuria, but also attenuates metabolic disorders associated with obesity, such as hypertension, altered lipid abnormalities, insulin resistance, and inflammation [14]. Morbid obese patients with ORG those underwent bariatric surgery and drastic weight loss had reversed glomerulopathy. A low-calorie diet induced AMK pathway activation, and reduced Fetuin-A level, led to increased level of adiponectin and prevented podocyte injury [13]. The effects of weight loss on renal function in patients with severe obesity have been studied by Chagnac and group. These obese patients, who had undergone gastroplasty and have achieved weight loss, improved their glomerular hemodynamic abnormalities, suggesting weight loss may delay the progression of renal insufficiency in obese patients with glomerular disease [50]. In obese individuals, calorie restriction and weight loss, along with amelioration of insulin resistance and other functional and metabolic abnormalities, achieved significant short-term reduction in the GFR that conceivably reflected amelioration of glomerular hyperfiltration and that resembled the reduction observed after an invasive procedure such as bariatric surgery [51].
In experimental animals (rats), a low-protein diet has been shown to have prevented uremia and death [52]. In patients with moderate renal insufficiency, a low-protein diet demonstrated slow decline in renal function after four months of initiation [53].
A low protein vegetarian diet (LPVD) reduces nitrogen waste products and decreases kidney workload by lowering RPF, GFR, intraglomerular hydraulic pressure, which may protect the kidney, particularly in patients with decreased kidney function [6,30]. It leads to favorable metabolic effects that can preserve kidney function and control uremic symptoms.
For a 70 kg individual 0.6 g/kg/per day comes to 42 g/day, which is considered a low-protein-diet (LPD). In addition, protein restriction has been demonstrated to reduce proteinuria by 20-50% in patients with CKD. It improves metabolic acidosis and offers better control of mineral bone disorder (MBD) [30]. On the other hand, animal-derived protein is high in phosphorus while plant protein is low in phosphorus. As a result, plant-protein is efficacious in lowering serum phosphorus, which results in reduction in serum parathyroid hormone (PTH), and fibroblast growth factor-23 (FGF-23). CKD-MBD may include slowing the progression of vascular calcification and improving cardiovascular outcomes. Also, LPD may attenuate insulin resistance and oxidative stress, the common features that may play a role in accelerating atherosclerosis in patients with CKD [29-31].
In many parts of the world a low protein diet (LPD, 0.6-0.8 g/kg/day) is routinely prescribed for the management of patients with non-dialysis-dependent CKD, however this practice is infrequent in North America [31].
Prevention and decreased progression of kidney disease can be achieved with a PBD. This can be achieved with reducing AGES from the dietary source. Dietary AGEs exist in naturally occurring in foods, but they are high in animal-derived foods. Secondly, food preparations by heating at high temperature, grilling, broiling, roasting, shearing, and frying leads to very high levels of AGEs. On the other hand, plant-foods such as whole grains, vegetables, legumes, fruits, and nuts; and using the cooking methods that preserve moisture - stewing, steam-cooking, boiling, or poaching generate fewer AGEs. This method of food preparation reduces AGEs by 50% or more. These in combination will prevent and reduce the progression of kidney disease [39-43].
Resistant starches obtained from vegetables, fruits, wheat, corn, and nuts are metabolized by intestinal microbiota and synthesize short chain fatty acids (SCFA). SCFA provide nutrients to the enterocytes and are associated with the integrity of the gut epithelial barrier preventing absorption of toxic byproducts to the systemic circulation, thereby preventing chronic inflammation [37]. In the CKD population, higher fiber intake not only had strong association with lower inflammation but also was associated with lower mortality [54,55].
Now there is a growing body of evidence indicates that a plant-based, fiber-rich diet has multiple beneficial effects in the prevention and management of CKD. It improves metabolic acidosis, hyperphosphatemia, hypertension, uremic toxins, need for kidney replacement therapy including dialysis, patient satisfaction, quality of life, and mortality [56].
A balanced low-protein plant-based diet, by virtue of its reno-protective biochemical composition and function, has the advantage in modulating metabolic effects that can prevent the progression of chronic kidney disease.