Extravasation of infusion fluids is a frequent complication in the neonatal patient population. The authors report the case of a newborn infant who had a necrotic soft tissue lesion due to extravasation in the lower arm, and discuss the therapeutic options from a surgical point of view.
Extravasation, Complication, Neonate, Necrosis, Scar
FTSG: Full-Thickness Skin Graft; STSG: Split-Thickness Skin Graft
Extravasation is the accidental leakage of infusion fluids in the subcutaneous tissues. Intravenous therapy can be required for a long period of time in premature infants and the intravenous access is hard to achieve, making extravasation unfortunately a common complication in the neonatal unit [1,2]. Vigilance is necessary to recognize extravasation lesions in a timely manner, because the potential subsequent soft tissue injuries in premature infants with thin and fragile skin can lead to severe necrosis and scarring. The reason for skin necrosis can be tissue pressure in case of neutral fluids leak out of the vein, but necrosis can also follow a cytotoxic mechanism due to potassium, calcium or other substances.
A newborn infant developed an Enterobacter sepsis and disseminated intravascular coagulation two days after a premature birth at 35.5 weeks and after a pregnancy burdened with maternal substance abuse. On day 2 after birth, a saline extravasation occurred (NaCl 0.9%) in the left arm. Thirteen days after the extravasation, our plastic surgery team was called to evaluate the lesion (Figure 1). A full-thickness defect on the dorsal side of the lower two-thirds of the lower arm was observed, which also involved the dorsal wrist. The extensor tendons seemed intact as the neonate was able to stretch wrist and fingers. A wound swab was negative and the neonate was stable.
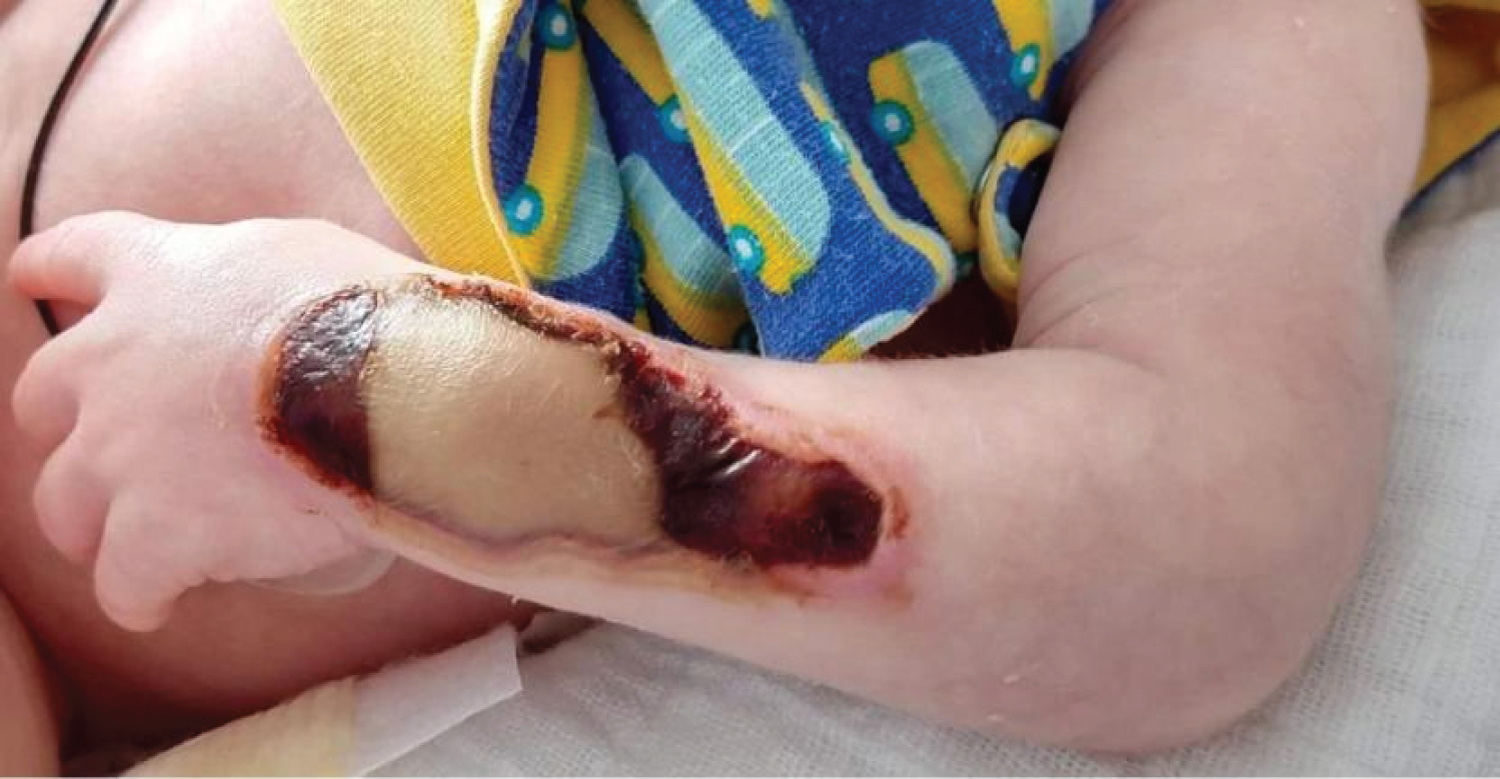 Figure 1: On day thirteen after the extravasation, a full-thickness skin defect was observed on the left lower arm, involving the distal dorsal two-thirds and the dorsal wrist. An adherent, necrotic, dry crust was present without underlying liquid collection. No infectious signs were observed. The newborn infant was able to actively stretch the fingers and wrist. View Figure 1
Figure 1: On day thirteen after the extravasation, a full-thickness skin defect was observed on the left lower arm, involving the distal dorsal two-thirds and the dorsal wrist. An adherent, necrotic, dry crust was present without underlying liquid collection. No infectious signs were observed. The newborn infant was able to actively stretch the fingers and wrist. View Figure 1
Four possible therapeutic options, each with their advantages and disadvantages, are discussed. Since neonates have a high spontaneous regeneration capacity, secondary healing was an option. It would exclude the necessity of undergoing anesthesia, which is not without risks in this age category. However, conservative management would require a long healing time and hospital stay. The necrotic lesion can infect and the wound is a potential entry point for development of sepsis. Desiccation of the underlying tendons and bone is possible, as well as general dehydration. Importantly, delayed healing can lead to unsightly scarring with hypertrophy, and skin contracture resulting in a limited range of motion.
In order to prevent the complications resulting from secondary healing, one can choose for early excision or avulsion followed by reconstruction with a full-thickness skin graft (FTSG). It would lead to a faster healing with a lower risk for desiccation and infection. Scarring and contracture would occur to a lesser extent. Anesthesia would be required. Donor site morbidity could involve both groins in order to harvest sufficient skin to cover the total defect.
To avoid additional scarring due to FTSG harvesting, a two-step surgical approach can be performed in which, firstly, the non-viable tissue is excised and a dermal matrix (Integra®) is applied, and, secondly, the dermal regeneration template is covered with a sheet thin split-thickness skin graft (STSG) [3,4]. This lengthens the hospital stay since at least a two-week delay is expected between both surgical interventions. Other disadvantages are the need to perform two operations under anesthesia, the high price of the dermal matrix product and the risk of infection of the product. The arm would have to be splinted in order to immobilize the product to allow optimal integration on the wound bed, ingrowth of host-derived cells and vascularization. An additional technical difficulty is the harvesting of a STSG in a neonate, which could lead to scarring or alopecia [5]. Arguments in favor of using this acellular dermal matrix are its superiority in outcome regarding skin pliability, scar thickness, range of motion and contracture, compared to a conventional STSG.
An elegant treatment modality would be the bromelain-based enzymatic debridement (Nexobrid®), which has the ability to selectively debride necrotic tissues, while sparing viable tissue [6]. Therefore, it causes less collateral damage and it is indicated in mid to deep burns with mixed patterns. It causes less blood loss and decreases the surface area that would ultimately require skin grafting. Usually, the enzymatic debridement is followed by the application of (a dermal regeneration template and) a STSG two days later. To date, the use of Nexobrid® is, however, off-label in the pediatric population, even though children are a good indication. On presentation, the eschar was thirteen-days-old, which increases the likeliness of being contaminated. Debridement of this eschar with proteolytic enzymes can lead to bacteremia and a second septic episode. The procedure is painful and would also require anesthesia.
After careful consideration, the decision was taken to pursue a conservative treatment, consisting of daily disinfection with chlorhexidine digluconate, and antiseptic dressings creating a moist wound environment, followed by a non-compressive bandage, and slight elevation of the arm. Contracture of the wrist was avoided with daily passive mobilization. A progressive re-epithelialization from the wound edges was observed (Figure 2). The necrotic eschar could be easily removed with minimal blood loss and discomfort, bed-side on day 40. No tendons were exposed. An enzyme alginogel (Flaminal® Hydro) was applied until complete re-epithelialization on day 51 (Figure 3). Afterwards, physiotherapy, sun protection and massaging with hydrating cream are performed, and custom-made silicon sheets and compressive garment are applied for optimal scar appearance, thickness and pliability.
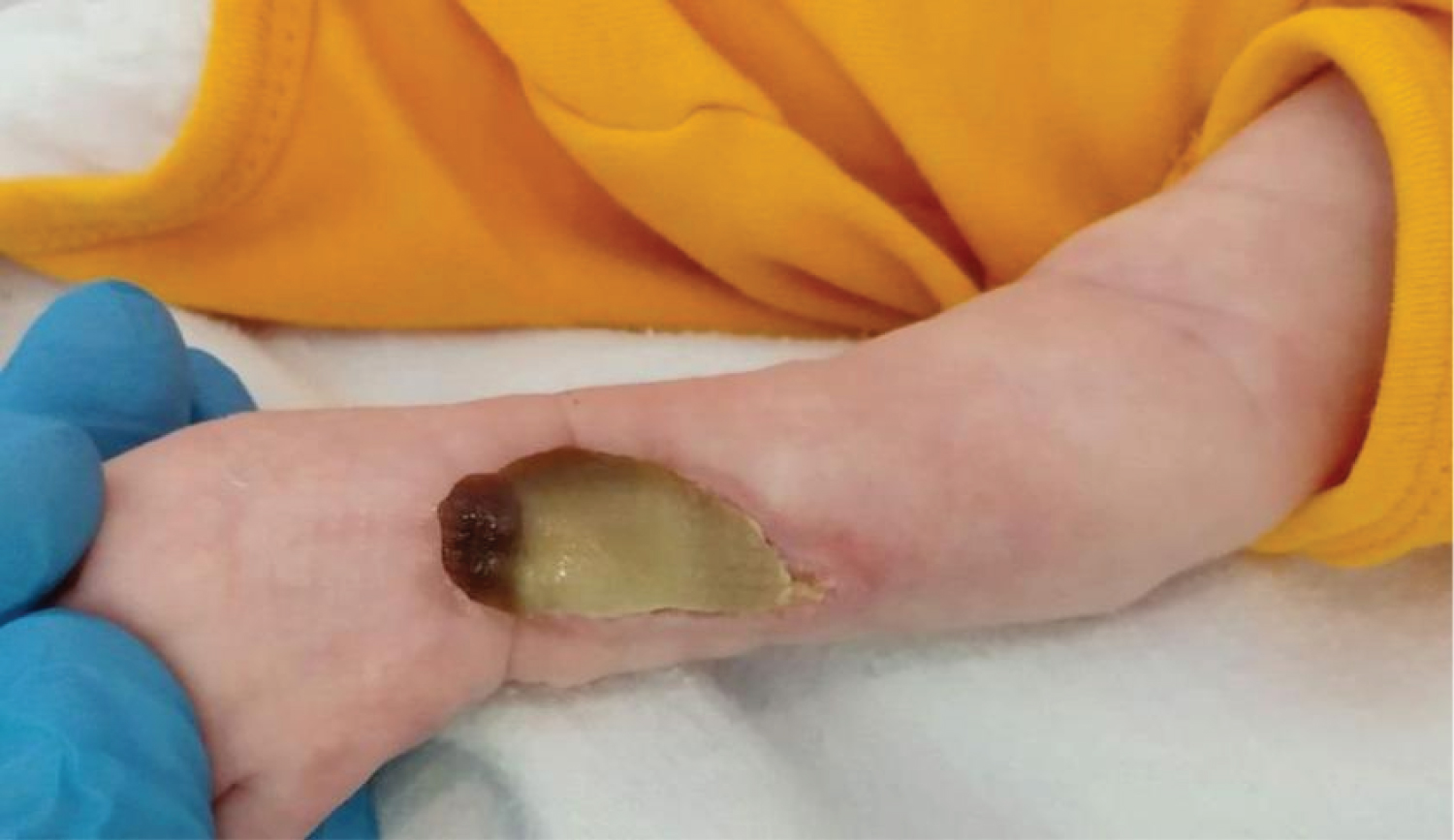 Figure 2: On day 35 after the extravasation, a remarkable decrease of the lesion was noticeable, and the necrotic crust detached almost completely from the underlying wound. View Figure 2
Figure 2: On day 35 after the extravasation, a remarkable decrease of the lesion was noticeable, and the necrotic crust detached almost completely from the underlying wound. View Figure 2
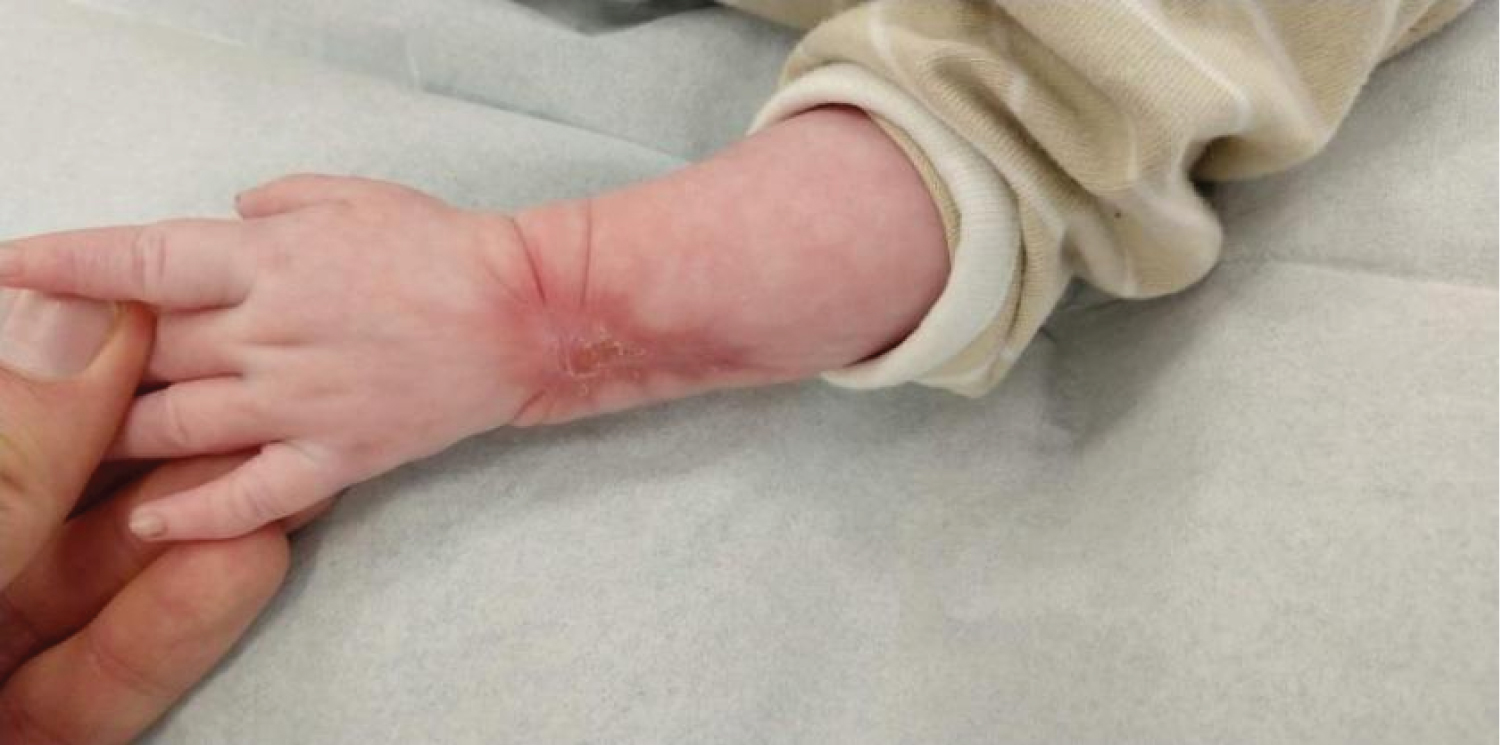 Figure 3: Complete re-epithelialization was achieved 7.5 weeks after the initial trauma. A contracture of the scar with limited palmar flexion of the wrist is treated through mobilization, massaging, silicon patches and compression. View Figure 3
Figure 3: Complete re-epithelialization was achieved 7.5 weeks after the initial trauma. A contracture of the scar with limited palmar flexion of the wrist is treated through mobilization, massaging, silicon patches and compression. View Figure 3
No therapeutic modality with clear benefits over other treatment options is currently preferred in the premature neonatal patient with extravasation-induced full thickness necrosis. In the presented case, closure of the full thickness defect was achieved after 7.5 weeks of conservative management without complications. This case demonstrates the exceptional healing capacity of the neonate. Surgical stress and anesthesia can worsen the general condition of the premature newborn. "Good surgeons know how to operate, better ones when to operate, and the best when not to operate" [7]: We should stay humble as every surgical option described in our report carries potential risks. It is our task to correctly inform the parents and colleagues, who are not surgeons, as they might pressure us towards a more pro-active surgical treatment.
However, a scar is unavoidable in the presented case, and hypertrophy and contracture with forced dorsal extension of the wrist can still occur [8]. In that case, serial excision, correction through scar plasty, reconstruction with tissue expansion, STSG with dermal matrix, FTSG or a thin microvascular free flap at a later stage can be offered, when the anesthesia procedure would be less cumbersome, and with consent of the adolescent patient.
Extravasation is a common complication in the neonatal unit. Therapeutic options are discussed with their advantages and disadvantages. A case is demonstrated, in which a conservative approach led to healing in 7.5 weeks.
The authors certify that they have obtained the written consent from the presented underage patient's parents to describe this case report and use the patient's photos for publication.
None.
The authors declare that they have no conflict of interest.