During the last decade the international debate on adverse health effects of silicone breast implants has intensified and recently the related disorder has been termed Breast Implant Illness (BII). This study aimed to explore the effects of explantation of silicone implants.
In a retrospective study, women with silicone breast implants, who consulted plastic surgeons for a variety of reasons, were requested to fill out a questionnaire, which was aimed at an evaluation of health complaints. In total, 101 questionnaires of the women with silicone breast implants and 34 questionnaires filled out by female hospital workers without breast implants, who served as a reference group, were evaluated. The women with silicone breast implants comprised 39 women who retained their silicone breast implants and 62 women who had explantation surgery.
The women with silicone breast implants, who had these implants for an average period of 15 years, reported various health complaints typical of BII, whereas women without such implants did not or hardly experience these complaints. Women who underwent explantation of the implants and a capsulectomy reported a significant improvement of various BII-related health complaints.
The results of this study demonstrate that explantation of silicone breast implants in women who experience BII-related complaints results in a resolution or improvement of their complaints and symptoms. Women should be informed about potential health effects resulting from silicone breast implants prior to implantation surgery.
Breast implants, Capsulectomy, Explantation, Gel bleed, Silicone
ASIA: Autoimmune/inflammatory Syndrome Induced by Adjuvants; BII: Breast Implant Illness; yrs: Years
In 1978 the first study on gel bleed from silicone gel-filled implants was published by Barker and co-workers [1]. They demonstrated the spontaneous leakage of material from silicone-gel-filled implants from different manufacturers on filter paper, without pressure being applied to them. The distribution by molecular weights of the leaked silicones reflected the silicone content of the implant. Brody [2] coined the term "bleeding" to describe this phenomenon of silicone diffusion from implants. Barker and co-workers also demonstrated in the same study the presence of silicones in the capsules of patients who underwent an open capsulotomy. In subsequent decennia many other studies demonstrated that silicone molecules can be detected in the body of individuals with breast implants [3-5], although evidence that these silicones were originating from their silicone breast implants was lacking. They could have come from different sources like nutrition, medication or cosmetics. Only when silicone deposition was found in the periprosthetic capsule [6], this could be directly linked to the silicone breast implant. Beekman and co-workers described the stages of migration through the capsule to the point of stage 4 migration, where the silicone material has moved through the capsule and appears on the outside of the capsule. The most recent study on this subject was published in 2016 [7]. For the first time silicone molecules were identified by three distinct methods, one of them being EDX-analysis, in every organ and tissue analysed, including the brain and spinal cord, of a woman who had silicone breast implants for 17 years. The silicon levels in the tissues were so high that there was no other explanation for their origin than bleeding from the implants. For instance, the number of Si-counts in the spinal cord was more than 20,000 per 0.1 μm2. Women without silicone breast implants never show such high quantities of Si-counts in their tissues, with Si-counts not higher than 500 per 0.1 μm2. The conclusion was that silicones escape through the intact shell as gel bleed into the surroundings, and it is likely that this process becomes intensified in case of a ruptured shell of the implant. Next, the molecules are either transported by the blood and lymphatic stream, as could be seen in the former study, or migrate through the tissue planes to become disseminated throughout the body.
The widespread dispersion of silicones and silicon-containing breakdown products throughout the body may cause a myriad of disruptions to the body's biochemical and enzymatic processes, which likely cause health complaints. Many publications support the assertion that this is associated with a systemic illness, which may gradually develop in women with silicone breast implants, which may be influenced by genetic and environmental factors. If the specific set of health complaints in patients with silicone breast implants are indeed resulting from these implants, it stands to reason that explantation of the implants in the affected women, will have an effect on these complaints. It was with this in mind that this study was undertaken.
Apart from the effects of explantation surgery on the health complaints other potentially relevant data were collected. In many cases it was impossible to find out which brand and size of implant had been inserted, mainly because these implants were inserted years ago by other surgeons and relevant information had been lost, while the deteriorated implant itself could not provide this information. In order to get an impression of the type of the silicone implants, the "generation of the implant" was used [8]. First generation implants were made between 1963 and 1972. According to Peters these were composed of a thick (firm) gel and a thick elastomeric wall. Second generation implants were made from 1972 to 1979 and they had a high degree of softness. These implants had a smooth elastomeric surface. The third generation implants, which had a stronger shell and an inner surface coated with a "barrier layer" to retard the gel bleed, became available in 1979. The fourth generation implants were introduced around 1992. They had a textured shell and the gel was more cohesive to resemble the anatomical form of the natural breast. The fifth generation soft touch implants were introduced in 2002. In the Peters' study it was demonstrated that after a mean implantation time of 15 years, the majority of the silicone breast implants had ruptured.
Here, we have analysed the effects of silicone breast implant explantation on the state of health of women who carried such implants for a mean period of 15 years, based on questionnaires filled out by the participants. As reference groups, women who kept silicone breast implants and women who never received breast implants were included.
When women who had silicone breast implants and who consulted the plastic surgery department of the Radboud University Medical Centre or a private clinic for a variety of reasons were given the opportunity to express their thoughts about their own health, they independently kept mentioning a certain set of complaints. For instance, extreme fatigue was almost always mentioned. From these complaints gradually a list emerged and became the basis of a questionnaire that was used to investigate the effects of explantation.
The questionnaire started with some general questions, which were followed by assessments of the severity of the complaints, graded from 1 (low) to 10 (high).
1. What was the reason for the breast operation?
2. In case implants were removed, was that a silicone breast implant?
3. On a scale of 1 to 10, please mark the following symptoms if they existed pre-operatively:
a. fatigue
b. painful left breast
c. painful right breast
d. hardening of the left breast
e. hardening of the right breast
f. painful muscles
g. painful joints
h. sleep disturbances
i. memory losses, difficulty with concentration, brain fogginess
j. sense of being unhealthy
k. burning sensation and/or redness of the eyes
l. itching
4. Same question as 3, but then assess "a." to "l." at least 6 months after your last operation.
In total, 152 of the 199 women who entered the study responded. Nine questionnaires could not be used for several reasons (incomplete anonymous questionnaires, Povidone and Trilucent implants, which were not part of this study, pregnancy, severe mastopathy and transfusion reaction). Of the 143 remaining questionnaires, 42 were also eliminated, because they had their first augmentation with a non-silicone-gel-filled breast implant. As a consequence, 101 of the questionnaires could be evaluated. In the control group 34 filled out questionnaires were received from a total of 60 female hospital workers (Group 1) who never had silicone breast implants in their history. Their mean age was 35 years. The women with silicone breast implants were divided in two groups. Group 2 consisted of 39 women with a mean age of 48 years, who had silicone breast implants for a mean period of 15 years at the time of filling out the questionnaire and continued with these implants during replacements that were executed in this 15-year period. There were 62 women in Group 3, with a mean age of 50 years at the time of filling out the questionnaire. They also had their implants for a mean period of 15 years and the mean follow-up period after explantation was 3.5 years.
The women with silicone implants came to the hospital for various reasons, such as breast pain, asymmetry, capsular contracture, advanced age of implant, being unhappy with the aesthetic appearance or uneasiness with their implants. Depending on the case, advice was given either to replace or remove them. Related complaints, such as capsular contracture, were also treated surgically.
Based on the questionnaires filled out by all participants the presence and severity of health complaints were assessed. The complaints that were specified in the questionnaire were not or only very infrequently reported by the reference group, Group 1 (Figure 1). The most frequently reported complaints were fatigue (8.8%), burning sensation of the eyes (8.8%), itching and painful joints (5.9%). In contrast, the women of both Groups 2 and 3 reported a number of health complaints, which arose during the 15-year period they had silicone breast implants. This is consistent with previous reports and substantiates that after implantation of silicone breast implants, regardless of the brand, in due course specific health complaints may develop that together define breast implant illness (BII). The frequency by which the complaints were reported by Group 2 was somewhat lower than that of Group 3 (Figure 1), in spite of the fact that they had silicone implants for a similar period. This suggests that the frequency, and possibly the severity of the complaints may affect the decision to remove the silicone implants or to replace them by non-silicone gel-filled implants.
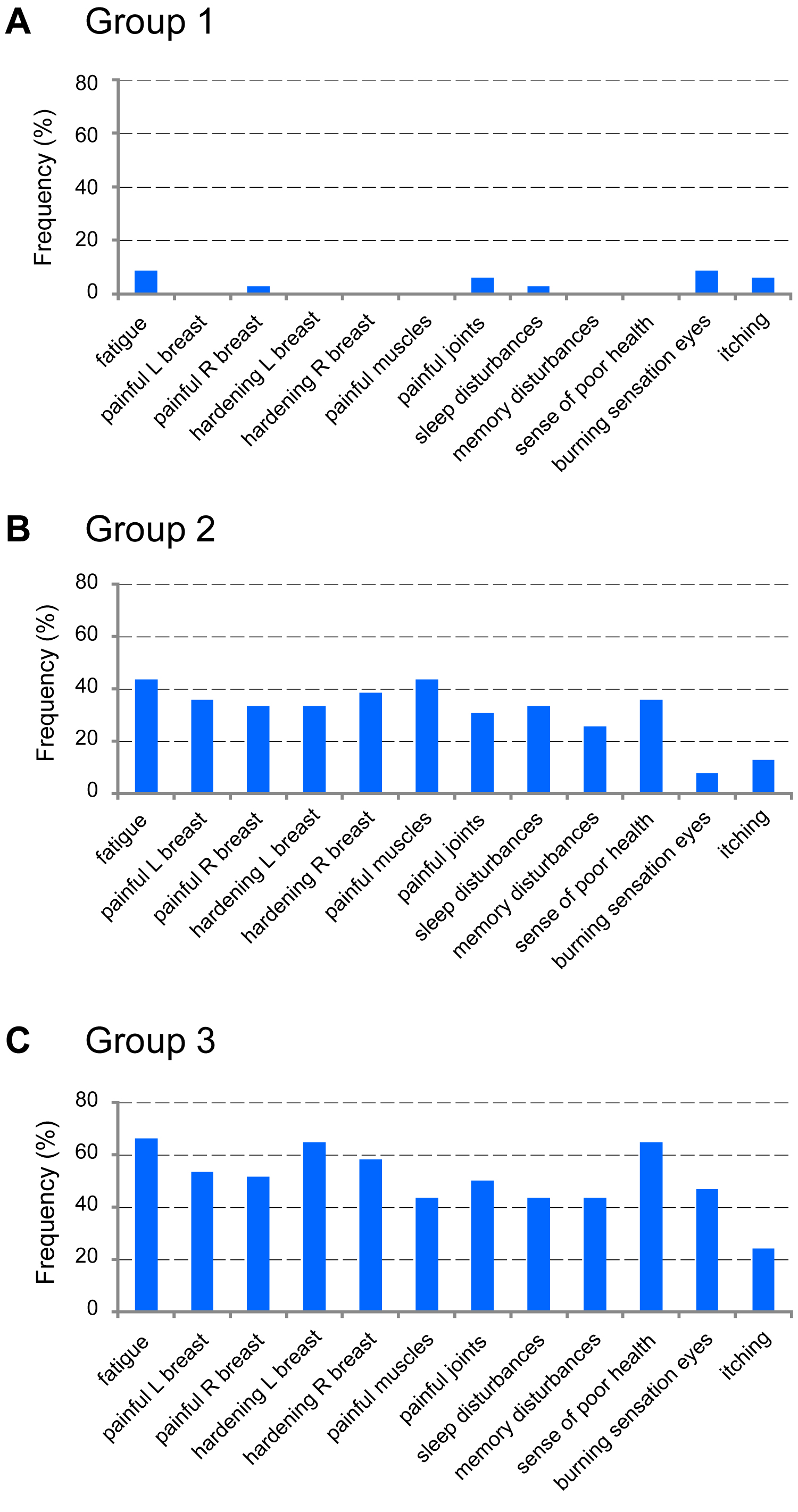 Figure 1: Health complaints reported by Groups 1, 2 and 3. The bars correspond to the percentage of subjects in each group reporting complaints (specified below the graphs) with scores of 4 and higher. A) Group 1 (Control group); B) Group 2; C) Group 3. View Figure 1
Figure 1: Health complaints reported by Groups 1, 2 and 3. The bars correspond to the percentage of subjects in each group reporting complaints (specified below the graphs) with scores of 4 and higher. A) Group 1 (Control group); B) Group 2; C) Group 3. View Figure 1
Upon explantation of the silicone gel-filled implants the women in Group 3 reported an improvement of their complaints after a mean follow-up of 3.5 years (Figure 2). Major improvements were experienced with respect to pain in and hardening of the breasts, but also the complaints affecting other parts of the body all improved. In both Group 2 and Group 3 some of the women had one or more replacements with silicone breast implants during the period preceding the current evaluation or explantation surgery (mean 15 years for both groups) and these replacements were generally done with later generation silicone implants.
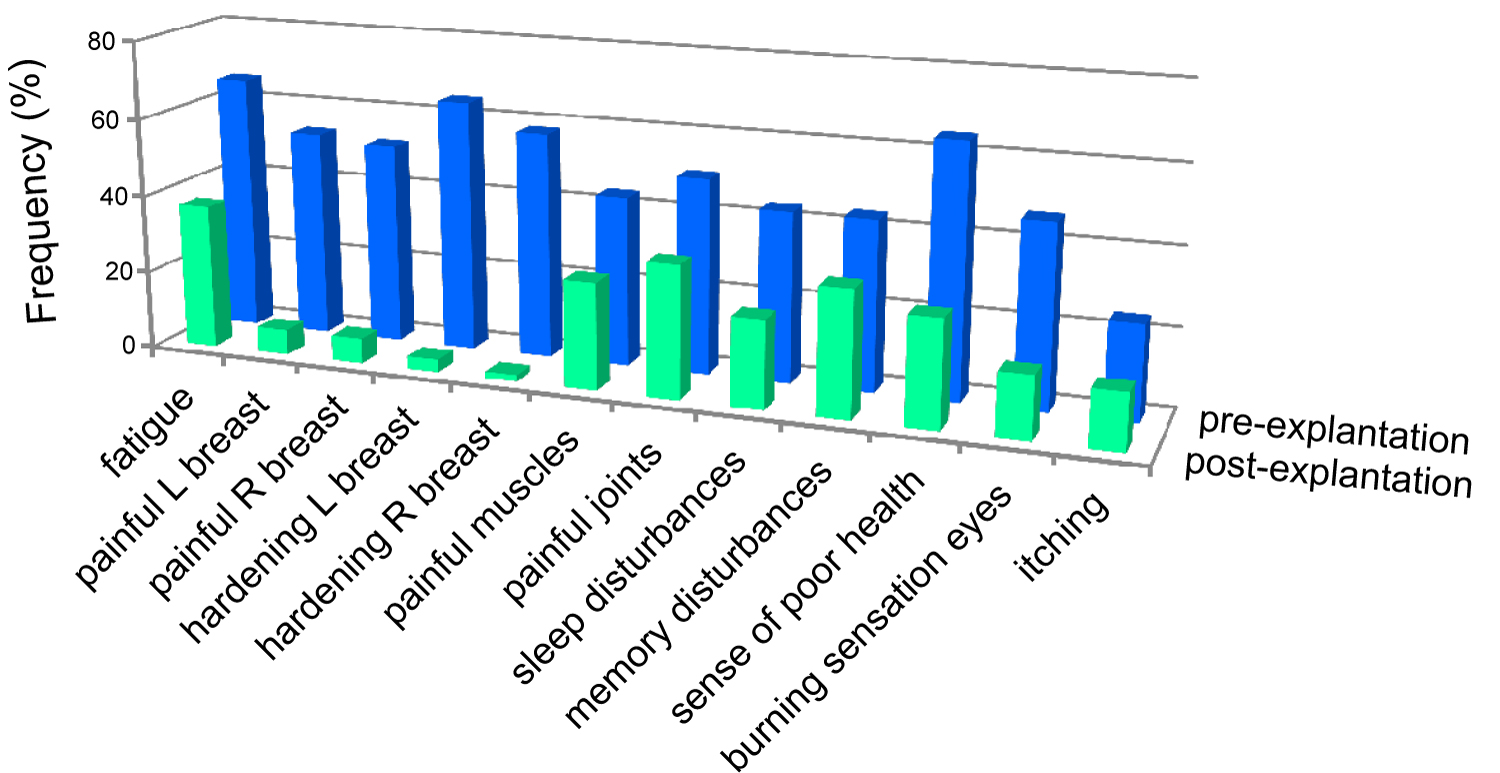 Figure 2: Effects of explantation on the health complaints reported by Group 3. The bars correspond to the percentage of subjects in Group 3 reporting complaints (specified below the graphs) with scores of 4 and higher. Pre-explantation: Before explantation; post-explantation: After a mean follow-up of 3.5 years. View Figure 2
Figure 2: Effects of explantation on the health complaints reported by Group 3. The bars correspond to the percentage of subjects in Group 3 reporting complaints (specified below the graphs) with scores of 4 and higher. Pre-explantation: Before explantation; post-explantation: After a mean follow-up of 3.5 years. View Figure 2
In addition to the information acquired by the questionnaires, information on capsular contraction and the presence of implant rupture was obtained from the clinical files, based upon observations made during the operation. The generation of silicone breast implant was derived from the year of implantation. These additional data are presented in Table 1. Of the complete group of 101 patients, 81% had health complaints that could not be ignored. In addition, 56% had a ruptured implant, and 57% had serious capsular contraction (Baker III and Baker IV combined). Hardly any difference was observed between Group 2 and Group 3 with respect to implant rupture and capsular contraction. From these data it becomes clear that the majority of both Group 2 and Group 3 patients had type 3 implants, which according to Peters and co-workers [8] already had a bleed retardation layer. Nevertheless, in both groups we observed extensive capsular contraction, implant rupture and health complaints.
Table 1: Characteristics of study groups. View Table 1
Up till now the existence of a disease that is related to gel bleed of silicone breast implants has for decennia been debated in the medical disciplines involved. Epidemiological studies do not support the notion that there is a strong connection between silicone breast implants and any illness [9]. In this respect it is important to note that the existence of silicone implant-related health complaints was largely denied by the clinicians involved and therefore these health complaints were not reported in their medical files. As a consequence, the potential relationship remained unnoticed in any meta-analysis. The only study that clearly demonstrated the presence of health complaints in relation to silicone breast implants was omitted from meta-analyses. This was a study on self-reported health complaints by female health professionals and was considered to be subjective [10]. Silicone gel bleed from implants is a well-established phenomenon, but one cannot technically determine the concentration of intact whole silicone molecules exuded from the implants into the body, although the number of their constituent silicon atoms can be measured. This makes it impossible to link them to silicone breast implants and likewise to develop a diagnostic test.
Animal studies have been proposed to study the effects of silicone gel bleed, but this will only be meaningful after a very long implantation time (> 15 years) in the animals. If feasible, silicone-containing molecules will probably be found just as they are found in tissues of women who had silicone breast implants for many years, but it might be difficult to assess health complaints in these animals. Apart from that, the accumulation of silicone molecules in various tissues of these animals cannot be extrapolated to humans and, therefore, the information may not be considered relevant for BII. All this leaves us therefore with no other option than deduction and reasoning of the known facts regarding BII or continue with an everlasting unsolvable scientific controversy, which has its advantages for some stakeholders.
The localization of silicone molecules in tissues of women with breast implants does not tell us how the molecules bring about the complaints. If explantation of silicone implants affects the health complaints, it stands to reason that they are related to one another. Our study demonstrates that explantation surgery of silicone breast implants in symptomatic woman ameliorates their complaints in the majority of cases.
There have been a number of other studies [11-15] that document the beneficial postoperative effects on health complaints in women who had explantation of their breast implants. The results of our study are in accordance with these publications. In addition it demonstrates that symptomatic women who continue with silicone breast implants can expect that, once present, these complaints will not disappear spontaneously upon the reassurance that the complaints are not caused by the silicone breast implants. In any case, health related complaints in patients with silicone breast implants demand explantation surgery or replacement with non-silicone gel-filled implants.
During the explantation surgery the surgeon should strive for a total capsulectomy. This is technically easier when the implant is located subglandularly and the capsule is contracted into a thick membrane. If the implant is located behind the breast muscle, than often a total capsulectomy is very difficult with regular surgery, risking further damage to the muscle. In that case one should accept a partial capsulectomy. An attempt can be made to remove the capsule with microsurgery. The remaining part of the capsule can disappear spontaneously, and this is more likely if the capsule is very thin, but a thick capsule can remain present even after many years [16]. A minority of cases will show no health improvement after explantation, not even after a total capsulectomy, and this is particularly observed after a very long time of implantation and in case of many health complaints.
The aim of explantation surgery is to remove the silicone breast implant and with that the gel bleed is terminated. However, surgically nothing can be done in terms of removing the molecules that have migrated throughout the body, and which most likely are responsible for the symptoms. Yet, when one evaluates the postoperative results, most of the time, but not always, health improvement is evident. It is important to note that very often, already starting right after explantation surgery, there is a substantial increase in the severity of the health complaints, which can last for days, weeks, and even months before they ameliorate. This phenomenon is not always present, but often enough to warrant a preoperative discussion of this subject with the patient. This may be caused by the cleaning of the area of the silicone breast implants during surgery, with pressure on the implant during the operation, thus increasing gel bleed, while blood vessels are being open.
The many generations of silicone breast implants will probably differ in their degree of bleeding, which is the reason why they were created in the first place. The normal body temperature will augment the motility of silicone oligomers and polymers and enhance the efficiency of bleeding. Moreover, gel bleed is associated with swelling of the elastomeric material, which leads to a decrease in tensile strength and weakening of the shell [17]. This has two effects. First, it becomes easier for subsequent molecules to bleed, which progressively increases the amount of bleeding, and second, it eventually leads to a spontaneous rupture of the shell, which is very often encountered during operation or by preoperative examination with imaging technologies.
Adverse effects of silicone breast implants have been addressed in many studies [5,18-20]. Various disease names have been proposed and many ideas on the pathogenesis have been postulated. However, when it comes to the real characteristics of the silicone breast implant related health issue, the neutral term "Breast Implant Illness" (BII) to describe this illness seems to be most suitable. There have also been attempts to classify the health complaints as belonging to a predominantly autoimmune disease (ASIA), but over decennia not a single marker has unequivocally been identified in relation to the health complaints of silicone breast implant carriers. Moreover, autoimmune therapy has never cured a patient with BII. Looking at the great variety of the types of symptoms, occurring in unison in these patients, and the variety of severity of the symptoms, one can only assume that many organ systems in the body are simultaneously involved, including the immune system, which should therefore not be considered to be the sole orchestrator of BII. There are also many more women with BII than there are women with a typical autoimmune disease in relation to silicone breast implants. Moreover, in the cadaveric study (7), there were no accumulations of cells belonging to the immune system identified. Therefore, the term ASIA in this context is misleading and should be refuted, but this does not deny that abnormal immune phenomena could be present is some patients with BII as part of their health complaints.
Taken together, the only intervention that seems to have an ameliorative effect on the health complaints in patients who have silicone breast implants, is explantation surgery.
Women with different types and generations of silicone breast implants have in common that gradually over many years various health complaints become manifest. This is most likely explained by gel bleed from the implants, because the complaints change, usually for the better, when the gel bleed is eliminated through implant removal. Women with BII should therefore be advised to have explantation surgery of their silicone breast implants, preferably in combination with a capsulectomy, in order to stop the exposure to continuously bleeding silicones and to give them a chance to recover to some extent. Women should also be informed of this phenomenon of gel bleed and the potential health effects before implantation surgery and likewise this should be included in their informed consent.
The authors thank all participating women for their contribution. Dr. Kappel owns and acts as a plastic surgeon at the Dr. Kappel Institute. Dr. Pruijn has nothing to disclose. No funding was received for this article.