Soluble ST2 might be a sensitive marker of rheumatic (peri-)carditis before and during antirheumatic treatment.
We describe a case of rheumatic heart disease with pericarditis and mild left ventricular dysfunction which was successfully treated by immunosuppression. An interesting pattern of activation of the cardiac marker ST2 was observed at detection and during treatment. It suggests ST2 as biomarker for diagnosis and monitoring of disease activity in rheumatic heart disease.
Rheumatic disorders have a prevalence of roughly 2% and often involve the heart. Cardiac involvement may include pericarditis, myocarditis, endocarditis, accelerated valve disease and conduction disturbance [1]. Although better treatment options have become available for rheumatic diseases, the presence of cardiac involvement is still a matter of great concern and a leading cause of morbidity and mortality. Nevertheless, the presence and severity of cardiac involvement in rheumatic disease is difficult to determine and often requires (repeated) imaging by echocardiography, angiography and/or MRI.
The cardiac biomarker NT-proBNP is a valuable prognostic marker for the assessment of cardiac risk in patients with rheumatic disorder [2,3]. However, it is still unclear, whether NT-proBNP reflects comorbidities such as (pulmonary) hypertension [4], coronary artery disease, left ventricular dysfunction and/or the risk associated with anti-inflammatory therapy [5] rather than direct affection by the rheumatic disease.
Therefore, a complementary cardiac biomarker, which directly indicates cardiac affection and allows for detection and monitoring of rheumatic heart disease is still lacking. Our current case suggests that ST2 might be such a marker.
A 46-year-old male patient was admitted to our hospital with progressive dyspnea and coughing. He had suffered from polyarthralgia, nocturnal sweats and progressive weakness for three months. Three years ago, an exsudative pleuritis was treated by pleurectomy and pleurodesis and an increased ANA-titer was detected. Current medical treatment was by NSAIDs only.
Upon echocardiography, a pericardial effusion and slightly decreased left ventricular pump function (EF 49%) were noted (Figure 1a). Upon cardiac MRI, slightly decreased left ventricular pump function was confirmed (EF 48%) and fibrosis was excluded by absence of late gadolinium enhancement (Figure 1b). Upon coronary angiography, there was no evidence of coronary arteriitis or obstructive coronary artery disease. On ventriculography, elevated filling pressures and slightly reduced left ventricular function were noted. Laboratory testing revealed an increased titer of antinuclear antibody (ANA, 1:1000) but no antibodies against ds-DNA, p-ANCA or c-ANCA. Further tests for rheumatoid arthritis (rheumatoid factor (RF), Anti-cyclic citrullinated peptide (Anti-CCP)), sarcoidosis (soluble interleukin 2 receptor (sIL-2R), angiotensin-converting enzyme (ACE)), borreliosis (serology test), tuberculosis (QuantiFERON, sputum) and other infectious diseases were negative. On the basis of these findings, a diagnosis of acute exacerbation of undifferentiated connective tissue disease with associated (peri-)carditis was established.
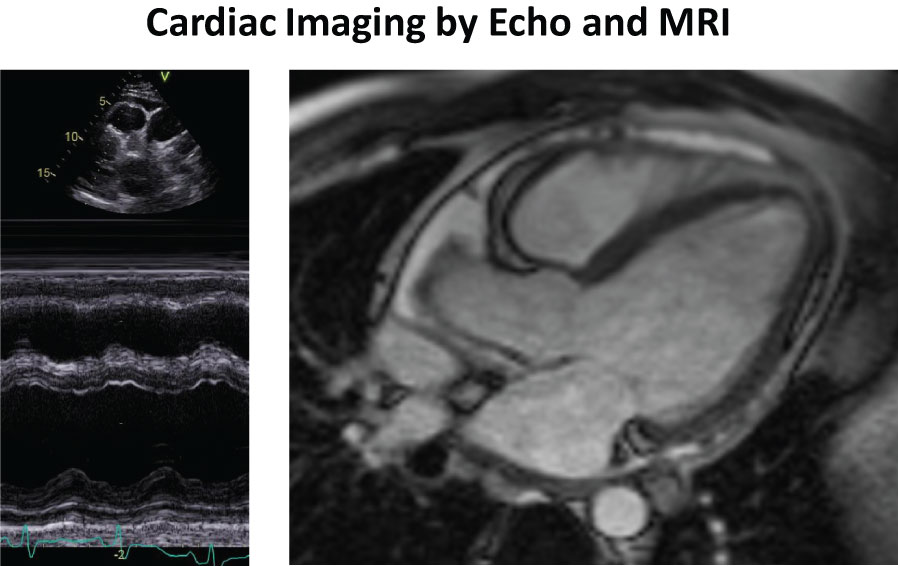 Figure 1: a, left) M-mode Echocardiography showing pericardial effusion and slightly decreased LV-function; b, right) Cardiac MRI showing presence of pericarial effusion and absence of late gadolinium enhancement.
View Figure 1
Figure 1: a, left) M-mode Echocardiography showing pericardial effusion and slightly decreased LV-function; b, right) Cardiac MRI showing presence of pericarial effusion and absence of late gadolinium enhancement.
View Figure 1
Immunosuppressive therapy with corticosteroids (0.5 mg/kg, tapering to maintenance dose over a period of 6 weeks) and azathioprine (50 mg/d starting dose) was initiated. The patient responded well to treatment and repeat echocardiography showed decreasing pericardial effusion and decreasing inflammatory markers. Six weeks later, during a follow-up visit, the pericardial effusion had disappeared and cardiac function had normalized.
Regarding the cardiac markers, ST2 was excessively elevated (214 pg/ml, cut-off 35 pg/ml, 644% of upper limit of normal) upon presentation, whereas NT-proBNP was only mildly elevated (271 pg/mL, cut-off 150 pg/ml, 217% of upper limit of normal). After initiation of immunosuppressive treatment, ST2 decreased rapidly whereas NT-proBNP increased slightly further. After six weeks of treatment, both cardiac markers had normalized completely (Figure 2).
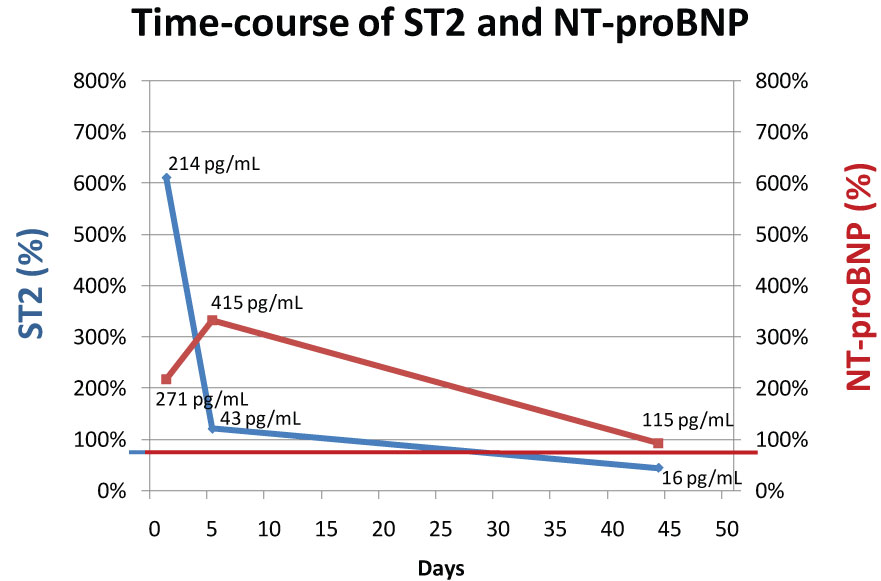 Figure 2: Time-course of ST2 (blue) and NT-proBNP (red). X-axes show % of upper limit of normal (ULN). ULN of ST2 is 35 pg/mL and of NT-proBNP 125 pg/mL. Numbers inside the figure depict absolute concentrations of ST2 and NT-proBNP.
View Figure 2
Figure 2: Time-course of ST2 (blue) and NT-proBNP (red). X-axes show % of upper limit of normal (ULN). ULN of ST2 is 35 pg/mL and of NT-proBNP 125 pg/mL. Numbers inside the figure depict absolute concentrations of ST2 and NT-proBNP.
View Figure 2
ST2 is a protein biomarker of cardiac injury and belongs to the interleukin 1 receptor family. It has two isoforms, soluble ST2 and the membrane-bound ST2 receptor. The ligand for ST2 is the cytokine IL-33. In the heart, binding of IL-33 to the ST2 receptor has a cardioprotective effect. The protective IL-33 signal is counter balanced by soluble ST2, which also binds to IL-33 and makes it unavailable to the ST2 receptor. Consequently, high levels of soluble ST2 promote negative effects on the heart and elevated ST2 has been reported as strong biomarker of adverse prognosis in heart failure [6,7]. The IL-33/ST2 axis has also been linked to adult-onset Still's Disease, Sjögrens Syndrome and Rheumatoid Arthritis, but not yet specifically to peri-(myocarditis) or rheumatic heart disease.
Although the source of ST2 in the current case is not known, it has been shown that endothelial cells seem to be the source of ST2 in the cardiovascular system [8,9] and in adipose tissue [10]. Release of ST2 has also been demonstrated from human and mouse mast cells [11]. Since our patient only had signs of cardiac involvement and no other current signs of rheumatic disease, we assume that the source of ST2 were endothelial cells, possibly of the epicardium and pericardium. Interestingly, the initial concentration of ST2 in our patient was higher than any other ST2 measurement in a series of patients with acute or chronic heart failure due to traditional causes and such points to a particularly important role for ST2 in rheumatic (peri-)carditis.
In summary, we speculate that the high initial concentration of ST2 during the acute attack of rheumatic heart disease and the sharp decline and normalization of ST2-concentration in response to antirheumatic treatment indicates that ST2 might be a potential marker of rheumatic (peri-)carditis as well as a marker of cardiac disease activity during antirheumatic treatment. Further, that ST2 might be a more sensitive marker than NT-proBNP in cases of rheumatic cardiac involvement without severe left ventricular dysfunction or overt heart failure. ST2 should therefore be assessed cross-sectionally in a larger cohort of patients with rheumatic disorder with and without cardiac involvement and longitudinally as a marker of disease severity and prognosis during antirheumatic treatment in rheumatic patients with cardiac involvement.
We gratefully acknowledge the technical support of Mr. Otto Lorenz, who performed the ST2 measurements.
RJ, AvM, MF and AL report no conflicts of interest.
Laboratory kits for measurement of ST2 (ASPECT-PLUS ST2 on ASPECT reader) were provided at no cost by bestbiondx diagnostics, Horbeller Str. 33, 50858 Köln, Germany.