Xanthelasmas are lesions containing lipid-filled histiocytes, which can be found on the skin or in the gastrointestinal tract. Gastrointestinal xanthelasmas (GX) are rare lesions typically found incidentally on esophagogastroduodenoscopy. Several risk factors have been identified for development of GX including dyslipidemia, fatty liver, radiotherapy, Helicobacter pylori infection, and immunosuppression. We present a case of a gastroesophageal xanthelasma in a 78-year-old male without risk factors. While his xanthelasma was anatomically located in the esophagus, the surrounding tissue was gastric mucosa, suggesting that it had occurred in a sliding hiatal hernia. The etiology of his GX was likely related to age and/or mucosal inflammation at the hiatal hernia. Recently, studies have suggested an association of GX with malignancy. However there are no current guidelines about surveillance of GX or screening for malignancy in patients with GX. Our aim is to review a unique case of gastroesophageal GX and discuss the current literature available on this topic to highlight the clinical significance of GX.
Xanthelasma, Gastrointestinal tract, Endoscopy, Lipid disorders, Premalignant
GX: Gastrointestinal Xanthelasmas; GI: Gastrointestinal; CXs: Cutaneous Xanthelasmas; EX: Esophageal Xanthelasma; EGDs: Esophagogastroduodenoscopies
Xanthelasmas, or xanthomas, are lesions containing lipid-rich histiocytes. They are most commonly seen on the skin but can also occur in the gastrointestinal (GI) tract. Cutaneous xanthelasmas (CXs) occur in 1.1% of females and 0.3% of males and are most commonly found in the periorbital area [1]. Gastrointestinal xanthelasmas (GXs) are extremely rare lesions with an estimated prevalence of 0.2-0.8% [2]. They have been found in various parts of the GI tract, most commonly in the stomach with a frequency of 76% of GXs [2]. They are rarely found in the esophagus, accounting for 12% [2]. To date, there are only 22 cases of esophageal xanthelasma (EX) reported in the English literature including the case presented here [3-5]. The characteristic endoscopic appearance of GX is that of a yellow-colored nodule or polyp [6]. They are typically solitary lesions.
GX are typically asymptomatic and are discovered incidentally on esophagogastroduodenoscopies (EGDs) done for a variety of indications. However, it is unlikely that symptoms such as abdominal pain or dyspepsia, which prompted endoscopic evaluation, are caused by solitary GX lesions. In the very rare case of multiple xanthomas, or xanthomatosis, there have been few reports of GI motility disorders [7]. However, this has not been seen with solitary lesions. Here, we present a case of GX presenting as an incidental finding on EGD, review the literature, and discuss the clinical significance.
A 78-year-old male with history of alcoholic cirrhosis presented to our gastroenterology clinic for evaluation of chronic normocytic anemia. A review of symptoms was positive for constipation, but negative for hematemesis, hematochezia, melena, and weight changes. An esophagogastroduodenoscopy (EGD) had been done 2 years prior and revealed portal hypertensive gastropathy with a normal esophagus and duodenum. Other past medical history included chronic obstructive pulmonary disease, hypertension, benign prostatic hypertrophy, stroke, and vascular dementia. He had no significant family or surgical history. Medications included metoprolol tartrate, lisinopril, atorvastatin, tamsulosin, finasteride, escitalopram, mirtazapine, gabapentin, and aripiprazole. His social history was significant for heavy alcohol use in the past, and a 15 pack-year smoking history.
At the time of endoscopy, his vital signs were within normal limits. He had a BMI of 22. He was an older male in no acute distress. He was normocephalic with moist mucus membranes, normal conjunctiva and clear oropharynx. His cardiopulmonary examination was normal. The abdomen was soft, nontender, and nondistended. There were no skin lesions, specifically no cutaneous xanthelasmas were noted. Recent lab work was significant for anemia with hemoglobin of 9.3 g/dL (ref. 13.5-17.5 g/dL), but ferritin and a lipid panel was within normal limits.
He underwent EGD for variceal screening which showed grade I esophageal varices, persistent portal hypertensive gastropathy which likely contributed to his anemia. There was also a single, small, round well circumscribed, pale yellow esophageal polyp above the gastroesophageal junction, measuring 0.3 × 0.5 cm (Figure 1). The lesion was biopsied due to its interval development since last EGD 2 years prior. The biopsied tissue demonstrated gastric cardia-type mucosa with abundant foam cells within the lamina propria (Figure 2). The tissue was positive for CD68, a marker of histiocytes. This was consistent with a diagnosis of xanthelasma. There was no histological evidence of metaplasia or dysplasia. Gastric biopsies were negative for Helicobacter pylori. Given the anatomic location in the esophagus but the gastric-type mucosa seen pathologically, this lesion likely originated in a segment of sliding hiatal hernia. A colonoscopy was performed at the same visit, and no colonic xanthomas were identified.
 Figure 1: Endoscopic view of the distal esophageal xanthelasma showing a yellowish, polypoid appearance. View Figure 1
Figure 1: Endoscopic view of the distal esophageal xanthelasma showing a yellowish, polypoid appearance. View Figure 1
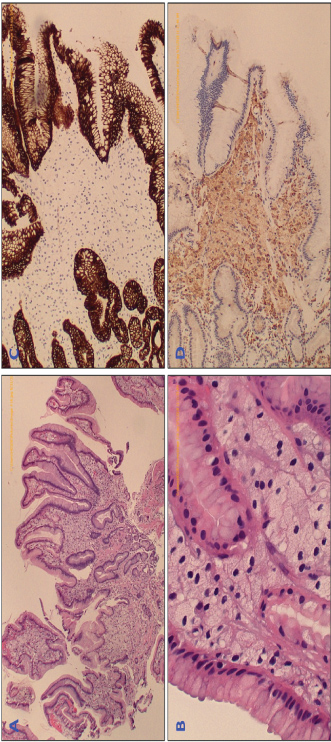 Figure 2: The biopsied tissue was prepared with H&E staining, which is seen at 4X (A) and 40X (B) and demonstrates many foam cells within the lamina propria (indicated by blue arrows). Special staining was performed to differentiate the type of lesion. Staining for cytokeratin CAM (C) was negative, excluding epithelial neoplasm, however the histiocyte-marker CD68 (D) was positive, confirming diagnosis of xanthelasma. View Figure 2
Figure 2: The biopsied tissue was prepared with H&E staining, which is seen at 4X (A) and 40X (B) and demonstrates many foam cells within the lamina propria (indicated by blue arrows). Special staining was performed to differentiate the type of lesion. Staining for cytokeratin CAM (C) was negative, excluding epithelial neoplasm, however the histiocyte-marker CD68 (D) was positive, confirming diagnosis of xanthelasma. View Figure 2
There are several proposed risk factors for development of GX, including dyslipidemia, fatty liver, H. pylori infection, and immunosuppression. Our patient had no significant risk factors, making GX an unexpected finding in this patient.
Dyslipidemia is one of the most well-studied risk factors in the development of xanthelasmas. Patients with CX have increased LDL levels and decreased HDL levels [8]. Unlike CX, the association of GX with dyslipidemia is unclear. One study showed that the occurrence of GX was higher in patients with elevated triglyceride and fasting glucose levels compared to those with normal levels [9]. However, there was no association with hemoglobin A1c, low-density lipoprotein or total cholesterol levels [9]. This study did not describe patient characteristics such as body-mass index, waist circumference, use of cholesterol-lowering or glycemic-control medications, and other factors which may have affected the metabolic profile of their study population. Inclusion of this information would have been valuable in the interpretation of the findings relative to the general population.
A study by Chen, et al. retrospectively evaluated 1370 patients with GX and reported a more frequent incidence in patients with fatty liver. However, there was no relationship with chronic liver disease [9]. Unfortunately, the authors did not define which conditions were included in "chronic liver disease". Therefore, it is difficult to extrapolate this finding to our patient's alcoholic cirrhosis as a risk factor for GX.
H. pylori has been shown to be more prevalent in patients with GX, at a rate of 31% [9]. The relationship between radiation and GX has been suggested in several case reports [10,11]. These observed associations support a pathogenic theory involving mucosal trauma. Radiation therapy of nearby structures may also have a role in development of GX. However, a study evaluating esophageal pathology following mediastinal radiation did not reveal any cases of EX [10,12]. Immunosuppression has been suggested as risk factor for GX by several case reports, however this association is not well supported in the literature [5,13]. Despite the numerous potential risk factors for GX, our patient appears to have had a relatively low risk. He had no history of immunosuppression, radiation therapy, dyslipidemia, or H. pylori infection.
While the exact etiology of GX is not clear, one proposed mechanism is reactive inflammation to mucosal damage or trauma [4]. The relative frequency of these lesions along gastrointestinal tract supports this theory. The esophagus is lined with squamous epithelium which is more resistant to trauma compared to gastric columnar epithelium [7]. Therefore, the prevalence of EX is significantly less. A case report by Uehara, et al. suggested that the histiocytes involved in GX formation consist of a subtype known as M2 histiocytes [4]. Macrophages are normally found in the lamina propria of the GI mucosa and remove unwanted particles using a scavenger receptor. The M2 histiocytes play a role in late stage inflammation, where M2 cells ingest the inflammatory debris [7]. In the case of GX, histiocytes phagocytose lipids, in addition to this inflammatory debris, to form foam cells. This is consistent with the theory that GX are reactive lesions, formed in response to mucosal inflammation. Because this xanthelasma occurred within an area of hiatal hernia, mucosal inflammation due to hernia sliding, friction and reflux, likely contributed to GX development. Others have proposed that xanthelasmas develop as a normal feature of the gastrointestinal mucosa as it ages [4,6,9,14]. This case supports this theory as this patient developed GX at age 78 since his last endoscopic exam 2 years prior.
At the time of this patient's EGD, the precise diagnosis of his esophageal lesion was not evident. The histologic findings delivered the final diagnosis of GX. Histologically, xanthelasmas are identified by the presence of foam cells, or histiocytes containing lipid-filled vacuoles in the lamina propria of GI mucosa or the superficial dermis layer in the skin [2]. The lipid, or oxidized low-density lipoprotein, can be distinguished with Sudan black or oil red O staining and may also help differentiate it from mucin- or glycogen- containing cells seen in Whipple's disease or other metabolic storage disorders [2]. Immunohistochemical staining is typically positive for CD68, CD163, and TGF-beta, which are markers of histiocytes [4].
Although GXs are generally considered to be benign lesions, recently there have been some studies raising concern that GX may be a precursor to gastric malignancy. In a matched-control study, gastric cancer was identified in 21% of patients with a GX, compared to 3% in patients without [1]. The authors concluded that GX was significantly associated with gastric cancer and GX may represent a "biomarker" for malignancy [1]. Additionally, gastric cancer was noted to be more common in patients with 3 or more GXs [6]. In that study, the prevalence of GX was 7.7%, which is significantly higher than the prevalence of the general population. Therefore, there may have been some population bias. Another study demonstrated a higher rate of intestinal metaplasia and dysplasia in patients with GX [9]. More specifically, intestinal metaplasia was seen in 49.6% of patients with GX and dysplasia was seen in 2.1%, which was 7-times greater than in patients without GX [9]. A case report also suggested a link between GX and malignancy, and proposed that underlying cancer cells may stimulate proliferation of xanthoma cells [15]. Due to the high mortality of gastric cancer, it is important to determine whether GXs are a precursor lesion of gastric cancer. The current data represent only statistical associations, and there is currently no clear link to any other GI malignancy. Additionally, it is unclear if there is a malignant potential for xanthelasmas in other parts of the gastrointestinal tract, such as the esophagus.
Our case highlights the pathogenesis of GX. Arising within a hiatal hernia, mucosal injury from sliding friction and reflux predisposed this patient to development of a GX. This case also raised the question about surveillance of these types of lesions given the possible risk for pre-malignancy. Currently, there are no guidelines regarding surveillance for of GX or screening for GI malignancy in patients with GX. While this patient may need further endoscopic evaluation for other reasons, we felt there was no need for additional endoscopic follow up for his GX. Overall, the potential of GX to "predict" gastrointestinal cancer requires further investigation.
Special thanks to Dr. Poormina Hegde of the University of Connecticut Department of Pathology for providing images and descriptions of pathologic components of this case.
None.
Mavilia: Drafting manuscript, literature review; Wu: Clinical supervision of case, major revisions.