Endometrial cancer is one of the most frequent gynecological cancers in women. According to a recent national survey, endometrial cancer has increased rapidly in the past 30 years in Taiwan and the incidence of endometrioid adenocarcinoma has increased more and more quickly in younger age. Although total hysterectomy is usually recommended to eradicate the malignant lesion, this procedure may not be suitable for a young nulliparous woman, especially when the endometrial cancer is early stage. We report a case of consecutive successful pregnancies by in vitro fertilization and embryo transfer (IVF-ET) after conservative management of early stage endometrial carcinoma. To our knowledge, this is the first such case ever been reported in Taiwan. We believe this case will be very encouraging to younger women with early stage endometrial cancer.
Endometrial cancer, Fertility-sparing therapy, Progestin
Endometrial cancer is one of the most frequent gynecological cancers in women. While the majority of cases are diagnosed in post-menopausal women, up to 14% of cases happen in the pre-menopausal women including 4% diagnosed in women less than 40 years of age [1,2]. According to a recent national survey in Taiwan, cases of hormone- dependent type I endometrial cancer have increased rapidly in the past 30 years and the incidence of endometrioid adenocarcinoma has increased more and more quickly in younger age [3]. Standard management of endometrial cancer at diagnosis involves hysterectomy with bilateral salpingo-oophorectomy, followed by chemotherapy with or without radiation therapy [4]. Since some of the young endometrial cancer patients may not have completed their family planning at the time of diagnosis, fertility preservation has become one of the most important issues in the past few years. Here we present the case of a patient who achieved consecutive successful pregnancies via assisted reproduction technologies after conservative management of early stage endometrial carcinoma.
The patient, a 39-year-old woman, BMI 26.6, came to our hospital due to primary infertility for the past seven years. She had an irregular menstruation and basal body temperature revealed no biphasic change. Multiple small follicles in both ovaries with necklace like appearance were noted during ultrasound examination. The serum level of follicular stimulating hormone (FSH) was 5.28 mIU/mL (reference range: 3.03-8.08 mIU/mL) and luteinizing hormone (LH) was 9.6 mIU/mL (reference range: 1.80-11.78 mIU/mL). Diagnosis of polycystic ovary syndrome was made according to Rotterdam criteria (Rotterdam ESHRE/ASRM-Sponsored PCOS consensus workshop group, 2004) [5]. The patient did not return after one cycle of unsuccessful artificial insemination. She visited our hospital again two years later requesting in vitro fertilization and embryo transfer (IVF-ET) directly due to advanced maternal age. Routine hysteroscopy (HSC) examination before treatment found a small vessel-rich mass in the uterine cavity (Figure 1). Biopsy was performed and endometrioid adenocarcinoma was diagnosed (Figure 2). Magnetic resonance imaging (MRI) of the uterus showed an intact endomyometrial junctional zone. There was no pathology regarding lymph nodes or metastatic disease nor suspicious adnexal mass (Figure 3).
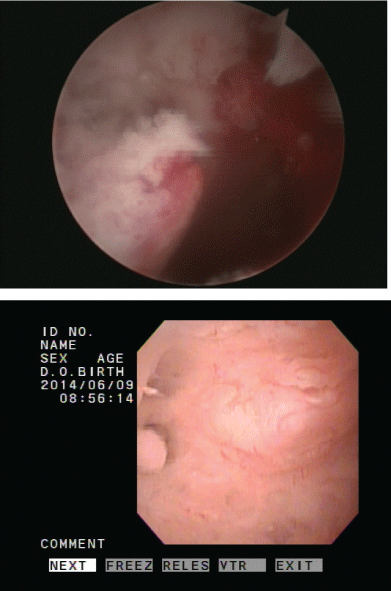 Figure 1: Hysteroscopic finding of the endometrial cancer.
View Figure 1
Figure 1: Hysteroscopic finding of the endometrial cancer.
View Figure 1
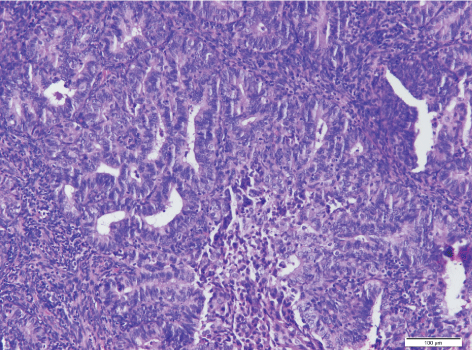 Figure 2: Pre-therapy. Endometrial glands with neoplastic transformation, H&E, 200×.
View Figure 2
Figure 2: Pre-therapy. Endometrial glands with neoplastic transformation, H&E, 200×.
View Figure 2
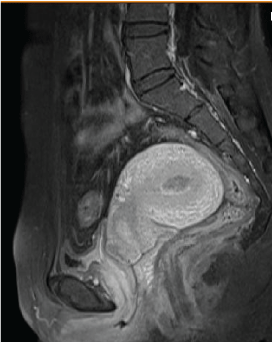 Figure 3: MRI finding before Fertility-sparing therapy.
View Figure 3
Figure 3: MRI finding before Fertility-sparing therapy.
View Figure 3
Due to the patient's desire to retain her child-bearing potential, conservative management was decided after thorough consultation. Dilation and curettage (D&C) was arranged and the report showed low grade endometrioid adenocarcinoma without evidence of lymphvascular space invasion (LVSI). There were no contraindications to medical therapy nor pregnancy. Ovulation induction followed by IVF was performed and all 4 embryos were frozen. Medroxyprogesterone acetate (MPA) 5 mg twice daily was given after the D&C for 24 days until oocyte retrieval. She then received medroxyprogesterone acetate (Farlutal) at a dose of 250 mg/d for four months. A repeat HSC with endometrial D&C revealed no residual cancer (Figure 4). Frozen embryo transfer (FET) with three embryos was arranged but was unsuccessful. Another IVF-ET was arranged immediately during the next cycle. Three embryos were transferred, and three embryos were frozen. A single gestational sac was noted at transvaginal ultrasonography ten days after the positive pregnancy test. The course of pregnancy was uneventfully, and she gave birth to a healthy female baby at term. Hysterectomy was recommended for her safety, but patient refused and requested FET during the following year. Farlutal at a dose of 250 mg/d was prescribed and FET with the remaining three embryos was performed after normal endometrial biopsy. She gave birth to her second female child nine months after the second FET. She is currently receiving regular follow up at our hospital and preparing for hysterectomy.
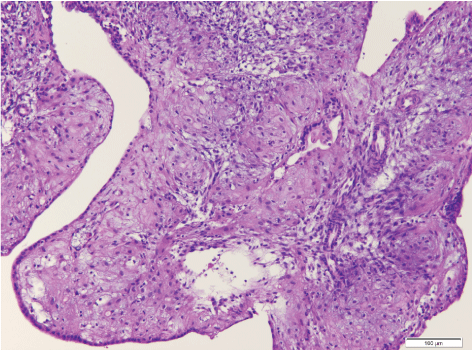 Figure 4: Post-therapy with high-dose systemic progesterone. Stroma with decidual transformation (drug-induced). Hyperplasia within the glandular tubes and on the endometrium surface - foci of residual hyperplasia, H&E, 200×.
View Figure 4
Figure 4: Post-therapy with high-dose systemic progesterone. Stroma with decidual transformation (drug-induced). Hyperplasia within the glandular tubes and on the endometrium surface - foci of residual hyperplasia, H&E, 200×.
View Figure 4
Progestin treatment of atypical hyperplasia and well differentiated carcinoma of the endometrium in women under age of 40 was first published by Randall, et al. at 1997 [6]. Since then, many patients with early stage endometrial carcinoma have been reported to have achieved successful pregnancies after conservative medical management [7-9]. Because recurrence after successful fertility-sparing therapy of endometrial cancer is very high, prophylactic hysterectomy is usually performed once patient has given birth to a child. Until now, there are no data regarding how many pregnancies a woman could support after fertility-sparing therapy of early endometrial cancer. The case we present here with two consecutive successful pregnancies is encouraging for women diagnosed with early stage endometrial carcinoma who wish to have more than one child.
According to the recommendation from American College of Gynecology (ACOG) in 2011, for women with low risk of endometrial cancer (grade 1 or 2, stage IA, endometrioid endometrial carcinoma, confined to the endometrium) who wish to preserve their fertility, conservative management such as progestin-based therapy can be considered as long as she fulfilled the following criteria; Absence of suspicious or metastatic disease on imaging, absence of lymphovascular space invasion (LVSI) on adequate curettage specimen, no evidence of a suspicious adnexal mass on CT or pelvic ultrasound imaging, strong and diffuse expression of progesterone receptors on immunohistochemical staining of an endometrial biopsy or curettage specimen, no contraindications to medical therapy or pregnancy were all basic requirements to perform this "fertility sparing therapy" [10-12].
Current fertility-sparing treatment modalities mainly comprise hormonal therapies involving progestins, progestin-releasing intrauterine devices, natural progesterone, oral contraceptives, selective estrogen receptor modulators, gonadotropin-releasing hormone agonist, and aromatase inhibitors. Among which, oral progestin therapy is the one of the most commonly used with well-established efficacy. Progesterone has long been known to suppress the growth of endometrial cancer by down-regulating estrogen receptors, activating enzymes in estrogen metabolism, involving cell cycle regulation by cyclin-dependent kinase (Cdk) [13], and reinforcing p27 (a cyclin E-Cdk2 complex inhibitor) expression resulting in suppression of the cell cycle [14]. Treatment of low-risk endometrial cancer with oral MPA in a range of doses from 2.5 to 1500 mg daily has been reported. The most commonly used dose is 600 mg daily. The range of doses of megestrol acetate (MA) has been reported from 10 to 400 mg daily and the most commonly used dose is 160 mg daily [15]. The choice of dose is based on a need to balance efficacy with toxicity. For patients who dislikes oral medicine, progestin-releasing intrauterine devices seem to be an optimal alternative. Wei J [16] conducted a meta-analysis comparing the efficacy of different fertility-sparing treatments in patient with early endometrial cancer or atypical complex hyperplasia. Complete remission (CR) was found in 71% of patients managed with progestin, 76% with progestin-releasing intrauterine devices and 87% for women treated with both. However, the pregnancy rate was 34% in progestin group (live birth rate of 20%), 18% in IUD group (live birth rate of 14%), and 40% in progestin plus IUD group (live birth rate of 35%). They concluded for patients with early endometrial cancer and atypical complex hyperplasia that treatments with progestin, with or without IUD or IUD alone, can achieve good CR rate; however, the pregnancy outcomes might be less in patients treated with IUD alone.
Though fertility-sparing treatments seem promising, these are not without risk. For example, the optimal dosage and duration of oral progestin remains to be determined and there is a high rate of recurrence after fertility-sparing therapy [17]. Recurrence risk ranges from 24 to 41 percent, with a mean time to recurrence between 15 and 35 months [18,19]. Therefore, thorough selection of patients through comprehensive pretreatment evaluation and counseling is needed before offering this nonstandard therapy. Close follow-up during therapy is very important and, hysterectomy after completion of childbirth is recommended as a definitive treatment. Our patient who achieved two consecutive successful pregnancies after diagnosis of early endometrial cancer is encouraging and demonstrates the feasibility of fertility sparing therapy.