An 82-year-old male patient was admitted to our clinic because of shortness of breath and chest pain. A grade 4/6 diastolic murmur was heard on auscultation. Physical examination revealed signs of congestive heart failure and poor peripheral perfusion. There was a diagnosis of acute Stanford Type A ascending aortic dissection in the history of the patient. The patient had refused emergency surgical intervention three years ago. Computed tomography revealed chronic Stanford Type A dissection and dilatation of the ascending aorta. The patient had 2°-3° aortic insufficiency and calcified bicuspid aortic valve in an echocardiography, was detected 90 mmHg aortic valve gradient. Graft replacement of the ascending aorta and serape aortic valve replacement were performed successfully under a cardiopulmonary bypass (using both femoral and innominate artery with a Y-shaped connector) with open aortic technique. The patient was discharged on the 10th postoperative day without any problem.
Giant aneurysm of ascending aorta, Stanford type A aortic dissection, Cerebral and systemic protect, Perfusion
The cases within the first 14 days after the onset of symptoms are considered as acute dissection [1,2]. If acute symptoms occur and there is no death within two months, these cases are considered as chronic dissections. In patients without death or rupture, aortic diameter begins to expand in days and weeks due to pressure on the aortic wall [1]. Consequently, it may cause giant chronic aortic aneurysm as in our case. The surgical procedures in ascending aortic diseases associated with dissection or aneurysm are important in terms of brain and body protection and perfusion, especially in elderly patients.
In this paper, we present procedure for successful systemic perfusion and surgical result in an elderly patient with a chronic giant aneurysm of Stanford Type A aortic dissection.
This concerns the case of an 82-years-old man who had previously been diagnosed with acute Stanford Type A aortic dissection and who refused surgical intervention. The third year following the diagnosis of aortic dissection, the patient was hospitalized because of chest pain, shortness of breath, fatigue and dizziness. A grade 4/6 diastolic murmur was heard on auscultation. Chest x-rays showed a right pleural effusion. Computed tomography demonstrated a Stanford Type A aortic dissection and dilatation of the ascending aorta (Figure 1). There was an intimal flap, enlarged patent false lumen and true lumen with mural thrombus in the ascending aorta. The lesion was approximately 10 cm in diameter - the largest ever reported - and resulted from chronic aortic dissection.
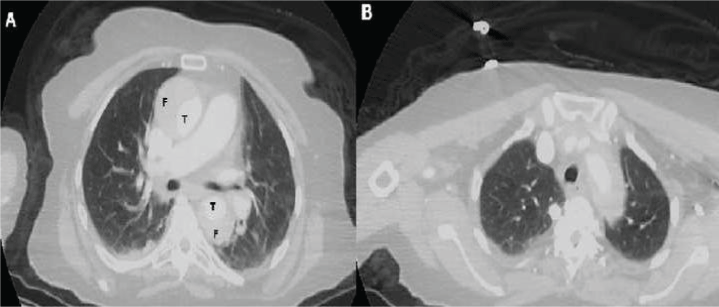 Figure 1: A) Computed sequential tomographic images of an 82-year-old man who diagnosed a chronic Stanford Type A aortic dissection. CT-scan illustrated dissection of the ascending aorta, dilatation of the ascending aorta and circulation in both the true (T) and the false (F) lumens. T denotes the true lumen and F denotes the false lumen; B) Contrast-enhanced tomography image showing aortic arch and branches in Stanford Type A dissection patient.
View Figure 1
Figure 1: A) Computed sequential tomographic images of an 82-year-old man who diagnosed a chronic Stanford Type A aortic dissection. CT-scan illustrated dissection of the ascending aorta, dilatation of the ascending aorta and circulation in both the true (T) and the false (F) lumens. T denotes the true lumen and F denotes the false lumen; B) Contrast-enhanced tomography image showing aortic arch and branches in Stanford Type A dissection patient.
View Figure 1
In transthoracic echocardiography, the patient had 2°-3° aortic insufficiency and a calcified bicuspid aortic valve. The aortic root angiography was carried out to measure the aortic gradient and condition of the coronary artery. In the angiography, advanced aortic stenosis (90 mmHg gradient), normal coronary arteries, and 2° aortic insufficiency were detected. The patient was recommended surgical treatment for advanced-stage aortic stenosis and chronic dissecting aortic aneurysm. The patient accepted surgery and operative risk, and then he taken operation, urgently. After a sternotomy, giant ascending aortic aneurysm was seen (Figure 2). Firstly the femoral artery and right atrium were prepared for a cardiopulmonary bypass following systemic heparinization (300 unit/kg). The femoral artery and two stage venous cannulas were placed. The ascending aorta was slightly pulled to the proximal and the innominate artery was made visible, and it was suspended with plastic tape. The diameter of the innominate artery was approximately 11 mm. The purse suture was placed on the innominate artery, and it was cannuled. The femoral arterial line (systemic line) was connected to the innominate arterial line (cerebral line) by the use of a Y-shaped connector, and the cardiopulmonary bypass was initiated. The vent tube was inserted into the left ventricle via the right upper pulmonary vein. Myocardial protection was provided by systemic hypothermia at 30 ℃ with antegrade administration cardioplegia solution, and then by cold retrograde blood perfusion. To start aortic surgery with open technique, the innominate artery was clamped 1 cm above the site of origin from ascending aorta, and the pump flow rate was decreased to 10 mL/kg/min. So, the antegrade cerebral and systemic perfusions were achieved with low flow with innominate and femoral arteries lines. Following this, a vertical incision was made to the ascending aortic aneurysm. The dilated ascending aorta was excised. A marked mural thrombus was present in the false lumen of the ascending aorta, but the dissection did not extend to the coronary ostium (Figure 3). A 30 mm tubular woven Dacron prosthesis (UB Shield Graft TM, Ube Medical Co. Ltd., Tokyo, Japan) was anastomosed - using 3-0 polypropylene under the open anastomosis technique - to the distal ascending aorta. The distal stump was reinforced with a strip of polytetrafluoroethylene felt on the outside of the aorta. After the clamp on the innominate artery was removed, a cross clamping was placed on the new ascending aortic graft, and the cardiopulmonary bypass was continued with a normal flow rate using both the femoral and the innominate arteries lines. Later, the aortic valve was excised and an aortic valve replacement was performed with number 21 mechanical aortic valve (Sorin Bicarbon bileaflet valve). Then, the new ascending aortic graft was sutured to the proximal aorta. That is, the procedures of a tubular ascending aortic graft and separated aortic valve replacement were completed (Figure 4). De-airing of the left heart was carried out via the aortic root catheter, followed by a de-clamping of the graft, and the patient was re-warmed. The heart spontaneously resumed beating into ventricular fibrillation, underwent cardioversion into a slow rhythm, and was then paced. About five minutes after declamping the aorta, the hemodynamics stabilized with good left ventricular contraction. Cardiopulmonary bypass was withdrawn uneventfully. The cannulas in the innominate and femoral arteries were removed. After sufficient hemostasis was achieved, the chest was closed. The hemispheric antegrade cerebral and systemic perfusion time was 35 minutes during open aortic technique. The average aortic cross-clamp time was 63 minutes. On the first postoperative day, the patient was conscious and extubated. A postoperative-enhanced CT showed no abnormal findings at the anastomotic site with the prosthesis, as well as no residual aortic dissection. The pathological diagnosis was aortic dissection without cystic medial necrosis. The patient was discharged on postoperative day 10 without any problems.
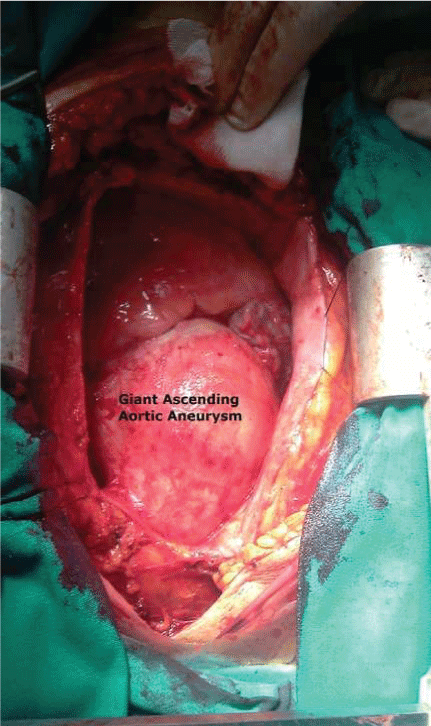 Figure 2: Intraoperative view of the chronic dissecting ascending aortic aneurysm. The ascending aorta dilated, extending 10 cm in diameter.
View Figure 2
Figure 2: Intraoperative view of the chronic dissecting ascending aortic aneurysm. The ascending aorta dilated, extending 10 cm in diameter.
View Figure 2
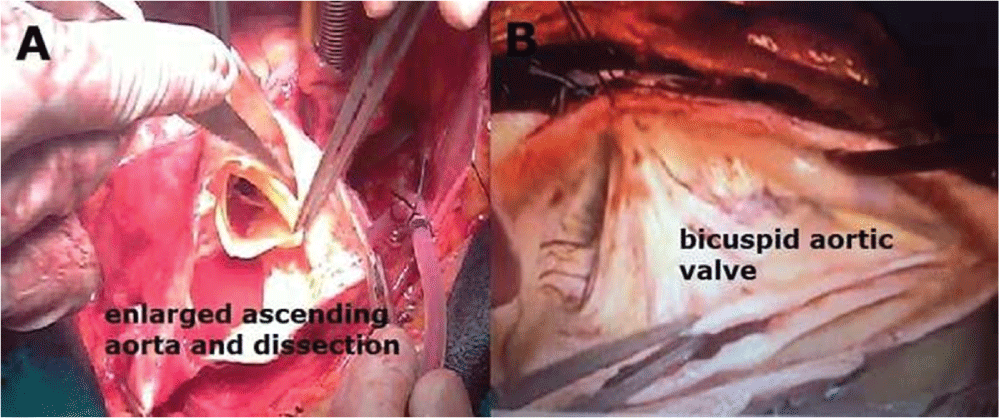 Figure 3: A) Intraoperative view after aortotomy, enlarged ascending aorta and dissected aortic tissue; B) Operative image of bicuspid aortic valve after proximal ascending aortic graft implantation.
View Figure 3
Figure 3: A) Intraoperative view after aortotomy, enlarged ascending aorta and dissected aortic tissue; B) Operative image of bicuspid aortic valve after proximal ascending aortic graft implantation.
View Figure 3
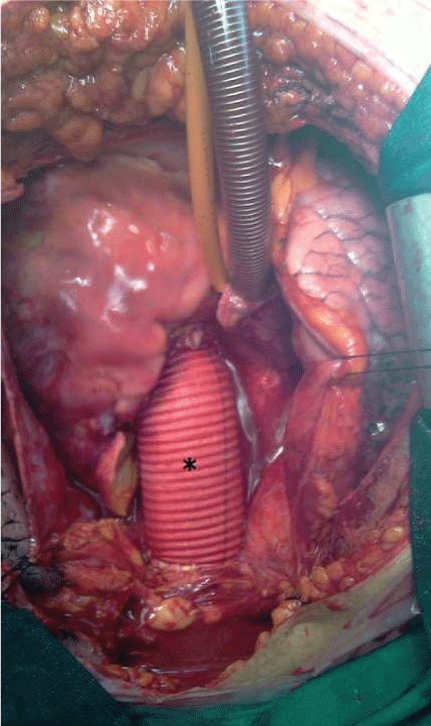 Figure 4: Postoperative view following ascending tubular graft replacement (asterisk).
View Figure 4
Figure 4: Postoperative view following ascending tubular graft replacement (asterisk).
View Figure 4
Diseases of the aorta are important contributory factors towards morbidity and mortality, and are related to cardiovascular disease. One of these diseases is aortic dissection (AD), with a prevalence of 5 to 30 cases per million people per year, and it is an exceptionally lethal condition. Almost three quarters of AD cases affect the ascending aorta and this, in the acute phase of the disease, carries a high risk of serious complications. Patients with aortic dissection may also present with acute aortic regurgitation, cardiac dysfunction, and congestive heart failure, ischemia to various organs, and neurologic deficits [1,2].
The few patients who survive the initial phase of an untreated Stanford Type A aortic dissection have an extremely high long-term risk of mortality and often have clinical findings different from those of acute dissections. Many patients with aortic dissection die before hospital admission. Mortality has been estimated at 1-2% per hour during the first two days [2,3]. Early clinical recognition is crucial for the emergent (usually surgical) management of these cases. However, in up to 38% of patients, the diagnosis is missed on initial clinical evaluation and, in more than 20% of patients, the diagnosis is only made at autopsy, and few have been reported as chronic dissections. Meticulous diagnostic imaging and urgent surgical treatment are compulsory to improve survival [1,2].
Aneurysms of the ascending aorta are generally caused by Marfan's syndrome, post-stenotic dilatation in aortic valve disease, aortic arteriosclerosis, or chronic aortic dissection. The aortic aneurysm's relation to chronic aortic dissection of the ascending aorta is one of a high rate of in-hospital mortality and poor long-term survival. The aneurysms caused by chronic aortic dissection are quite rare [2-4]. The incidence of chronic ascending aortic dissection ranges from 21% to 31% in clinical and pathological series, and these include patients with previous cardiac surgery. Our patient hadn't undergone either aortic or cardiac surgery before. Our patient was diagnosed with Stanford Type A aortic dissection three years earlier, and had rejected surgical intervention. The patient hadn't had any potentially mortal complication or rupture due to the ascending aortic dissection, and the dissected aortic segment became chronic and aneurysmatic. Besides, the patient developed aortic valve calcification and the clinical picture of advanced-stage aortic stenosis because of a bicuspid aortic valve. Bicuspid aortic valve is one of the most common congenital heart anomalies (incidence ranges from 0.9% to 2%). In the classifications made by Sievers, et al. 3 properties of the aortic valve were used (number of raphes, spatial position of cusps or raphes, and functional status of the valve) for classification. With these three structures, the bicuspid aorta is divided into 3 main types as type 0 (no raphe), type 1 (a raphe) and type 2 (two raphes) [5]. The aortic vessel pathologies that may be associated with bicuspid aorta may cause life-threatening pathologies in terms of patients. Ascending aortic aneurysms and dissections are one of them and these are pathologies with high morbidity and mortality in terms of patients. Our patient had a bicuspid aortic valve and aortic dissection developed 3 years ago.
The operative method is determined by the extent of the lesion. Bentall operations, total arch replacement, only ascending aortic replacement can be performed [3,4,6]. During surgical interventions on the ascending aorta itself, its branches or on aortic valves, cerebral protection can be achieved using techniques such as deep hypothermic circulatory arrest, retrograde cerebral perfusion, or antegrade selective cerebral perfusion, individually or in combination. In our patient, we ensured both systemic and cerebral perfusion using the open aortic technique under moderate hypothermia (30 ℃), performing direct innominate artery and femoral artery cannulation. During the open technique, the dacron graft was sutured to the distal native aorta by providing cerebral perfusion with the innominate artery line and systemic perfusion with the femoral artery line. Then, the cross clamp was positioned over the sutured graft and a cardiopulmonary bypass was continued through both innominate and femoral arterial routes. Finally, both the aortic valve replacement and a proximal graft suturation were completed with innominate and femoral lines perfusion. As there may be problems associated with cerebral and systemic functions in elderly patients, we have perfused both systems during cardiopulmonary bypass with this method. Using this method, we realized that we made the right choice because the patient woke up postoperatively and had normal systemic function.
In summary, the cardiopulmonary bypass can be achieved by using the innominate artery and femoral artery together in elderly patients whose systemic organ perfusion is thought to be insufficient. The cardiopulmonary bypass without a circulatory arrest with a moderate hypothermia will protect the patient from side effects of both deep hypothermic circulatory arrest and complications of retrograde cerebral perfusion.