Introduction: Atypical hemolytic uremic syndrome (aHUS) can be triggered by pregnancy and may lead to both irreversible and severe consequences. We herein present the case of a woman who will develop postpartum aHUS, with the consequent diagnostic and therapeutic challenges that followed.
Case report: It is the case of a pregnant woman who develops postpartum hemorrhage in addition to thrombocytopenia and acute kidney injury (AKI). She is transferred to the intensive care unit for probable pre-eclampsia with severe features or HELLP syndrome. 72 hours later, her clinical situation worsens. aHUS is therefore considered after being linked to C3-C4 hypocomplementemia. Eculizumab being unavailable in a country suffering from an economic crisis, a supportive treatment based on corticosteroids, rituximab and plasmapheresis was initiated. The patient slowly recovered both clinically and biologically; however, she still suffers from chronic kidney failure.
Conclusion: aHUS is an exclusion diagnosis. First line treatment is eculizumab but, when unavailable, the patient may improve with the proper supportive treatment.
Acute kidney injury, Coagulopathy, Hemolytic anemia, Atypical hemolytic uremic syndrome, Pregnancy
Atypical hemolytic uremic syndrome (aHUS) is most often caused by defects in the alternative pathway of the complement system or defects in the proteins regulating it. Several situations can contribute to the development of secondary aHUS, including pregnancy, malignant hypertension, kidney transplantation, certain medications (calcineurin inhibitors, antiplatelet agents, etc.), and hematopoietic stem cell transplantation [1,2]. aHUS triggered by pregnancy can lead to severe and irreversible consequences through thrombotic microangiopathy (TMA) [1]. This syndrome is characterized by a triad of intravascular hemolytic anemia, thrombocytopenia, and acute kidney injury [2,3]. The differential diagnosis of aHUS in the peripartum period is often challenging because pre-eclampsia, HELLP syndrome, and postpartum hemorrhage can all mimic aHUS. The clinical course can be severe, with a 46% risk of developing end-stage chronic kidney disease [3-5]. Treatment includes corticosteroids, plasma exchange, dialysis, and eculizumab. Eculizumab is a monoclonal antibody against C5 that inhibits complement activation [2,5]. Although women with aHUS can safely take eculizumab during pregnancy, such data is limited [1]. It is important to note that eculizumab is a medication that may be unavailable in some countries, especially in developing ones. We present the case of a woman with postpartum aHUS and the challenges faced in controlling her disease in the absence of eculizumab.
The case is about a 39-year-old woman, primipara with a history of a spontaneous miscarriage at 9 weeks of gestation for which she takes 100 mg/day of acetylsalicylic acid. Her blood pressure was labile during pregnancy, but not exceeding 130-140/80 mmHg. Her tests never showed any proteinuria. It is worth noting that the sFLT1/PlGF ratio was not performed due to the unavailability of the test. Her family history includes two cases of preeclampsia with her two sisters, one of which evolved into severe preeclampsia and lead to premature birth.
She presented to the hospital in labor at 34 weeks and 2 days of gestation. Her blood tests on admission were unremarkable except for a platelet count of 81,000/µL (previously 100,000/µL at the start of the 3 rd trimester). Labor was augmented with oxytocin after spinal anesthesia. A control blood test at 23h showed the platelet count at 83,000/µL. The patient delivered vaginally 5 hours after her admission, with the use of a vacuum extractor and a left medio-lateral episiotomy. The placenta was delivered manually. The newborn had an APGAR score of 10 at 1 minute and 10 at 5 minutes, with a birth weight of 2400g. Uterine revision was performed, and a single dose of amoxicillin with clavulanic acid was administered. The uterus was well-contracted, and bleeding was within the normal range. At 3 hours postpartum, vaginal bleeding worsened despite a well-contracted uterus, estimated at 600 cc. Two misoprostol tablets were administered intrarectally, and one sublingually, with moderate improvement. At 9 hours postpartum, the patient developed a fever of 39 °C and fatigue. Amoxicillin with clavulanic acid was systematically administered at 1.2 g/8h in addition to tranexamic acid at 1 g/8h. Blood tests revealed the following, with “N” being the normal range:
- Hemoglobin 10.6 g/dL (12 < N < 16)
- White blood cells 15100/µL (4000 < N < 11000) with neutrophils at 84% and lymphocytes at 8.7%
- Platelets 46 000/µL (150000 < N < 450000)
Hemolysis panel:
- Reticulocytes 4% (0.5 < N < 2.5)
- LDH 1665 U/L (105 < N < 333)
- Haptoglobin < 0.01 g/L (0.5 < N < 2.2)
- CPK 767 U/L (26 < N < 192)
- Negative direct Coombs testing
- Rare schizocytes
Liver panel:
- SGOT 114.6 U/L (9 < N < 32)
- SGPT 15.7 U/L (7 < N < 45)
Coagulation studies:
- PT 13.3/11.9
- PTT 35.1/33.6
- Fibrinogen 2.19 g/L (2 < N < 4)
- Urine dip stick: proteins 2+
At 16 hours of postpartum, a follow-up test showed worsening anemia, severe thrombocytopenia, and acute renal failure with a creatinine level of 2.42 mg/dL. The patient was thus transferred to the intensive care unit with a preliminary diagnosis of preeclampsia with severe features and an estimated total blood loss of 1000 cc. Her vital signs were stable. Clinical examination showed clear cardiopulmonary auscultation, normal neurological examination, and a soft abdomen upon palpation. The initial care plan included appropriate fluid resuscitation with 1 L/24h of normal saline and 1 L/24h of lactated Ringer's solution. A 4g bolus of magnesium sulfate was administered, followed by a maintenance dose of 2 g/24h. Antibiotic therapy was continued, and the patient received two units of platelets.
On the 2 nd day of postpartum, the patient developed oliguria and a worsening renal function. Cardiac ultrasound showed preserved systolic and diastolic function. Follow-up blood tests indicated persistent hemolytic anemia, thrombocytopenia, and liver function abnormalities.
On the 3 rd day of postpartum, the patient's condition deteriorated further. A diagnosis of atypical hemolytic uremic syndrome (aHUS) was suspected. Blood tests revealed hypocomplementemia (low C3 and C4 levels), and additional tests for anti-ADAMTS13 antibodies and ADAMTS13 activity were conducted. Anti-ADAMTS13 antibodies were negative, and ADAMTS13 activity was normal, but antigen levels were low, confirming a diagnosis of aHUS.
The patient gradually improved with the ongoing treatment regimen, including rituximab, plasma exchanges (stopped after 12 sessions), corticosteroids, hydration, red blood cell and platelet transfusions and fresh frozen plasma transfusion as needed.
On the 18 th day postpartum, the patient was transferred back to the maternity ward while still receiving rituximab and corticosteroids. Urine protein electrophoresis did not show any suspicious bands.
On the 20 th day postpartum, a kidney biopsy confirmed renal damage due to atypical hemolytic uremic syndrome complicating a preeclampsia, further corroborating the aHUS diagnosis. The patient was discharged with a creatinine level of 1.61 mg/dL, hemoglobin of 9.7 g/dL, and platelets of 211,000/µL. Her home treatment included prednisone, bisoprolol, losartan, and blood pressure monitoring.
A follow-up at three months postpartum showed normalization of most parameters in her blood tests, except for persistently elevated creatinine (0.97 mg/dL).
Figure 1, Figure 2, Figure 3 and Figure 4 found at the end of the manuscript, illustrate the respective evolutions of acute kidney injury, hemolysis and thrombocytopenia.
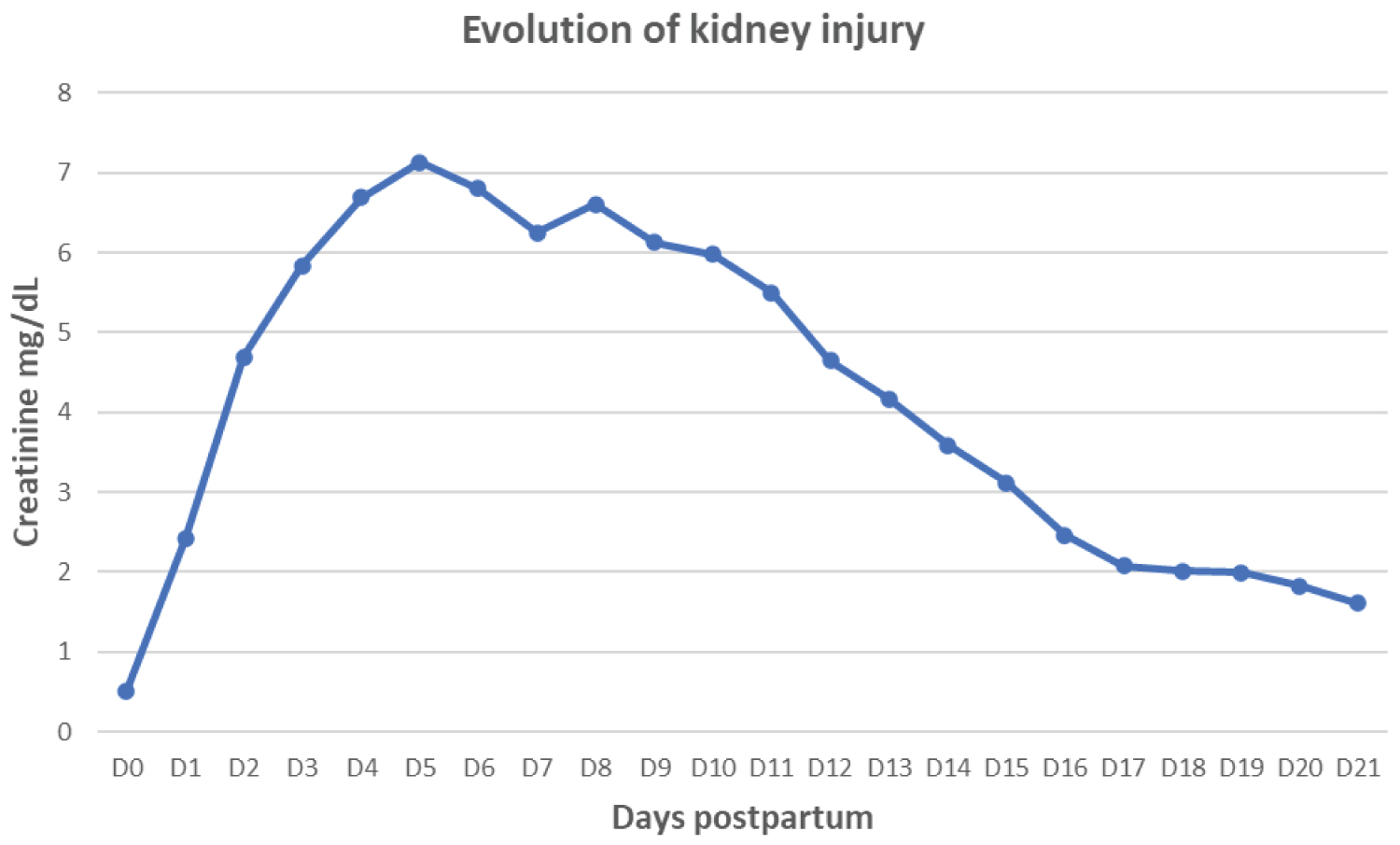 Figure 1: Evolution of kidney injury illustrated by creatinine.
View Figure 1
Figure 1: Evolution of kidney injury illustrated by creatinine.
View Figure 1
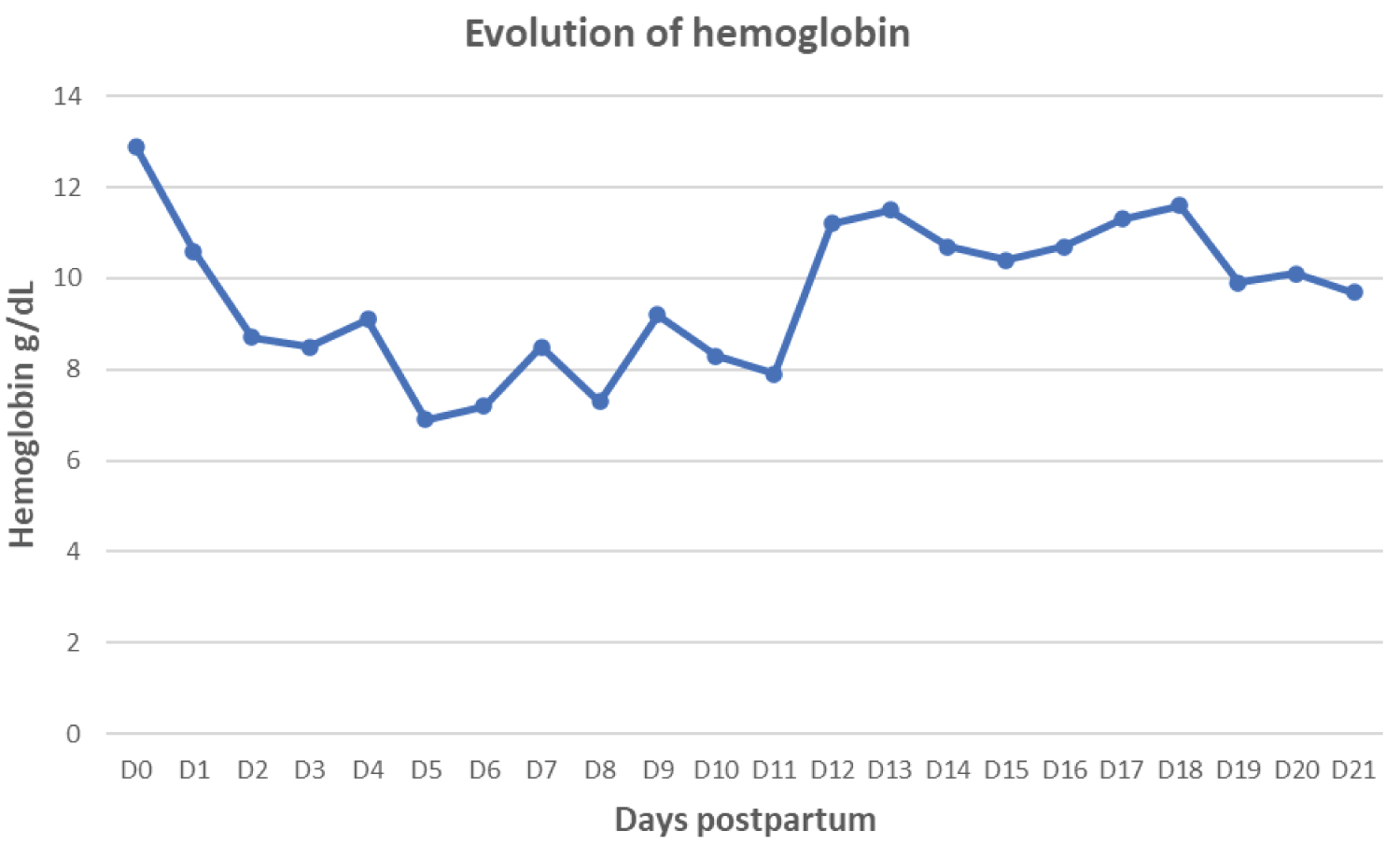 Figure 2: Evolution of hemolysis illustrated by hemoglobin.
View Figure 2
Figure 2: Evolution of hemolysis illustrated by hemoglobin.
View Figure 2
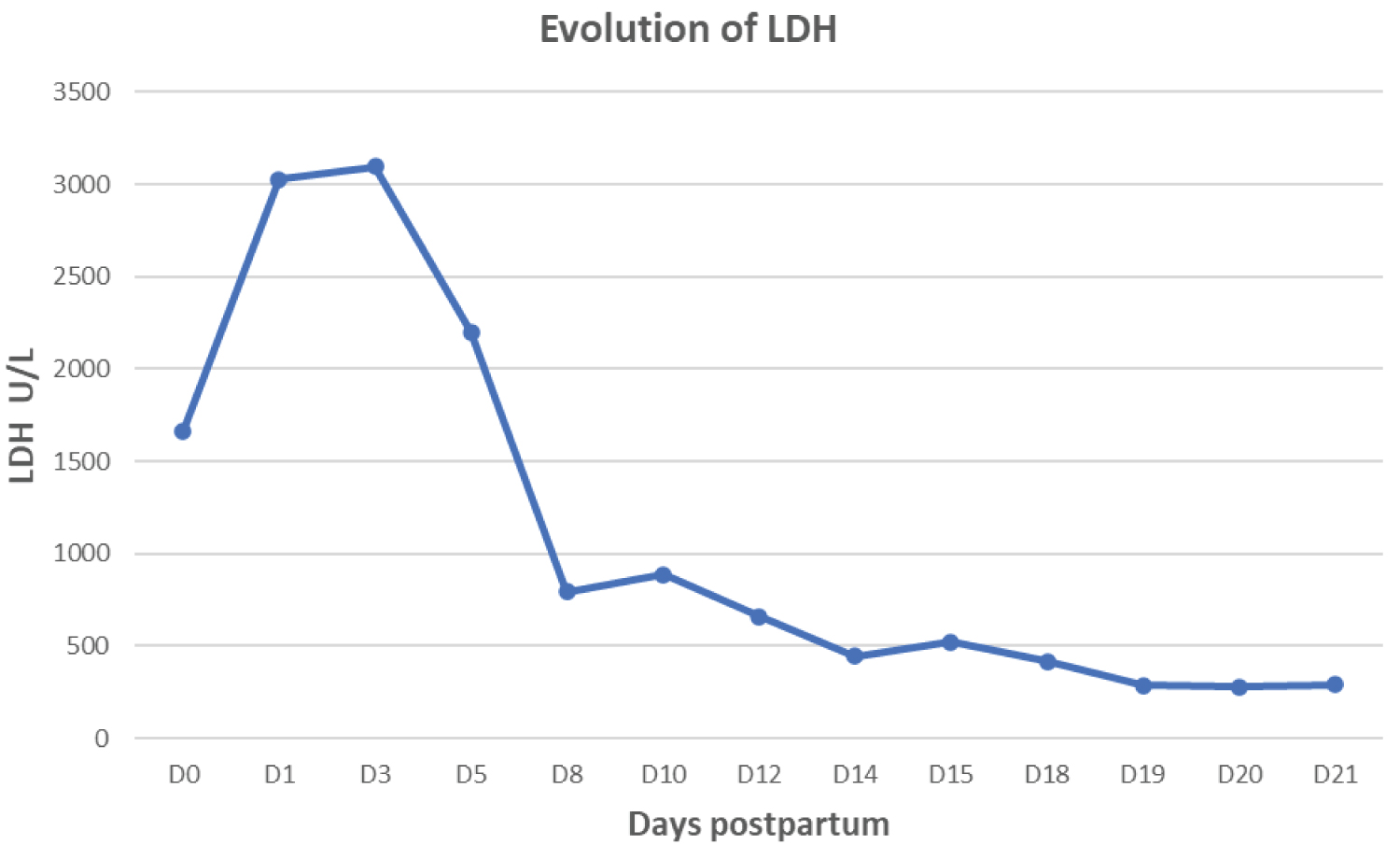 Figure 3: Evolution of hemolysis illustrated by LDH.
View Figure 3
Figure 3: Evolution of hemolysis illustrated by LDH.
View Figure 3
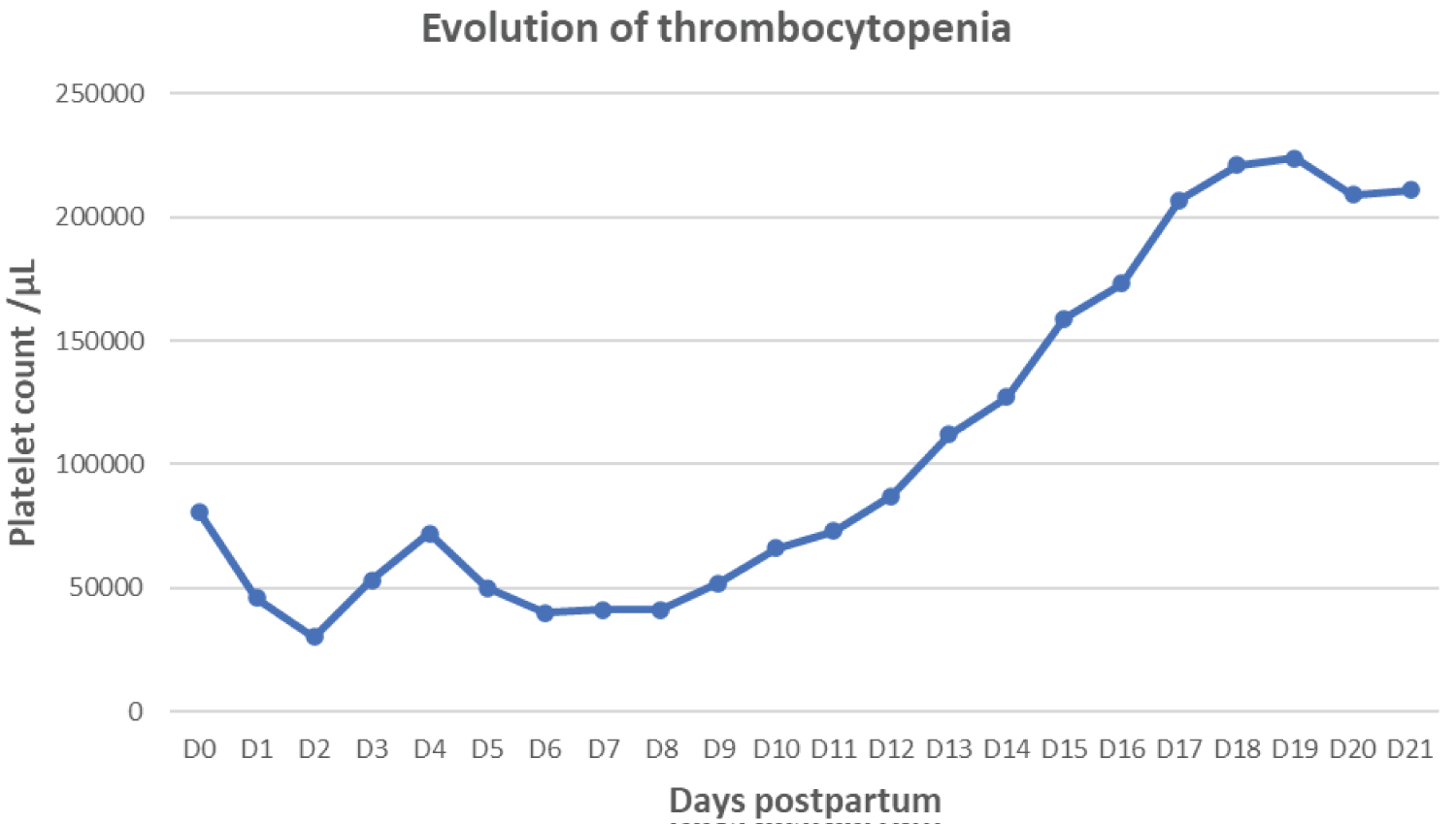 Figure 4: Evolution of thrombocytopenia illustrated by platelet count.
View Figure 4
Figure 4: Evolution of thrombocytopenia illustrated by platelet count.
View Figure 4
The patient experienced three simultaneous complications in addition to her postpartum hemorrhage: Severe acute renal failure, thrombocytopenia, and microangiopathic hemolytic anemia. The differential diagnosis initially favored HELLP syndrome due to the presence of microangiopathic hemolytic anemia, low platelet count, elevated SGOT/SGPT ratio and elevated creatinine. However, the absence of epigastric and right upper quadrant abdominal pain, the absence of neurological symptoms and the lack of improvement within 72 hours of postpartum weigh against HELLP syndrome. The diagnosis of acute fatty liver of pregnancy (AFLP) was considered due to an elevated SGOT/SGPT ratio and elevated creatinine, but a platelet counts as low as 33,000/µL, severe hemolytic anemia, and the absence of jaundice, hypoglycemia, and significant coagulopathy made this diagnosis unlikely. The diagnosis of thrombotic thrombocytopenic purpura (TTP) was initially suspected due to severe thrombocytopenia and fever, but the normal ADAMTS13 activity and other clinical findings did not support this diagnosis. Given the absence of initial digestive symptoms such as bloody diarrhea, typical hemolytic uremic syndrome (HUS) was not initially considered. Eventually, the patient's diagnosis of atypical hemolytic uremic syndrome (aHUS) was determined through exclusion. The postpartum hemorrhage, severe thrombocytopenia, severe acute renal failure, microangiopathic hemolytic anemia and hypocomplementemia (low C3 and C4) all favored the diagnosis. Another crucial factor was the persistence of symptoms beyond 72 hours of postpartum. Eculizumab is the first-line treatment for aHUS, but it was unavailable in this case due to the economic crisis of the country. The patient's improvement with plasma exchange, despite a longer recovery time, underscores the importance of prompt diagnosis and treatment in aHUS. This case also highlights the potential for long-term complications, such as chronic renal failure, in patients with aHUS who do not receive eculizumab.
Atypical hemolytic-uremic syndrome (aHUS) is an exclusion diagnosis to consider when facing postpartum hemorrhage with microangiopathic hemolytic anemia, thrombocytopenia, and severe acute renal failure. Another supporting factor would be a clinical worsening after 72 hours of postpartum. The first-line treatment is eculizumab, but in case of unavailability, supportive treatment with plasma exchanges and corticosteroids can lead to remission, albeit at a slower pace and with a risk of sequelae. Pregnancy in women with a history of aHUS is a high-risk pregnancy. The management and decisions regarding a future pregnancy should involve a multidisciplinary team including an obstetrician, nephrologist, and hematologist.
The author and all co-authors declare no conflicts of interest.
The four authors have equally contributed to the writing of this article.