Introduction: Volume overload, whether overt or sub-clinical, is associated with increased cardiovascular mortality in chronic haemodialysis patients. The assessment of dry weight remains a challenge and relies on clinical and paraclinical methods. Pulmonary ultrasound could thus prove useful as an additional tool in improving the management of haemodialysis patients, possibly in addition to bioimpedance metry data.
Method: To assess the interest and feasibility of lung ultrasound in the evaluation of dry weight overload in comparison with clinical examination in hemodialysis patients. We conducted a study in our dialysis unit at the MOHAMMED V military training hospital. The patients were all dialysed on a NIKKISSO machine. Each patient had a pulmonary ultrasound 30 minutes after the HD session, with measurement by B-line counting on 8 zones. Cardiac ultrasound and chest X-ray were performed to assess volume overload. Clinical parameters scheduled and performed UF were also recorded.
Results: Between March and May 2022, 37 patients were included with a mean age of 57.4 ± 16.7 years [26; 83] and a female predominance of 54.1%. Subclinical overload was detected on lung ultrasound in 45.9% versus 18.9% on clinical examination. A significant correlation between the Kerley B line and pre-dialytic hypertension and overload was found, with a correlation coefficient of 0.038 (p < 0.05).
No significant correlation was found between B-lines and clinical examination (oedema, NYHA stage, crepitus).
Conclusion: Pulmonary ultrasound appears to be easy to use in routine and effective for dry weight assessment, in addition to other assessment methods.
Dry weight, Lung ultrasound, Kerley line B, Morbi-mortality, Chronic haemodialysis
Hyperhydration in haemodialysis patients is an important morbidity and mortality factor. Dry weight is defined as the lowest tolerated post-dialysis weight corresponding to a state of euvolaemia. It is traditionally determined in haemodialysis by clinical examination data based on the search for signs of hypervolemia (oedema, hypertension, and symptoms of left heart failure) [1]. However, such an empirical approach rarely avoids the problems of hypertension, intra-dialytic hypotension and sub-clinical hyperhydration.
For these reasons, other additional means are used, including bioimpedance methods, echocardiography, pro-BNP measurement and the introduction of lung ultrasound as a complementary tool in the assessment of volume overload in chronic haemodialysis patients [1,2].
Lung ultrasound is a new, reliable, easy-to-learn, reproducible and non-invasive approach to objectively quantify the presence of extravascular lung water at a sub-clinical stage and has led to progress in this field. Impaired left ventricular function and increased pulmonary capillary permeability are key factors in the genesis of subclinical pulmonary congestion and are positively correlated with Kerley B-lines. The B-line is the basic symptom of water in the pulmonary alveoli [3].
The aim of this work is to clarify the contribution and feasibility of lung ultrasound in the assessment of dry weight overload in comparison with clinical examination in haemodialysis patients.
This is a descriptive monocentric cross-sectional study conducted in the department of nephrology, dialysis, and renal transplantation at the Mohammed V military hospital of Rabat. We included patients with chronic end-stage renal failure undergoing periodic hemodialysis at a rate of 03 sessions per week for more than 03 months and who had undergone pulmonary ultrasound. We collected 37 patients dialyzed in the hemodialysis centre of the hospital during the study period from March 1 to May 1, 2022.
Lung ultrasound was performed in the supine position at the patient's bedside, 30 minutes after each dialysis session and in a quiet room at room temperature, by 2 different experienced operators.
We used the LOGIQ P6 Pro GE healthcare ultrasound machine with a convex probe from 3 to 9 MHz.
The estimation of the fluid status after the haemodialysis session is done by means of the lung ultrasound. Pulmonary congestion by fluid overload was retained in view of the presence of several KerleyB-lines. These are defined as vertical bands of hyperechoic comet-tail artefacts that originate at the pleural line and cross the entire surface of the lung, the pleural line and vertically cross the entire ultrasound screen to the bottom of the screen and are considered to be the sonographic sign of interstitial oedema [4]. Pulmonary congestion due to fluid overload was retained when multiple B-lines were present (number > 2) and we adopted the 8-zone method. This method takes into account the scanning of 2 anterior and 2 lateral zones in each hemithorax located and bordered by the virtual para-sternal line, the mid-clavicular and mid-axillary line of each hemithorax [5].
The majority of authors have adopted the 28-zone method. This method is easy to perform, but it is not very reproducible in daily clinical practice.
In our work, we have opted for the use of the 8-zone method of analysis, which saves time and is less cumbersome, making it more suitable for routine clinical use.
Clinical, dialysis, biological and radiological data of the patients were collected from the hospital records:
Clinical data: Age, sex, associated pathologies and treatment, initial nephropathy as well as the length of time on dialysis, blood pressure, clinical signs of hydrosodic overload (oedema of the lower limbs (OMI), crepitus rales).
Dialysis data: Vascular approach, dialysis dose, dialysis modality (HDF or HDI), PPID, dry weight, scheduled ultrafiltration, achieved ultrafiltration and the difference.
Biological parameters: Blood urea, creatinine, blood ionogram, haemoglobin and albumin.
Radiological parameters: Trans-thoracic ultrasound (TTE), chest X-ray.
Lung ultrasound data: Look for the presence and number of Kerly's B lines and the number of zones.
All data were entered and analysed using EXCEL 2010 for WINDOWS and SPSS version 20 data processing software.
The statistical analysis used descriptive analysis of quantitative variables expressed as mean ± standard deviation. Qualitative variables were expressed as frequency and percentage.
An overall analysis of the risk factors for overload was performed by logistic regression. A p-value of less than 0.05 was retained as significant.
The main demographic, clinical and radiological characteristics: Table 1 and Table 2.
Table 1: The main demographic, clinical and radiological characteristics. View Table 1
Table 2: Statistical analysis of anamnestic, clinical and para-clinical characteristics. View Table 2
A total of 37 cases were included with a mean age of 57.4 ± 16.7 years (extremes 26 and 83 years). An average age of 60 years was found in the studies. In our series we noted a predominance of the female sex with a frequency of 54.1%. This female predominance was inconsistent with the data reported in the literature [6,7].
The assessment of hydration status in chronic haemodialysis patients remains a real challenge despite the multitude of available methods, notably bioimpedance metry and cardiac ultrasound.
Pulmonary ultrasound remains an easy to reproduce technique that allows the objectification of pulmonary congestion by visualising B-lines at an early and asymptomatic stage [8].
The table below shows us the added value of lung ultrasound in terms of lung congestion compared to clinical examination.
Like Torino C, et al. [9], we detected lung congestion on lung ultrasound at a sub-clinical stage (Table 3).
Table 3: Comparative study of the contribution of lung ultrasound and clinical examination. View Table 3
Our work confirms the data of the literature on the preponderant contribution of the pulmonary US in the assessment of the overload in comparison with the data of the clinical examination [10,11].
The majority of authors often criticise the clinical method for its imprecision, lack of sensitivity and objectivity, whereas pulmonary echo allows an earlier and more objective diagnosis of pulmonary interstitial oedema [9,12,13]. The combination of crepitus and peripheral oedema does not show an improvement in the diagnosis of pulmonary congestion.
Nevertheless, lung ultrasound remains an additional tool in the assessment of overload in the subclinical stage. Due to its ability to accurately estimate lung congestion by measuring extravascular lung liquid, this modality is increasingly used in renal failure patients.
Due to its non-invasive, non-irradiating nature, ease of use, availability of portable devices and acceptable intra- and inter-operator reproducibility, this technique can be considered one of the most interesting offered to the nephrologist in the assessment of extravascular lung liquid during end-stage renal disease [12]. The majority of authors have adopted the 28-zone method. Although easy to perform, this method is not very reproducible in daily clinical practice.
In our work, we have opted for the use of the 8-zone method of analysis, which saves time and is less cumbersome, making it more suitable for routine clinical use (Figure 1).
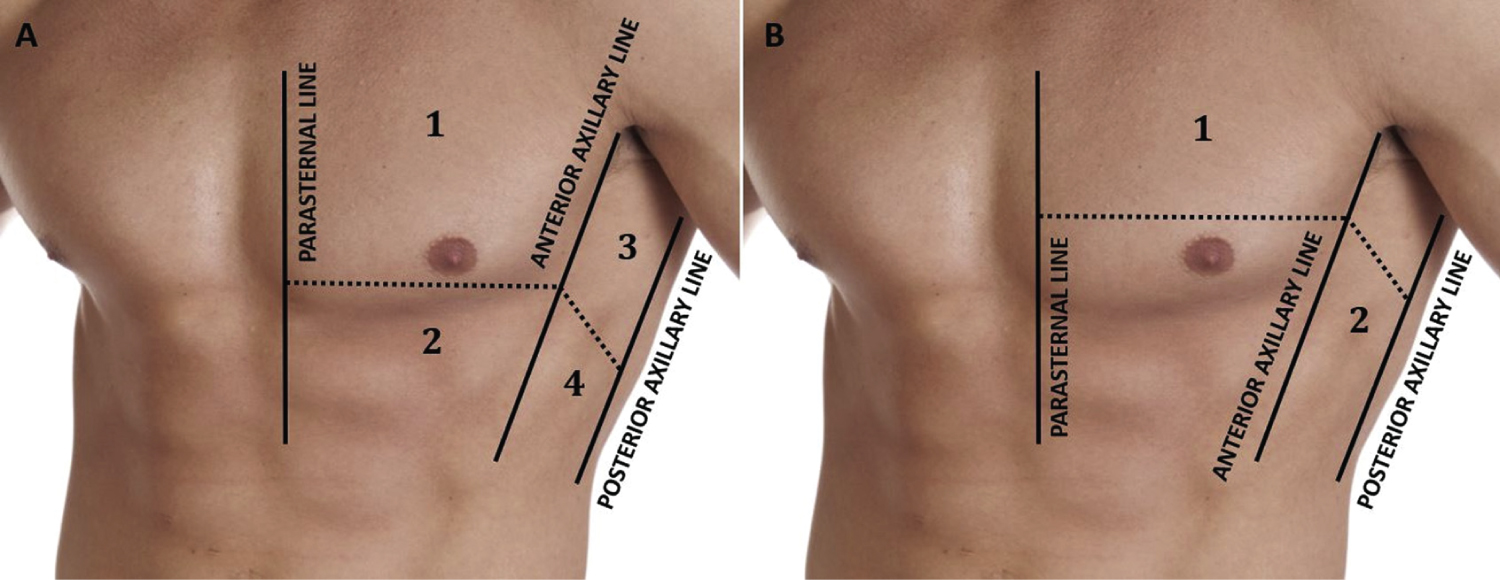 Figure 1: The 8 zones of analysis on lung ultrasound identified by the parasternal line, the mid-clavicular line and the posterior axillary line.
View Figure 1
Figure 1: The 8 zones of analysis on lung ultrasound identified by the parasternal line, the mid-clavicular line and the posterior axillary line.
View Figure 1
In patients with chronic renal failure on haemodialysis, the eight-zone method showed good agreement with the standard 28-zone method in terms of lung congestion and the two approaches have a similar prognostic value in this population.
The 8-site approach is faster and can be applied in most circumstances to detect pulmonary congestion in this population [5,14].
Therefore, we can assume that irrespective of the type of extrarenal replacement therapy considered (haemodialysis or peritoneal), subclinical extravascular lung water is often present. Its clinically "silent" nature may have a negative impact on the quality of life and survival of patients with end-stage renal disease [13].
Regarding the contribution of clinical parameters in the assessment of fluid overload, upper limb edema was found in 13.5% in our series. The study by Ishwarya, et al. [15] found a frequency of 37%. This difference can be explained by the lower number of cases in our series. Indeed, the presence of lower limb oedema was conspicuously absent in 87 and 80% of the evaluations where lung ultrasound indicated moderate and severe pulmonary congestion respectively [9,13]. The correlation between peripheral oedema and the number of B-lines was even lower.
In our study, crackles were observed in 18.9%, close to the result reported in the study by Ishwarya, et al. [15]. The hypothesis that the presence of crackling rales was significantly associated with the diagnosis of overload for the diagnosis of pulmonary congestion has recently been specifically evaluated in the dialysis population.
The investigators of the LUST (Lung Water by Ultra-Sound Guided Treatment to Prevent Death and Cardiovascular Complications in High Risk ESRD Patients with Cardiomyopathy Trial) adopted lung ultrasound as the gold standard tool to examine the diagnostic reliability of crackles as a sign of pulmonary congestion. It was found that 61% of patients with moderate or severe pulmonary congestion by lung ultrasound (B-line score ≥ 15developerSCORE) did not have crackles [9,13,15].
Similarly, the absence of edema, despite marked pulmonary congestion, has been reported in ultrasound studies of haemodialysis patients [11].
We found that routinely assessed clinical signs and symptoms were quite insensitive in detecting interstitial pulmonary oedema, as evidenced by their low sensitivity. The combination of crepitus and peripheral oedema did not show any improvement in the diagnosis of pulmonary.
The absence of oedema and crepitus, which are considered to be one of the end points of fluid removal during dialysis, was not present in almost one third of our patients with significant pulmonary congestion on lung ultrasound. These results highlight the errors inherent in estimating dry weight on the basis of clinical examination alone. Pulmonary ultrasound can identify interstitial oedema at a much earlier stage of lung congestion, which is largely consistent with the literature.
In our work, pre-dialytic hypertension was significantly associated with echo graphic overload (p = 0.038) in contrast to other studies where the authors [16,17] admitted that the reversal of excess volume is mainly attributed not to the decrease in blood pressure per se but to the reduction in extra cellular fluid volume, as the reduction in B-lines on pulmonary ultrasound was independently associated with the reduction in LV filling pressures. Thus, reversal of excess volume may reduce left ventricular preload and consequently decrease right atrial preload which would in turn lead to a decrease in right atrial afterload and in the long term may reverse chronic and widespread chamber enlargement.
In our series, 03 patients presented with clinical global cardiac decompensation (peripheral edemas, jugular venous hypertension, cardiomegaly and hilar overload) significantly correlated with pulmonary echofindings (p = 0.005). Pulmonary echo is a method with a very short learning curve and can be performed with standard equipment, providing good reproducibility with good agreement.
It is important to note that the time to dialysis is highly dependent on left ventricular function according to a study of ESRD patients with NYHA stage III to IV heart failure [18].
The close and inverse association between pulmonary congestion and ejection fraction before and after dialysis clearly indicates that left ventricular dysfunction is a major factor in pulmonary congestion. In our study, we do not have left ventricular dysfunction. This study provides evidence that chest ultrasound detects pulmonary congestion at a preclinical stage in most patients.
Indeed, 57% of asymptomatic dialysis patients had moderate to severe congestion. The detection of preclinical pulmonary oedema in dialysis patients may be an important clue to prevent decompensated heart failure [18].
Our study has several limitations due to its small sample size and monocentric nature. Secondly, the majority of patients did not have significant Kerley B lines.
Pulmonary ultrasound is a simple bedside examination for the assessment of dry weight in haemodialysis patients. It can detect lung overload at a sub-clinical stage.
The authors declare that they have no conflicts of interest.