Hemangiopericytoma is an uncommon vascular tumor derived from the pericytes of Zimmermann, representing only a small proportion of head and neck tumors, and mostly occurring in the sinonasal tract.
We report two cases of hemangiopericytoma of the sinonasal tract in a 78-year-old woman and a 77-year-old man, both presenting with symptoms of frequent epistaxis. In Case 1, a purple tumor was identified originating from the posterior olfactory fissure in the left nasal cavity. To reduce bleeding from the tumor, the anterior and posterior ethmoidal arteries were cauterized via a transconjunctival incision approach prior to tumor removal. The tumor in Case 2 was arising from the posterior region of the right nasal septum. In Case 2, intensive bleeding was seen during biopsy. To reduce the bleeding and surgical time for the subsequent operation, preoperative angiography and embolization were performed. We removed the tumors en bloc in both cases by endoscopic operation.
In these cases, postoperative histopathological and immunohistochemical examinations confirmed sinonasal hemangiopericytoma. Endoscopic and radiological follow-ups showed no recurrences within 5 years.
Sinonasal hemangiopericytomas were diagnosed histologically. Immunohistochemical analyses were helpful for tumor diagnosis. Herein we discuss the diagnosis and treatment of hemangiopericytoma.
Sinonasal hemangiopericytoma, Epistaxis, Immunohistochemical examination
Hemangiopericytomas are unusual vascular tumors that were first described by Stout and Murray in 1942 [1]. These tumors originate from extravascular cells called pericytes, which presumably represent modified contractile smooth muscle cells found on the external surface of capillaries and post-capillary venules.
Approximately 15-30% of hemangiopericytomas occur in the head and neck. Of these, approximately 5% arise from the nasal cavity. Sinonasal hemangiopericytomas are believed to behave less aggressively than hemangiopericytomas that occur in other parts of the body.
Hemangiopericytomas are relatively radioresistant and show a major tendency to bleed profusely, so wide surgical resection under an open approach and preoperative embolization are the main management options for large sinonasal tumors. Endoscopic resection of sinonasal hemangiopericytoma is a novel method with special advantages. We report two cases of hemangiopericytoma of the sinonasal tract in a 78-year-old woman and a 77-year-old man, both of whom presented with symptoms of frequent epistaxis.
A tumor in the left nasal cavity was detected in a 78-year-old woman on gastro fiberscopy at an internal medicine clinic. She was referred to our hospital because of frequent epistaxis after the examination. Nasal endoscopy showed a purple tumor occupying the left nasal cavity. The base of the tumor was seen originating from the posterior olfactory fissure (Figure 1a). Imaging modalities including computed tomography (CT) and magnetic resonance imaging (MRI) showed a hypervascular tumor originating from the posterior olfactory fissure in the left cavity and right sphenoid sinusitis, with no infiltration into surrounding structures (Figure1b, Figure 1c and Figure 1d). Preoperative biopsy was not performed, as biopsy of the tumor was considered to carry a high risk of excessive bleeding. We therefore decided to dissect the tumor and identify the specimen on pathological examination. Preoperative vessel embolization was not performed because the base of the tumor was limited and the feeder vessel of the tumor was thought to be a branch of the internal jugular artery. However, to reduce bleeding from the tumor, the anterior ethmoidal and posterior ethmoidal arteries were cauterized via a transconjunctival incision approach prior to tumor removal (Figure 2a). As a result, the tumor was completely removed endoscopically while suppressing the amount of bleeding (Figure 2b, Figure 2c and Figure 2d).
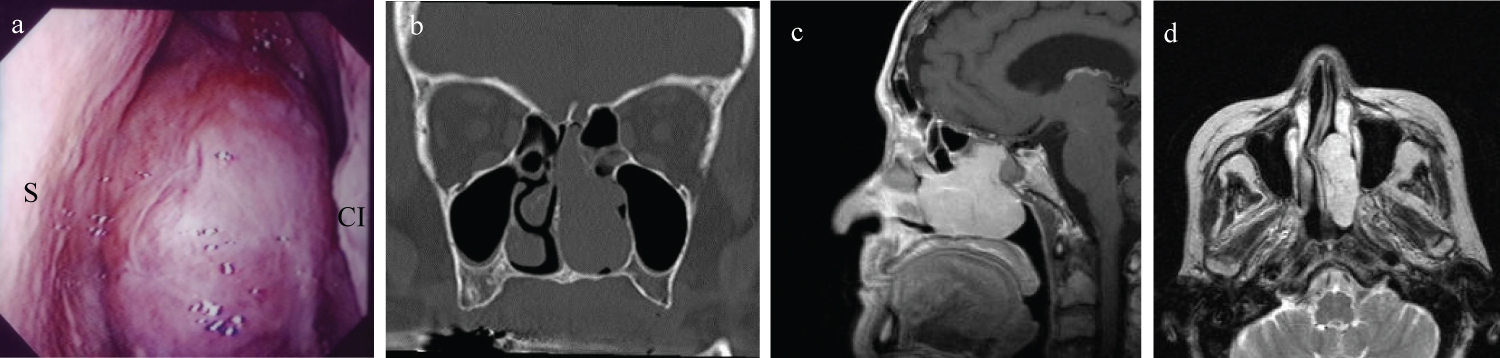 Figure 1: Preoperative findings.
Figure 1: Preoperative findings.
a) Endoscopic findings show a purple tumor occupying the left nasal cavity. The surface of the tumor appears highly vascular. The base of the tumor is seen originating from the posterior olfactory fissure. S: Nasal septum; CI: Concha nasalis inferior; b) Plain computed tomography (CT) of the paranasalsinuses reveals a mass occupying almost the entire left nasal cavity. No bony destruction is evident; c) Sagittal T2-weighted MRI shows the base of the tumor localized and originating from the posterior olfactory fissure; d) Contrast-enhanced T2-weighted MRI shows a uniformly strong contrast-enhanced mass in the left nasal cavity. A vascular tumor is suspected.
View Figure 1
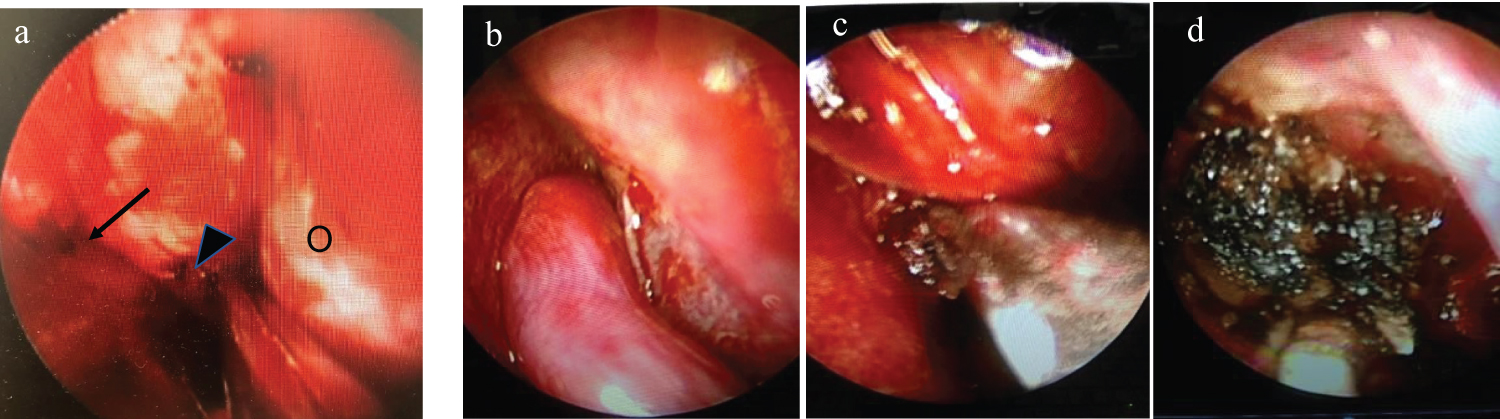 Figure 2: Intraoperative findings.
Figure 2: Intraoperative findings.
a) Prior to tumor removal, a transconjunctival incision is made to ablate the anterior ethmoidal artery (arrow) and posterior ethmoidal artery (arrowhead). O: Orbital side; b) The tumor has a base in the posterior olfactory fissure direction; c,d) The tumor is resected with an electric scalpel while the assistant removes bleeding with a suction curette.
View Figure 2
Histopathological examination revealed sinonasal hemangiopericytoma (Figure 3 and Figure 4). The patient had an uneventful recovery and no recurrence has been seen as of 5years postoperatively.
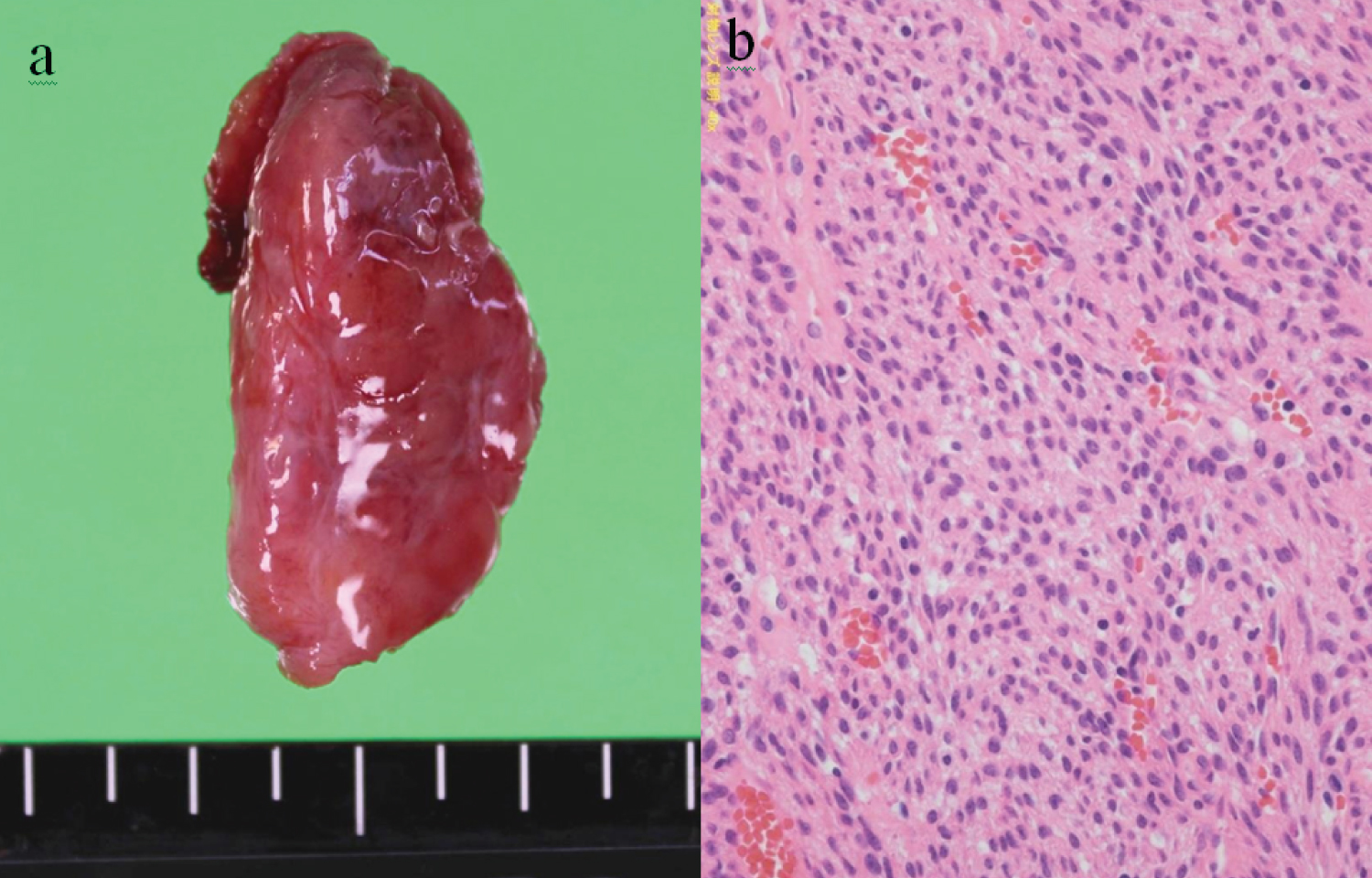 Figure 3: Histochemical staining.
Figure 3: Histochemical staining.
a) Excised tumor specimen; b) Spindle-shaped tumor cells with rounded nuclei are seen growing in sheets and whorls around vascular channels. Poor cellular atypia and no mitotic figures in tumor cells (HE, ×100).
View Figure 3
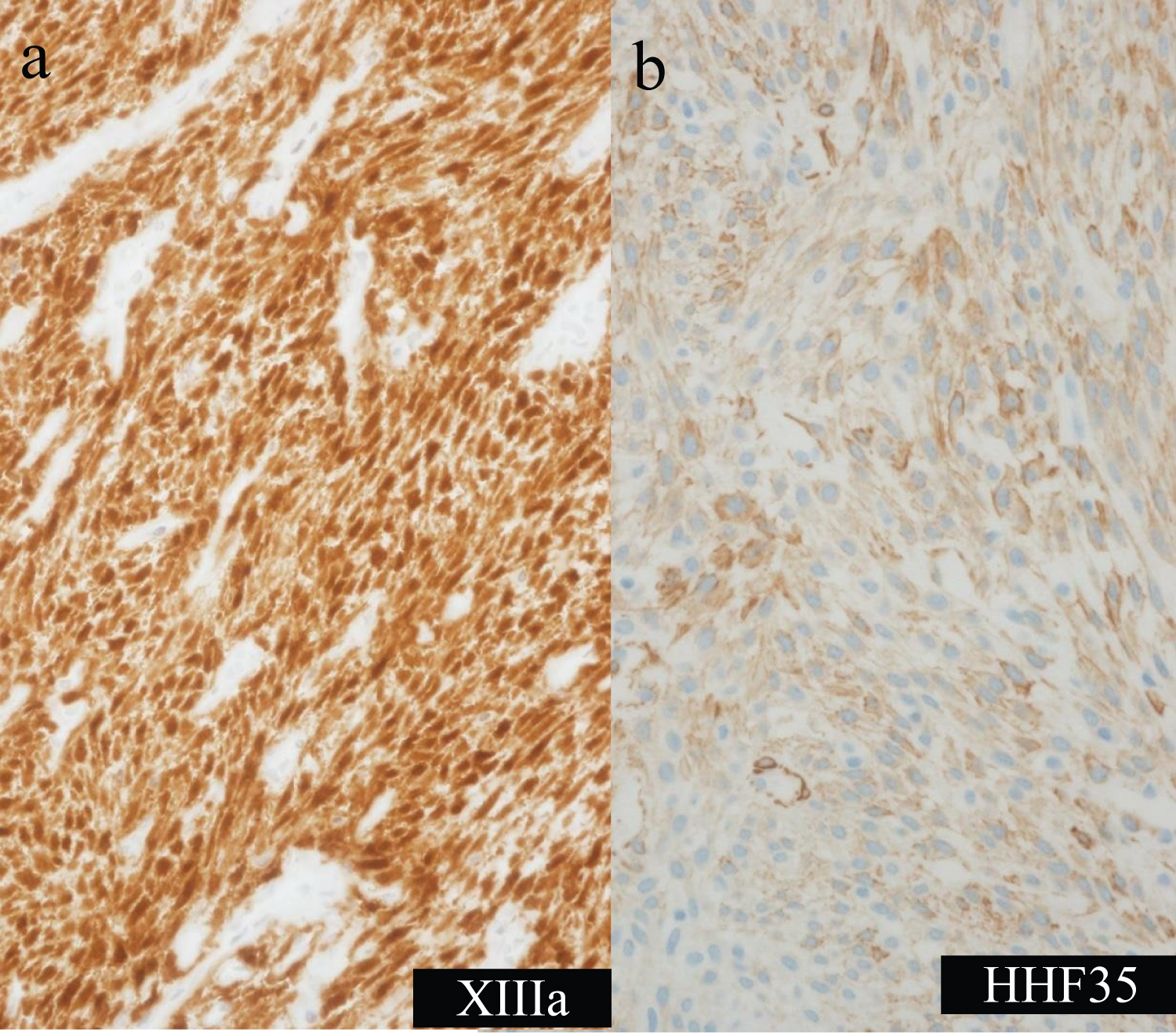 Figure 4: Immunohistochemical staining.
Figure 4: Immunohistochemical staining.
Tumor cells appear strongly positive for factor XIIIA (a) and mildly positive for HHF35 (b).
View Figure 4
A 77-year-old man was referred to our hospital because of frequent epistaxis. Nasal endoscopy showed a reddish tumor arising from the posterior region of the right nasal septum (Figure 5a). CT revealed a well-marginated soft tissue mass in the posterior region of the olfactory cleft (Figure 5b). During biopsy, intensive bleeding was seen. Histological examination of the tumor identified hemangiopericytoma. To reduce bleeding during the subsequent operation, preoperative embolization of the tumor was carried out (Figure 6), and transnasal endoscopic surgery was performed with complete tumor resection the next day. The amount of bleeding at the time of tumor removal was also very small (volume of blood loss is 10 cc). As of 5years postoperatively, the patient remains asymptomatic and has shown no signs of recurrence.
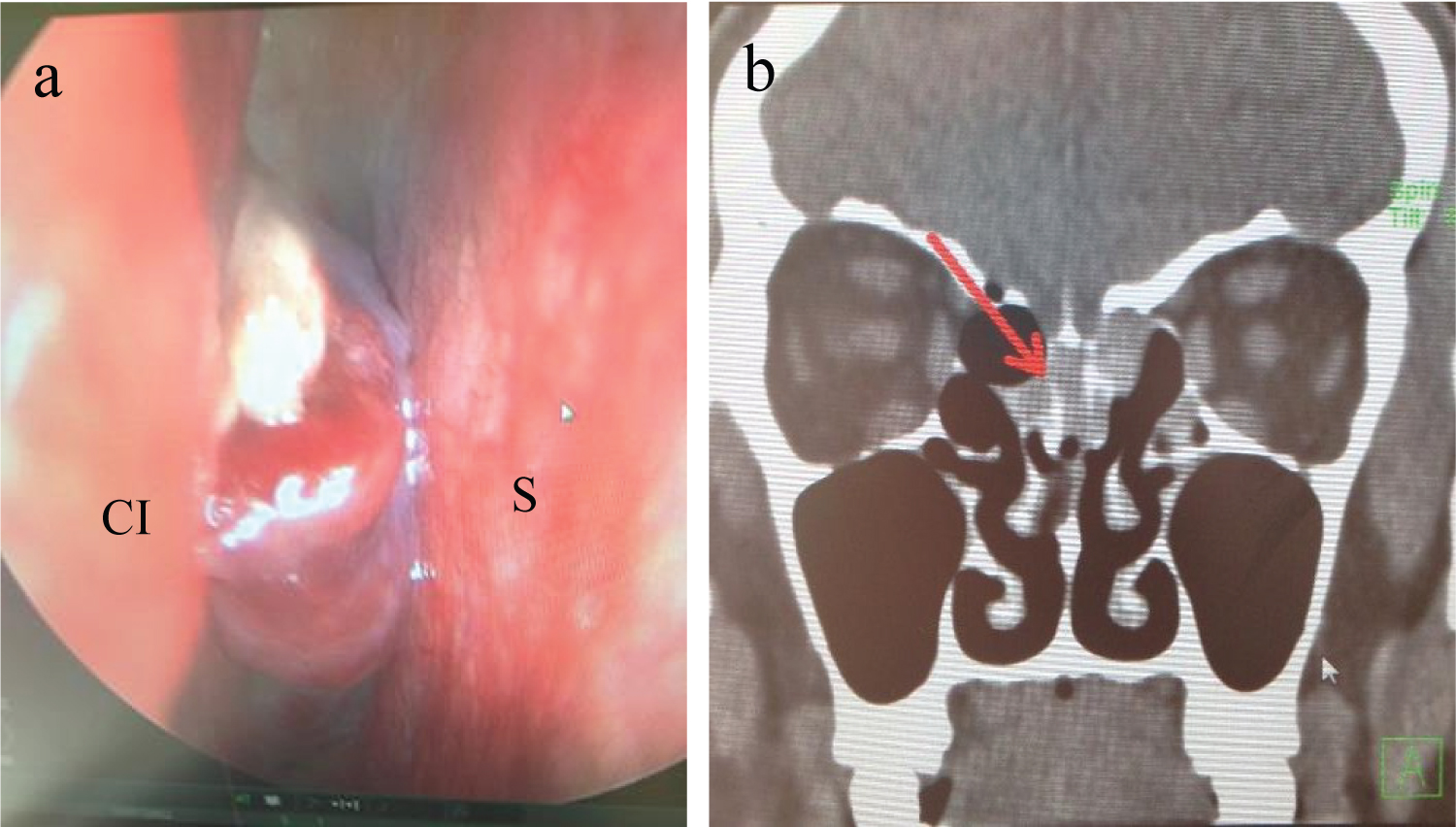 Figure 5: Preoperative findings.
Figure 5: Preoperative findings.
a) Endoscopic findings demonstrate a mass in the posterior region of the olfactory cleft that seems to be arising from the nasal septum; b) Coronal CT shows a soft tissue mass in the posterior region of the olfactory cleft.
View Figure 5
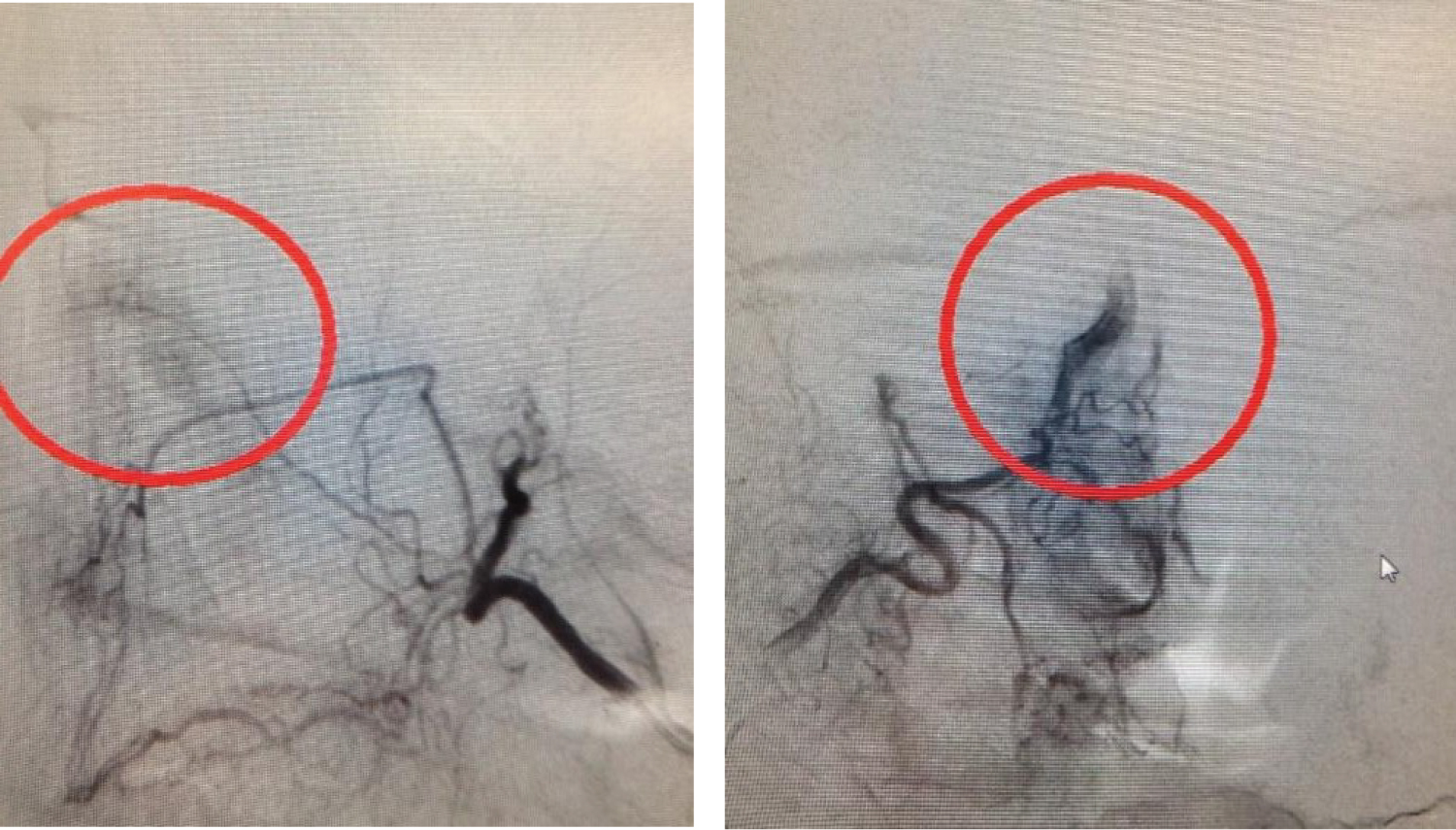 Figure 6: Preoperative angiography.
Figure 6: Preoperative angiography.
Angiography shows bilateral sphenopalatine arteries and the posterior lateral nasal branch of the sphenopalatine arteries supplying the tumor.
View Figure 6
Sinonasal hemangiopericytomas are low-grade, malignant vascular soft tissue tumors that can arise anywhere in the body, slowly invading into surrounding tissues. Generally, these tumors are considered to have malignant potential with late recurrence rates reportedly within the range of 8-53% [2-6] and metastasis occurring in 12-60% of cases [5,6]. Complete tumor removal is thus very important, because local recurrences tend to show metastasis.
Differential diagnosis from other more common pathologies in the sinonasal region (nasal polyposis, fungus ball, malignancy, encephalocele, etc.) should be undertaken preoperatively. The first step in making a diagnosis is to suspect a vascular tumor based on endoscopic findings. Radiological examinations are very important. Contrast-enhanced CT and MRI of the paranasal sinuses reveal vascular tumors as strongly contrast-enhanced. CT also allows a check for infiltration of bone around the tumor. On T2-weighted MRI, tumor masses are iso- to hypointense, whereas inflammatory fluids appear hyperintense [7]. Contrast-enhanced MRI is the best way to show tumor infiltration into the skull. The best visualization of the vessel supply for sinonasal hemangiopericytoma can be achieved by conventional digital angiography, which also helps to plan preoperative embolization [8].
Even though the final diagnosis is obtained from histopathological examination, several authors have suggested [9-12] avoiding biopsies if the tumor size suggests a risk of severe bleeding [2]. However, misdiagnosis can lead to an incorrect surgical approach, with incomplete excision, which may favor recurrences or metastases. Preoperative tissue biopsy is thus warranted whenever possible, while preparing for bleeding. In cases where biopsy is not possible before surgery, frozen section analysis should be performed intraoperatively to determine the suitable range for tumor resection.
Morphologically, hemangiopericytoma is very similar to a wide range of soft tissue tumors. Differential diagnoses include solitary fibrous tumor, myopericytoma, glomus tumor, perivascular epithelioid cell neoplasm, extra-gastrointestinal stromal tumor, and synovial sarcoma [13]. Histochemical and immunohistochemical techniques are essential, mainly to exclude morphologically similar soft tissue tumors. No specific immunohistochemical markers for sinonasal-type hemangiopericytoma have been identified to date.
However, the tumor cells in hemangiopericytoma are typically positive for vimentin (98% of specimens) and smooth muscle act in (92%) [14]. Positive staining for cytokeratin excludes a diagnosis of hemangiopericytoma. Intimate relationships between hemangiopericytoma and solitary fibrous tumor have been reported. Differentiating between these tumors is important, because most solitary fibrous tumors of the soft tissues are benign. CD34 is an extremely useful marker for this purpose [15,16].
Preoperative angiography is very useful for identifying whether the tumor has a large blood supply and judging the necessity of preoperative embolization of the tumor. In Case 1, preoperative embolization was not performed because the base of the tumor was limited and the feeder vessel to the tumor was thought to be a branch of the internal jugular artery. However, preoperative embolization is needed in cases of very large lesions or those showing intracranial lesions to reduce blood loss intraoperatively and tumor size. However, depending on the facility, angiography and embolization may be difficult. Angiography itself also carries a risk of complications such as cerebrovascular accident [17,18]. In cases where angiography cannot be enforced, the amount of bleeding may be able to be reduced by cauterizing blood vessels that seem to be feeding the tumor during surgery. If the anterior or posterior ethmoidal artery is considered to be the feeding vessel, coagulation of the vessel can be performed under the canopy of the ethmoid sinus or within the orbit. For posterior tumors, coagulation of the sphenopalatine artery at the sphenopalatine foramen may reduce bleeding from the tumor to some extent.
Due to its latent potential for malignancy, wide local excision is still considered the gold-standard treatment for sinonasal hemangiopericytoma [19]. For tumors in paranasal sinuses, an external procedure is most frequently chosen to achieve total tumor excision. In particular, a craniofacial approach is necessary when the cribriform plate or base of the skull is breached, but tumor located purely intranasally or strictly within the ethmoid or sphenoid sinus can be removed via an endonasal approach. We were able to remove both tumors en bloc in endoscopic operations leaving behind bone of the cribriform plate.
No complications such as leakage of cerebrospinal fluid (CSF) were seen in either case. However, endoscopic skull base surgery must be performed after tumor resection at the time of CSF leaks during the operation. Most recurrences are probably the result of inadequate surgical excision. Because recurrence can develop years after treatment, lifelong follow-up is necessary [2-4].
We encountered two cases of sinonasal hemangiopericytoma. We removed the tumors en bloc inendoscopic operations. Prior to tumor resection, the anterior and posterior ethmoidal arteries were cut via a transconjunctival incision in Case 1, and preoperative angiography and embolization were performed in Case 2 to reduce both surgical time and blood loss.
Tumor excision must be complete due to the latent potential for malignant behavior of these tumors. This may be achieved using an endonasal endoscopic approach. If this approach is not possible, an external approach is needed and can potentially be combined with endonasal endoscopic control. No recurrences have been found so far in either of our patients, but recurrence can develop years after treatment, so lifelong follow-up will be continued.
The authors declare no potential conflicts of interest.
The authors received no specific funding for this work.
Dr. Arimoto (Area of interest: Sleep medicine), Dr. You and Dr. Kawade (Area of interest: Rhinology) consulted on treatment methods as part of the same nasal sinus group, and performed the surgery together. Dr. Kishimoto (Area of interest: Audiology), Dr. Uchida (Area of interest: Audiology), Dr. Ogawa, and Dr. Hujimoto (Area of interest: Speech and language) were asked to read and critique the paper and identify points for improvement. All authors have read and approved submission of the manuscript.
We would like to thank Yasuhiro Takahashi from the Department of Ophthalmic Plastic and Reconstructive Surgery for his cooperation with the transconjunctival incision surgery.
Conference presentation: This manuscript was presented as a poster at Rhinology World Congress, September 1-3, 2017, Hong Kong, China.