Hypertension is the most prevalent risk factor for cardiovascular disease, including coronary heart disease, heart failure, myocardial infarction, stoke, end-stage renal disease and erectile dysfunction. Cardiovascular diseases (CVDs) are the leading cause of death worldwide, and although their etiology were multifactorial, eating habits have been associated with its development. There has been a great deal of interest in studies associating the beneficial effects of food with prevention and treatment of many diseases. Prevention and treatment of diseases can be provided by nutraceuticals, which are defined as foods or part of food that provides health benefits, as well as by isolated nutrients and dietary supplements.
Narrative review of literature. The search for the articles was done in the databases PubMed, Cochrane, Virtual Health Library and academic google, without language restriction or period of publication. The descriptors used in the search strategy were: nutraceuticals, diet, treatment, clinical trials, cardiovascular diseases and hypertension. In addition to these, the terms "flavonoids", "vitamins", "lycopene" and "garlic" were used.
In this review, we discuss if there are benefits in the use of nutraceuticals for the prevention and treatment of cardiovascular diseases, and its relationship with risk reduction and maintenance of physical and mental well-being.
Vitamins, garlic, lycopene and tomato have already been shown to be able to reduce the blood pressure and prevent or delay cardiovascular diseases; however, further studies are needed to confirm their effects, especially with respect to supplementation in humans.
Nutraceuticals, Dietary, Hypertension, Cardiovascular disease
ADMA: Asymmetric Dimethylarginine; AGE: Aged Garlic Extract; AT1: Angiotensin II Type 1; AT2: Antiogentin II Type 2; CAD: Coronary Artery Disease; CVD: Cardiovascular Disease; DADs: Diallyl Disulfide; DAS: Diallyl Sulfide; DATs: Diallyl Trisulfide; H2S: Hydrogen Sulphide; L: Liter; LDL: Low Density Lipoprotein; MATs: Methyl Allyl Trisulfide; mcmol: Micromol; mg: Milligrams; miRNA: Micro-Ribonucleic Acid; mL: Milliliters; NO: Nitric Oxide; ROS: Reactive Oxygen Species; Vitamin C: Ascorbic Acid
Essential, primary, or idiopathic hypertension accounts for 95% of all hypertension cases, which is defined as high blood pressure and involves comorbidities, such as renal and vascular disease, renal failure, or other causes of secondary hypertension or mendelian forms, monogenic, are not present [1], being a complex and multifactorial disease [2]. Hypertension is responsible for 45% of heart attacks and 51% of strokes and is a responsible factor for 9.4 million worldwide deaths related to CVDs [3]. It is estimated that over the next 25 years, the cost of not investing in the prevention and treatment of cardiovascular disease could reach $47 trillion worldwide [4].
If uncontrolled, diseases related to high blood pressure will be a substantial burden. More specifically, the direct burden on health will be drastically affected, as approximately 30% of patients with hypertension are not treated properly, demonstrating a need for new treatment options [5], as well as new forms of prevention and management of this condition. Based on this, several studies have demonstrated the importance of nutraceuticals in the management of hypertension [6] and cardiovascular diseases [7].
Nutraceuticals are foods or food derivatives that provide medicinal and health benefits, including the prevention, management and/or treatment of a disease, and included a class the isolated nutrients, encapsulated dietary supplements, herbal products and processed foods, such as cereals, soups and beverages [8-11]. Most nutraceuticals still need support from large clinical trials to demonstrate their efficacy and safety profile, prior to their use in cardiovascular prevention and treatment [12].
There are many studies reporting diet and reduction of blood pressure improve cardiovascular health [13-15], however, the association between different nutraceuticals and hypertension is still unclear. This is mainly due to the fact that there are many biases, and inconclusive, inconsistent studies, which have lead to limited guidelines for the safe and effective use of nutraceuticals for the treatment of hypertension when compared to a regimented diet plan and treatment with medication [16]. The aim of this review is to summarize the effects and mechanism of action of many of the most well known nutraceuticals used in treatment of cardiovascular diseases, in particular hypertension.
It is a study of a narrative review of the literature guided by the following research question: How is adherence to nutritional guidelines evaluated? Search of articles was carried out in electronic databases Scientific Electronic Library Online (SciELO), Literatura Latino-Americana e do Caribe em Ciências da Saúde (LILACS), Public Medical (PubMed), Cochrane, Virtual Health Library, without language restriction or period of publication. The following descriptors were used: "Nutraceuticals", "diet", "treatment", "clinical trials", "cardiovascular diseases" and "hypertension". In addition to these, the terms "flavonoids", "vitamins", "lycopene" and "garlic".
Inclusion criteria were: articles available in full, approaching the issue of the use of nutraceuticals and hypertension and/or cardiovascular diseases and discussing the subject by establishing some relationship with prevention and/or treatment. Articles which were excluded: those who have presented conflicts of interest or scientifically irrelevant or contradictory data. Thus, 91 articles were selected from journals.
The most abundant secondary metabolites in the plant kingdom are polyphenols, which contain the molecules of the flavonoid (Anthocyanins, flavan-3-ols, flavonols, flavanones, flavones and isoflavones) and non-flavonoid family (Phenolic acids and stilbenes) [17]. Many studies associate the consumption of foods and beverage rich in polyphenols (such as some vegetables, fruits, cocoa contained in chocolate, red wine, grape juice and tea) with the reduced risk of cardiovascular diseases [18-20].
Flavonoids are a group of phenolic compounds found naturally in fruits, vegetables, tea and red wine that have cardioprotective and anticarcinogenic properties [21]. Many studies have demonstrated that the properties of flavonoids, including antioxidants [22], antiinflammatory [23], may also improve vascular functions [24,25]. In addition, an increased intake of tea and flavonoids may contribute to the primary prevention of ischemic heart disease [26], as well as reduced risk of coronary heart disease mortality, demonstrated by high dietary intake of flavonols [27].
Although several studies have demonstrated the cardioprotective effects of flavonoids, there exists conflicting evidence as well. There is no certainty of the association between flavonoid intake and total coronary disease, although studies have not ruled out the possibility that flavonoids may have a protective effect on established coronary disease [28]. The acute and repetitive consumption of flavonoids in foods, have been shown to improve endothelial function in patients with coronary artery disease, after heart transplantation, or in healthy volunteers with or without cardiovascular risk factors [12].
Experimental studies in both animals and humans suggest that vascular activities of flavonoids occur, at least in part, because of the increased activity of nitric oxide synthase [29-31]. Soy isoflavones also belong to the class of flavonoids, in which a diet rich with these substances has been suggested to promote vasodilation, improve endothelial function and reduce blood pressure in animals' models [32]. In human trials, the results have not been reliable due to experimental bias.
To verify and confirm the effect of diet with soy isoflavones on blood pressure, a meta-analysis study of randomized controlled trials was performed, which concluded that isoflavones soy derived decrease blood pressure in hypertensive individuals but not in normotensive individuals, however, additional trials were needed to confirm these results. Therefore, it is still not reliable to state the effects of soy isoflavones on blood pressure [33] that is possible to confirm at Figure 1.
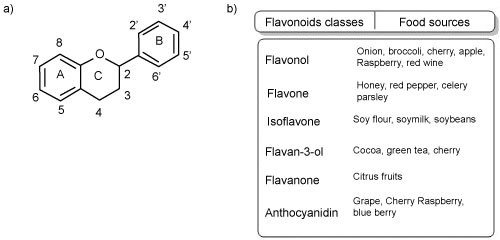 Figure 1: a) Basic structure of the flavonoids; b) Flavonoids classes and food sources of flavonoids.
View Figure 1
Figure 1: a) Basic structure of the flavonoids; b) Flavonoids classes and food sources of flavonoids.
View Figure 1
A systematic review with meta-analysis concluded that the dietary intake of subclasses of flavonoids is inversely associated to CVD risks, thus, the intake of flavonoids reduces the risk of CVDs, however, this meta-analysis based on observational studies and among other biases, possible error of dietary classification, presence of unmeasured/uncontrolled risk factors were observed [34].
Another flavonoid-rich food is extra virgin olive oil. Extra virgin olive oil intake was inversely associated with increased systolic and diastolic blood pressure [35]. Blood pressure reduction was also observed in hypertensive twins treated with an olive leaf extract, at 500-1000 mg per day for 1-8 weeks, compared with placebo [36]. Extra virgin olive oil may also contribute to reduce blood pressure by increasing plasma nitrite/nitrate and decreasing serum asymmetric dimethylarginine (ADMA) [37], which is an endogenously produced molecule capable of inhibiting synthesis of nitric oxide (NO) [38]. Taken together, a diet rich in polyphenols may contribute to the improvement of endothelial function by improving the bioavailability of NO.
Grape seed and red wine extracts are also known to contain high levels of polyphenols, and the beneficial health effects have been attributed to consumption of these compounds. Grape and wine polyphenols can potentially reduce elevated blood pressure and exert beneficial effects in cardiovascular diseases by inhibiting platelet aggregation, reducing oxidative stress, and decreasing LDL oxidation [17].
Despite the fact that the mechanism of action of grape seeds still is not fully understood, its extract has might act in an antioxidant capacity, leading to the reduction of the amount of reactive oxygen species (ROS). This capability demonstrates its potential in CVD as elevated ROS are commonly observed in cardiovascular and metabolic diseases [39].
A large body of evidence exists demonstrating the effects of flavonoids on the cardiovascular system, however, more studies are needed to better clarify the appropriate intake amount that results in significant and safe outcomes. Additionally, defining which specific subclasses of flavonoids lead to these desired outcomes and in which specific disease conditions they would be beneficial.
Hypertensive patients have a higher production of ROS and present a greater than normal response to oxidative stress [40,41]. Additionally, the endogenous and exogenous antioxidant defense mechanisms of the hypertensive patient are impaired [40]. Several epidemiological studies have shown that antioxidant deficiency have been implicated in hypertension and cardiovascular diseases [42-44]. Considering the epidemiological growth of chronic non-communicable diseases, such as diabetes mellitus and cardiovascular events, a variety food are of interest and have been studied due to the antioxidant nutrients that are contained, such as ascorbic acid (vitamin C) [45].
The importance of vitamin intake is well known and accepted, but to the extent how vitamins influence health and the cardiovascular system remains poorly elaborated. Several studies have shown that reduced levels of plasma vitamin E were related to an increase in cardiovascular risk. Similarly, lower ascorbic acid or vitamin C plasma levels were associated with an increased risk of cardiovascular events [12,46,47].
Higher blood concentrations of vitamin C in humans are associated with lower blood pressure [48,49]. In a 17-week controlled-diet depletion-repletion of vitamin c study, confirmed an inverse correlation of plasma ascorbate levels, diastolic and systolic blood pressures and affirmed that the efficacy of fruits and vegetables in reducing blood pressure may be related to vitamin C. In addition, tissue ascorbic acid levels may be important in maintaining low blood pressure [50]. Thus, in order to obtain a beneficial effect on blood pressure, it is recommended to maintain a serum level of plasma ascorbate of at least 100 mcmol/L. However, additional long-term intervention studies should be performed to confirm this finding [51].
Ascorbic acid decreases the binding affinity of the AT1 receptor, without modifying its maximal binding capacity and without altering the binding affinity of the AT2 receptor for angiotensin II. These results provide a mechanistic explanation for the reported blood pressure lowering effect of ascorbic acid, since most of the angiotensin II-induced cardiovascular effects, such as vascular contraction, aldosterone secretion, cardiovascular cell growth and proliferation, among others, are mediated by AT1 receptor. This reduction of the AT1 receptor affinity for angiotensin II appears to occur by disrupting the ATR1 disulfide bridges [52].
A randomized, double-blind, placebo-controlled factorial trial of vitamin E and vitamin C performed between 1997 and 2007, which included 14641 US male physicians, who were initially aged 50 years or older and included 754 men with prevalent cardiovascular disease at randomization, was performed to confront studies suggesting that vitamin E or vitamin C may reduce the risk of cardiovascular disease. According to the authors of this study, few long-term trials have evaluated men at an initially low risk of cardiovascular disease, and no previous trial in men has examined vitamin C alone in the prevention of cardiovascular disease. Thus, in this large, long-term trial of male physicians, neither vitamin E nor vitamin C supplementation reduced the risk of major cardiovascular events. In middle-aged and older men, these data provided no support for the use of these supplements in the prevention of cardiovascular disease [46]. At another study, the effects of vitamin C resulted in the reduction of blood pressure with an increase in nitric oxide, leading to an improvement in endothelial function and arterial compliance [47].
The inverse relationship between plasma ascorbic acid and blood pressure were observed in the attenuation of increases in blood pressure in healthy young adults that reduced the risk of associated vascular events. This study suggests that vitamin C may be an important factor in blood pressure regulation, even among healthy young adults [53]. Ingestion of foods rich in vitamin C appear to be important for lowering blood pressure, however, more long-term studies in patients of different ages, as well as different stages of hypertension need to be performed to clarify which patients are may benefit.
Other important vitamin involved in cardiovascular diseases is vitamin D. This vitamin is synthesized in the skin after exposure to sunlight and is also taken up with the diet and is contained particularly in some species of fish [54]. After vitamin D enters the body, it is rapidly converted to 25-hydroxyvitamin D (25 (OH) D), its main circulating form [55]. Vitamin D has a classical role in regulating calcium-phosphate homeostasis and also appears to exert immunoregulatory and anti-inflammatory effects [56].
Many studies suggest that there is a strong association between vitamin D deficiency and cardiovascular disease [12,57,58] and describe inverse associations between low sun exposure, that is, low serum 25(OH)D levels, and increased risks of stroke, myocardial infarction, peripheral vascular diseases and heart failure [59-65]. Besides that, systemic and local vitamin D deficiency in coronary artery disease (CAD) patients are assciated an increased activation of inflammatory pathways at epicardial adipose tissue (EAT) level [56], and its accumulation may be a risk factor for CAD [66]. Although Dozio and co-workers have been the first to associate, in humans, vitamin D and inflammation at EAT level in CAD patients, they could not assert if vitamin D supplementation would be able to oppose local EAT inflammation in these patients. Using animal model, Gunasekar and co-workers [67], demonstrated that vitamin D deficiency increases both the recruitment of M1 macrophages, as the proinflammatory expression of adipokine in the EAT, furthermore, it suggests that vitamin D supplementation decreases inflammatory processes and promotes anti-inflammatory responses in EAT. Vitamin D deficiency it is also capable promoted the progress of atherosclerosis and decreased plasma HDL-C level, and the supplementation of vitamin D3 in the diet can significantly increase the level of HDL-C, as macrophage pro-inflammatory polarization, in animal model, resulting in protection against the development of atherosclerosis [68].
Vitamin D plays an important role in cardiovascular protective effects through various mechanisms, such as modulation of vascular tone, regulation of blood pressure, vascular smooth muscle cell functions and maintenance of healthy endothelium [69-71]. The anti-inflammatory and anti-mitotic activities of vitamin D aid in the stabilization of the endothelium, which justifies, at least in part, its cardiovascular effects [72,73].
Although several evidences suggest an association between vitamin D deficiency and endothelial dysfunction and CVDs, there is no evidence of randomized clinical trials demonstrating that CVDs are prevented with vitamin D supplementation [69,74,75]. There still remains a great need to understand mediates cardiovascular function to improve target therapies, as well as prospective clinical interventions to advance the evidence of the efficacy of vitamin D repletion on cardiovascular health [76].
Lycopene is a carotenoid found in red fruits and vegetables likes papayas, tomatoes, red peppers, watermelons and others [77]. Tomatoes are well recognized for their culinary versatility and also contribute to dietary nutritional value [78]. Almost 90% of the total carotenoids that make up the tomato are composed of lycopene [79], although it is also composed of carotene, folate, phenolic compounds and vitamins C and E [80]. The association between tomato and lycopene consumption with cardiovascular disease risk reduction has gained research interest [78].
According to the Framingham Heart Offspring Study, there is a strong inverse association between the consumption of lycopene and the incidence of myocardial infarction, coronary insufficiency and angina pectoris [81]. In addition, plasma levels of lycopene in hypertension, acute myocardial infarction, stroke, and atherosclerosis were described as low [82,83].
Among the major carotenoids, lycopene is the most powerful antioxidant and plays an important role in preventing cardiovascular disease in humans [84]. Lycopene bioavailability in supplements has not been as extensively studied and remains an important area for research [78].
An experimental study using wistar rats showed that tomato supplementation for 3 months promoted changes in miRNA expression and reduced oxidative stress. In addition, these changes appear to be responsible for the reduction of the cross-sectional area of left ventricular myocytes and improvement of diastolic function. Despite this, further studies need to be conducted to investigate the potential targets of tomato and lycopene in the prevention and treatment of cardiovascular diseases and to clarify their beneficial cardiac effects, which have not yet been studied [85].
It is well known that endothelial function reflects vascular health (CITE). Furthermore, endothelial dysfunction is involved in the development of atherosclerosis and, in addition, coronary or peripheral vascular endothelium dysfunction appears to be an independent predictor of cardiovascular events [86]. Increased oxidative stress may be a contributing mechanism for endothelial dysfunction [87].
Interestingly, in a randomized, double-blind, placebo-controlled study, the 8-week supplementation with lycopene has been shown to promote beneficial effects of oxidative stress biomarkers (through the reduction of oxidative DNA damage and increased plasma superoxide dismutase enzymatic activity). Additionally, their antioxidant effects may play a substantial role in improving a endothelial function, which is deeply related to inflammatory markers. Those results demonstrate the beneficial effect of daily lycopene intake [88].
Many studies have associated tomato and lycopene with lower incidence rates of cardiovascular diseases [82,83,89], however the effect of lycopene supplementation remains unclear and unconfirmed [90]. Although almost 90% of the total carotenoids that are found in the tomato are composed of lycopene [79], the other bioactive compounds that are found should also be studied to better understand its beneficial effects. In addition, until new research clarifies and confirms the significant benefits of lycopene supplementation in humans, it is recommended to consume fruits and vegetables rich in natural carotenoids [90].
Garlic (Allium sativum) has played a substantial historic role in both food and medicinal importance [91]. Garlic is mostly composed of water (65%), and carbohydrates as fructose, sulfur compounds, proteins and free amino acids [92]. The sulfur compounds present or generated from garlic are responsible for their dietary and medicinal functions [93]. It has recently been established that part of the cardiovascular benefits of garlic is due to the presence of allicin, also known as diallylic thiosulfinate [94].
When the cellular structure of garlic is damaged, C-S-lyase (alliinase) acts on alliin (S-allyl-L-Cysteine sulfoxide) and converts it into allicin (allylthiosulfinate). Allicin forms several sulfides by non-enzymatic reaction as diallyl sulfide (DAS), diallyl disulfide (DADs), diallyl trisulfide (DATS), or methyl allyl trisulfide (MATS) (Ariga & Seki, 2006), all of which act with potent antiplatelet activity [95]. Garlic intake can inhibit the platelet aggregation in human individuals [96], enhances vasodilatation and fibrinolysis in animals and humans [97].
An important and well studied garlic preparation is aged garlic extract (AGE), which is prepared by storing sliced raw garlic in an ethanol solution for 20 months, at room temperature. Further studies are required to assess the benefit of this garlic preparation. In order to further investigate the properties of AGE, future studies could examine a combination of antioxidants over a longer duration and examine a diabetic cohort with higher cardiovascular risk, such as those with established cardiac disease [98].
In addition to AGE, there are other preparations of garlic, such as garlic oil, garlic powder and garlic oil that may present beneficial outcomes [94,99]. In addition to the afore mentioned cardiovascular effects, garlic was found to act in a cardioprotective manner as promoted by H2S, which can be produced by the conversion of garlic derivatives into human erythrocytes, protecting the heart by antioxidant, antiapoptotic and anti-inflammatory actions [97]. H2S is a cardioprotective gas signaling molecule and is responsible for a wide range of physiological roles in mammalian tissues. It is important for cell function and protects the cell against apoptosis, oxidative stress and necrosis [100].
Treatment with exogenous H2S is possible and may be responsible for a significant reduction in the size of myocardial infarction in mice with murine lesions and ischemia/reperfusion [101]. A tremendous amount of scientific literature supports the proposal that garlic consumption has a significant cardioprotective effect [102]. Clinical trials to evaluate the effects of garlic on cardiovascular diseases were performed after very promising in vitro studies suggesting beneficial effects of garlic, however many of these clinical trials were inconclusive [103]. The different forms of garlic compounds are possible to see at Figure 2.
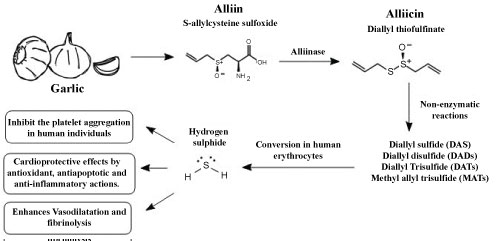 Figure 2: Garlic and its cardiovascular effects via hydrogen sulfide signaling.
View Figure 2
Figure 2: Garlic and its cardiovascular effects via hydrogen sulfide signaling.
View Figure 2
A systematic review and meta-analysis concluded that in hypertensive patients treated with garlic-only preparations, demonstrated a reduction of blood pressure in individuals with hypertension, when compared to placebo [104]. A recent study concluded that garlic has the potential to reduces blood pressure in hypertensive subjects, similar to standard blood pressure medication, such as angiotensin-converting enzyme inhibitors, angiotensin II - receptor blockers, calcium-channel blockers and diuretics [105].
Furthermore, the supplementation with garlic preparations may be a complementary treatment option for hypertension and experiments should be performed to investigate whether garlic could have an anti-hypertensive effect, in pre-hypertensive individuals, which would be important in the prevention of hypertension progression [104]. In a meta-analysis, garlic presented important results in subjects with mild hypertension, however, insufficient evidence was gathered to recommend for clinical therapy [106] and despite studies of blood pressure and garlic preparations have been inconclusive [103,107-109].
Further testing need to be performed to clarify the benefits of garlic in the treatment of hypertension and cardiovascular disease, and although there is strong evidence of its beneficial cardiac effects, caution should be exercised in the use of garlic due to the appearance of gastrointestinal problems [110]. Taken together, the clinical trials suggest that garlic play a role preventing or delaying cardiovascular disease, but more studies are needed to confirm its effects.
A recent randomized controlled pilot study, with thirty menopausal healthy women of rural communities (45-65 years of age) with mild hyperlipidemia were divided and randomly assigned in three groups: control group, subjects without any treatment and 2 experimental groups, one group treated with curry leaves powder (5 g) and other group treated with cucumber (100-125 g), once daily for 45 consecutive days. Low density lipoprotein-cholesterol (LDL-C), total cholesterol (TC), and triacyl glycerol (TAG) levels were significantly reduced in the curry leaves powder and cucumber treated groups. Only in the curry leaves powder treated group, the high density lipoprotein - cholesterol (HDL-C) level was significantly elevated. This result suggests the potential nutraceutical role of cucumber and curry leaves in treating CVD [111].
To investigate whether the consumption of beet juice in addition to a normal diet, would produce a measureable reduction in blood pressure, a double-blind, randomized, placebo-controlled, crossover study with 30 free-living adults was realized. Volunteers were randomized to receive 500 g of beetroot and apple juice (BJ) or a placebo juice (PL). Volunteers had BP measured at baseline and at least hourly for 24-h following juice consumption using an ambulatory blood pressure monitor (ABPM). This study concluded that beetroot juice will lower BP in men when consumed as part of a normal diet in free-living healthy adults [112].
Nutraceutical preparations of Allium cepa (onion) are indicated for treating various of acute and chronic diseases [113,114]. A randomised double-blinded placebo-controlled cross-over trial with sixty-eight subjects (thirty-four male, thirty-four female), randomised to receive quercetin (162 mg/day) from onion skin extract powder or placebo with 6-week treatment periods separated by a 6-week washout period, evaluated the effects of a quercetin-rich onion skin extract on 24 h ambulatory blood pressure and endothelial function in overweight-to-obese patients with (pre-)hypertension. This study concluded that supplementation with quercetin (162 mg/day) from onion skin extract lowers ambulatory blood pressure in patients with hypertension, suggesting a cardioprotective effect of quercetin [115].
The management, prevention and treatment of cardiovascular diseases, such as hypertension, are based on lifestyle, hereditary factors and physical activity. The nutritional supplementation represents a possible strategy for management and prevention of hypertension and other cardiovascular diseases.
The nutraceuticals act not only direct by antioxidant or anti-inflammatory properties but also indirectly, via modulation on various physiological pathways. A lot of the nutraceuticals that was described, such as oil extracts, plants, fruits and vegetables as well as botanical extracts, have shown beneficial effects on the cardiovascular system through substances such as phenolic compounds or polyphenols, lycopene and flavonoids.
Nutraceutical supplementation and their intake could improve blood pressure control and must be considered strategic management aimed at preventing hypertension, when considering its benefits and low costs. This review demonstrates the importance of new strategies in the control and treatment of hypertension with non-pharmacologic therapy, and through the use of the nutraceuticals established antihypertensive activity in humans, in association with a coherent improvement in diet and lifestyle.
The authors would like to thank to the Gonçalo Moniz Institute (Oswaldo Cruz Foundation - Bahia/Brazil), Federal University of Bahia, Conselho Nacional de Desenvolvimento Científico e Tecnologico (CNPq) and Fundação de Amparo à Pesquisa do Estado da Bahia (FAPESB) for the support. We thank Josiane Barbosa for the kindness in drawing the figures of this paper.
All the authors agree with this publication and confirm that none conflict of interest exists.
• Quiara Lovatti Alves worked at all stages from the conception of the study to the revision of the final version of the article.
• Samuel Barbosa Camargo worked on study design and article writing.
• Darízy Flávia Silva was the final reviewer of the article and the supervisor responsible for the work.