Older adults who qualify for cochlear implantation demonstrate significant improvements in speech recognition and quality of life with cochlear implant use. Cochlear implantation is a safe and effective procedure for patients with hearing loss, with some additional medical considerations specific to older adult patients. The indications for cochlear implantation have expanded from cases of bilateral severe-to-profound hearing loss to cases of normal-to-moderate low-frequency hearing sensitivity and severe-to-profound high-frequency hearing loss. The expansion of indications for cochlear implantation was prompted by evidence of low-frequency hearing preservation due to modified electrode array designs and surgical techniques, and significantly improved performance when listening with the combination of acoustic and electric stimulation (EAS) in the same ear. Older adults who underwent cochlear implantation as part of clinical trials investigating the effectiveness of EAS have been shown to experience significantly improved speech recognition and quality of life as compared to preoperative performance with conventional hearing aids.
Cochlear implantation is the standard treatment for cases of moderate-to-profound sensorineural hearing loss who experience limited speech recognition with appropriately-fit hearing aids. Though originally indicated for patients with severe-to-profound hearing loss bilaterally, candidacy criteria for cochlear implantation have expanded over the past several decades. Populations including those with normal-to-moderate low-frequency hearing in the ear-to-be implanted and those with normal hearing in the contralateral ear (known as single-sided deafness) have demonstrated significantly improved performance with cochlear implant (CI) use as part of clinical trials, prompting the expansion of candidacy criteria to include these patient populations [1-6]. The prevalence of hearing loss increases with age, which negatively impacts quality of life and has shown to be associated with cognitive impairment [7-9]. Providing older adults (≥ 65 years of age) with effective treatment options such as cochlear implantation for their hearing loss supports improved speech recognition and quality of life [10-20]; however, insurance coverage of this procedure is currently restricted to beneficiaries with bilateral moderate-to-profound sensorineural hearing loss. The aim of the present report is to review aspects of cochlear implantation unique to older adults and data that demonstrate the effectiveness of CI use for older adults who meet the new criteria for cochlear implantation.
The overall health of an older adult patient is an important consideration during the candidacy evaluation for cochlear implantation. A combination of comorbidities, medications, and physiologic changes associated with aging can contribute to increased risk for undergoing anesthesia [21]. Specific attention should be focused on cardiovascular, pulmonary, neurological, and renal function in older adults being evaluated for preoperative clearance [22]. The overall risks of anesthesia and the surgical procedure itself should be weighed against the expected benefits from CI use.
Generally, there are few systemic effects following cochlear implantation, though there are potential postoperative considerations for older adults including flap-related issues, dizziness and vertigo, and psychological considerations [23]. Flap-related issues are considered the most common complication following cochlear implantation. It is possible that chronic vascular changes from comorbidities such as long-standing hypertension, diabetes mellitus, or other vascular disease may increase this risk. Postoperative dizziness is rare following cochlear implantation, though older adults have an increased likelihood of underlying multisensory balance issues that may contribute to transient dizziness postoperatively [23]. The patient's psychological health, including attention to symptoms of dementia, should be included as part of the cochlear implantation candidacy evaluation as these may limit the overall success of CI use, though some studies have suggested CI use may slow cognitive decline [9,20,24].
Several studies examining perioperative and postoperative outcomes of older adults undergoing cochlear implantation report overall low complication rates with no long-term morbidity or mortality [11,25,26]. Though Coehlo, et al. (2009) found that advanced age was not an independent risk factor for general anesthesia, a study by Carlson, et al. (2010) showed older adults were more likely to experience anesthetic complications but found no difference in surgical complications as compared to younger adults. Cochlear implantation is generally considered to be a safe procedure for older adults that are deemed suitable to undergo general anesthesia.
Recently, some CI centers have developed methods for cochlear implantation under local anesthesia [27-30]. A modified surgical approach is used in combination with local anesthesia and minimal sedation. Preliminary studies have suggested that this can be a safe and effective alternative for patients that may not be able to tolerate general anesthesia. Studies have been performed comparing cochlear implantation under conscious sedation/local anesthesia and those undergoing general anesthesia. There reportedly are decreased surgical times, lengths of stay, and drug costs in patients implanted with conscious sedation/local anesthesia, with no differences in perioperative morbidity compared to patients implanted under general anesthesia [29,30]. Though further research and surgical training may be required before widespread use, this surgical technique may be an alternative for older adult CI candidates with multiple comorbidities that may be poor candidates for general anesthesia.
A primary measure of postoperative success with CI use is speech recognition, which assesses the listener's understanding of spoken words. Typically, recorded word or sentence materials are presented to the listener in a soundbooth to assess preoperative performance with appropriately-fit hearing aids and postoperative performance with the CI. Multiple studies demonstrate significant improvement in speech recognition scores for older adults listening with a CI as compared to their preoperative performance with hearing aids [10-15,19]. Typically, patients demonstrate significant improvements in speech recognition within the initial months of CI use and experience asymptotic performance between 6-12 months of listening experience [31]. The findings are mixed when comparing the speech recognition of younger and older adult CI recipients, with some reporting similar performance between the age groups [10-12,19] and others reporting younger adults experience better performance than older adults [13,15]. A consideration of previous comparisons between age groups is that older adults may require longer CI use than younger adults before experiencing asymptotic speech recognition performance. Dillon, et al. (2013) observed that older adults continued to experience significant improvements in speech recognition in quiet and in noise out to 5 years of CI use, and maintained superior speech recognition through 10 years. While the rate of performance growth and acclimatization for speech recognition may differ from younger adult CI recipients, an important consideration is older adults experience a significant improvement with CI use as compared to their preoperative performance with hearing aids [10-15,19].
Though speech recognition measures are valuable clinical tools to measure performance growth with CI use, the influence on the patient's quality of life is another important measure of the success of cochlear implantation. Older adult CI recipients report significant improvements in their overall quality of life as well as significant improvements on measures specific to their social life, confidence, emotional wellbeing, telephone use, and levels of depression [15-20]. These subjective benefits have largely been found to be similar to younger adult CI recipients, though a study by Olze, et al. (2012) suggesting older adult CI recipients may experience greater improvements in quality of life as compared to younger adult CI recipients [19]. This was speculated to be secondary to the more severe effects of hearing loss on functioning capabilities and decline of health with age of elderly individuals as well as the realistic expectations of those undergoing cochlear implantation. Collectively these studies substantiate that restoring the hearing of older adults via cochlear implantation and CI use can significantly improve quality of life and communication abilities in this population.
Indications for cochlear implantation have expanded to include patients with normal-to-moderate low-to-mid frequency hearing sensitivity, severe-to-profound high-frequency hearing loss, and limited speech recognition with appropriately-fit hearing aids (Figure 1). Modified electrode array designs and surgical techniques have been shown to support low-to-mid frequency acoustic hearing preservation in the implanted ear following cochlear implantation [1,2,32-34]. In cases of postoperative hearing preservation, patients are fit with a device that combines acoustic amplification and CI technology in the same ear, known as electric-acoustic stimulation (EAS). Recipients with postoperative hearing preservation demonstrate significantly better speech recognition in quiet and noise when listening with EAS as compared to listening with a CI alone [1,2,33,35-37]. Additionally, listeners of EAS devices have shown significant improvements in localization (identifying the location of a sound source), and quality of life as compared to preoperative abilities with hearing aids [1,2,33,53,37-40]. Successful low-frequency hearing preservation has been reported for older adult CI recipients [40], supporting those older adults with hearing configurations meeting the expanded indications criteria should be considered candidates for cochlear implantation.
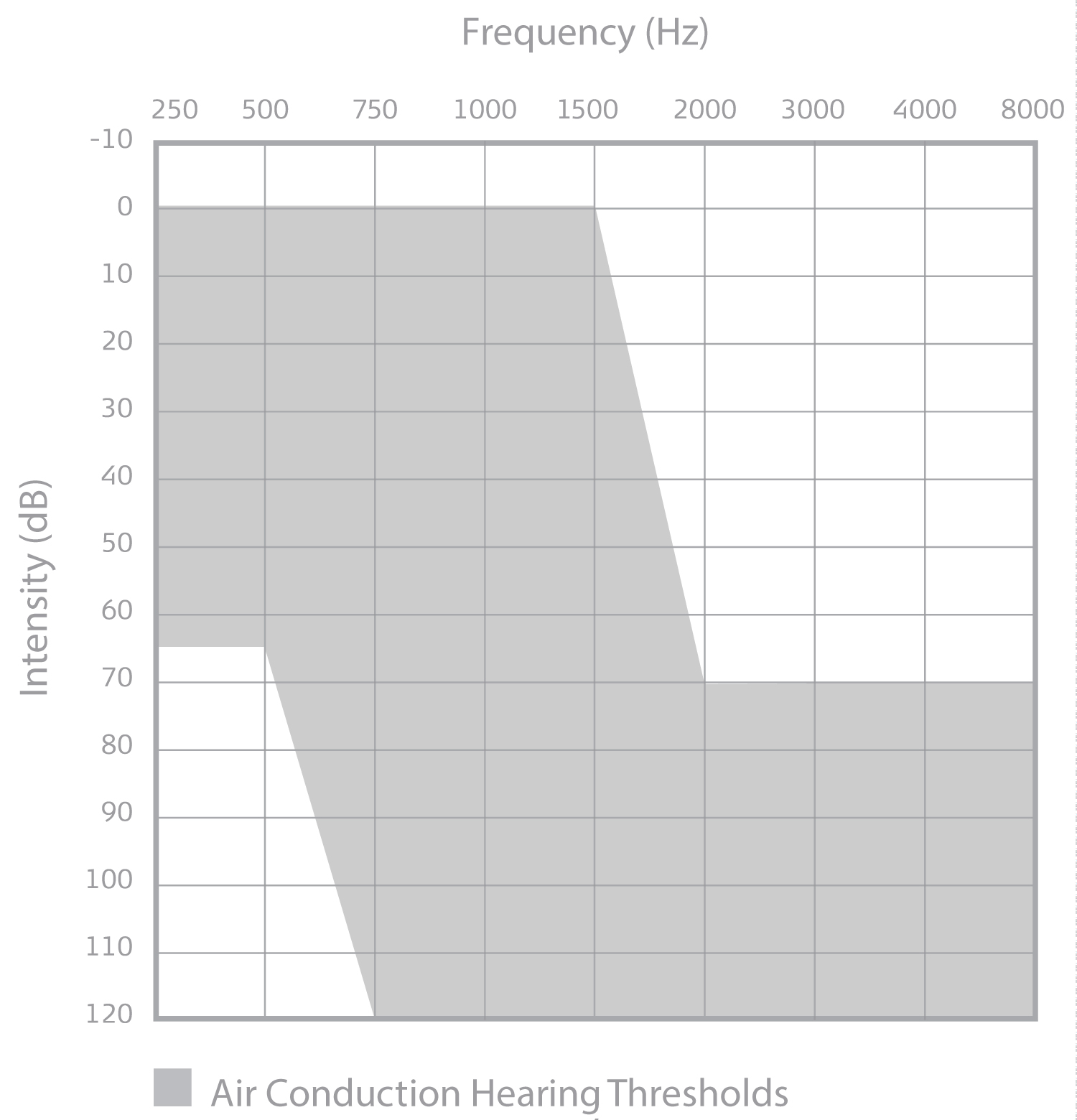 Figure 1: Audiogram displaying the unaided detection thresholds candidacy criteria (in gray) for the MED-EL Corporation Cochlear Implant System.
View Figure 1
Figure 1: Audiogram displaying the unaided detection thresholds candidacy criteria (in gray) for the MED-EL Corporation Cochlear Implant System.
View Figure 1
The addition of acoustic low-frequency hearing with electric stimulation provides the patient with cues that are typically not well replicated by a CI. Low-frequency cues help to resolve the fundamental frequency [37,41] and interaural timing differences [36,38,39], which support better speech recognition in noise and in challenging listening situations. Auditory aging has been shown to reduce the temporal acuity [42]. The initial clinical trials investigating the effectiveness of EAS included subjects above 65 years of age at implantation. Results are mixed regarding whether advanced age at implantation influences performance with EAS, with one study reporting a higher age at implantation yielded lower speech recognition scores [2] while another found no significant effect of age at implantation on performance [1]. A consideration of these studies is the study endpoints were 12 months post-activation. Considering older adult CI users have shown significant improvements out to 5 years of listening experience [14], older adult EAS listeners may also experience further improvements with long-term listening experience. While it is currently unclear to the extent that age at implantation may have on speech recognition with EAS, patients - regardless of age at implantation - demonstrate a benefit with EAS over preoperative abilities with hearing aids. A similar but more general conclusion has been made by Sprinzl and Riechelmann (2010), discussing that EAS devices can be an effective treatment for the older population, but more research on the topic is needed [43].
As observed in CI-alone users, EAS users have reported significant improvements in quality of life as compared to their preoperative perceptions [1,2]. Subjects listening with EAS report a decrease in perceived disability and an increase in device satisfaction as compared to their preoperative listening condition [1,2]. For instance, when analyzing the results from the Abbreviated Profile of Hearing Aid Benefit (APHAB), Pillsbury, et al. (2018) found that the overall score decreased (improved) by 30.2% at the 12-month interval. The APHAB has a total score as well as scores for four subcategories, which include Ease of Communication, Reverberation, Effectiveness in Background Noise, and Aversiveness of Sound. When analyzing performance on the individual subscales, 90% of subjects reported their EAS device provided benefit for the Ease of Communication and Reverberation subscales, and 91.7% of subjects reported benefit for the Effectiveness in Background Noise subscale [1]. Gantz, et al. (2016) reported similar findings, observing significant improvements on the Ease of Communication, Reverberation, and Effectiveness in Background Noise subscales. A significant difference was not observed on the Aversiveness subscale, which is a positive outcome as the Aversiveness subscale measures whether environmental sounds are bothersome or uncomfortably loud [2]. While investigations are ongoing, it appears that older adult EAS users may perceive postoperative improvements that support an improved quality of life.
The candidacy criteria for cochlear implantation continue to expand with evidence from prospective clinical trials demonstrating the effectiveness of CI use in new patient populations. Recently, the FDA approved single-sided deafness and asymmetric hearing loss as indications for cochlear implantation for the MED-EL CI system. Investigations of the effectiveness of CI use in subjects with single-sided deafness or asymmetric hearing loss included older adult recipients. While prospective clinical trials demonstrate the effectiveness of CI use on measures of quality of life, localization, and speech recognition in quiet and noise for subjects with single-sided deafness and asymmetric hearing loss [5,6,44], the current criteria for Medicare beneficiaries are restricted to those with moderate-to-profound hearing loss in both ears [45]. Ongoing work is investigating outcomes of CI use specific to older adult recipients with single-sided deafness and asymmetric hearing loss.
Older adult CI recipients demonstrate significant improvements in speech recognition and quality of life. Patients report significant improvements in their social life, confidence, emotional wellbeing, telephone use, and levels of depression. Indications for cochlear implantation continue to expand and now include those with single-sided deafness and asymmetric hearing loss. Older adult patients who meet the new indications for cochlear implantation may experience significantly improved speech recognition and quality of life as compared to their abilities with alternative treatment options.
Figure 1 was provided by MED-EL Corporation (Innsbruck, Austria).
MER and MTD are supported by a research grant from MED-EL Corporation to the university. HCP is a consultant for MED-EL Corporation. NJT has no conflicts of interest.