Many studies have shown the association between acute kidney injury (AKI) and morbidity and mortality in the Intensive Care Unit (ICU). Traditional measures of AKI, such as serum creatinine and urine output, fail to show dynamic changes in renal function. A feasibility study was performed to evaluate the Nephrocheck® test system which comprises of two biomarkers (Insulin-like growth factor binding protein 7 (IGFBP7) and tissue inhibitor of metalloproteinases-2 (TIMP-2)) to screen for patients at risk for AKI in the ICU and to guide management.
Patients were enrolled between November 2016 and March 2017 with respiratory failure or sepsis with no pre-existing elevation in the creatinine admitted to the ICU. In patients with an initial Nephrocheck® value ≥ 0.3, the primary team was notified and a kidney protective bundle was suggested. A repeat Nephrocheck® test was performed within 24 hours in those patients with initial value ≥ 0.3
Twenty patients were enrolled. Six (30%) patients developed AKI during first week of ICU stay. All six patients had initial Nephrocheck® values ≥ 0.3 that subsequently increased with the second Nephrocheck® test at 24 hours. Seven patients had normal Nephrocheck® values on admission and did not develop AKI. Interestingly, seven patients who had initial Nephrocheck® values ≥ 0.3 that decreased at 24 hours did not develop AKI.
The trend of the Nephrocheck® values was more significant than the initial value for the development of AKI. The Nephrocheck® test may be used as an early indicator of development of AKI. However, further studies are needed to determine if nephron-protective interventions can be helpful in mitigating this risk.
Renal biomarkers, Early detection, AKI, Critically ill, TIMP-2, IGFBP7, Nephrocheck®
The development of acute kidney injury (AKI) in critically ill patients has been associated with increased morbidity and mortality [1]. Various definitions have been developed to classify these patients according to severity of kidney injury for purposes of identification, management, and prognostication of the need for renal replacement therapy (RRT), re-hospitalization, and death. These scales include RIFLE (Risk, Injury, Failure, Loss, and End-stage Kidney Disease), AKIN (Acute Kidney Injury Network), and KDIGO (Kidney Disease: Improving Global Outcomes) [2]. However, these definitions use serum creatinine and urine output in a scoring system which reflect an insult or injury that occurred hours ago and do not reflect real time changes in renal function [1]. The lack of a gold standard in the diagnosis of AKI implies that we do not have any data on sensitivity and specificity of creatinine based definition of AKI. Currently, prevention and treatment strategies of renal injury and failure are limited due to the delay between the start of renal injury and rising creatinine or decreasing urine outputs. Earlier identification of acute kidney injury may lead to earlier interventions, which potentially could prevent the progression of the renal injury and improve patient outcomes.
Insults such as sepsis and respiratory failure are significant risk factors for the development of AKI in intensive care unit (ICU) patients [3-5]. Tubular injury leads to release of biomarkers which can be detected as a marker of AKI prior to a rise in creatinine or decrease in urine output. Neutrophil gelatinase associated lipocalin (NGAL), kidney injury molecule-1 (KIM-1), interleukin-18 are examples of biomarkers that have shown promising results [6].
In the face of injury, renal tubular cells use protective mechanisms such as de-differentiation and energy conservation, which includes arrest of the cell cycle in the G1 phase. Insulin-like growth factor binding protein 7 (IGFBP7) and tissue inhibitor of metalloproteinases-2 (TIMP-2) are inducers of G1 cell cycle arrest and can be detected in urine. The Nephrocheck® test system is a single use cartridge designed to detect IGFBP7 and TIMP-2. The results are obtained in 20 minutes with the use of Astute140 meter kit [7].
Using the Nephrocheck® kit, a feasibility study was performed to see if this test kit helped to identify patients with severe sepsis, septic shock, or respiratory failure at high risk to develop AKI. Those who were identified at risk where intervened on while these patients still normal kidney function by instituting a renal protective management bundle ("nephroprotective bundle") to prevent further development of AKI.
Patients with sepsis or respiratory failure admitted directly to an urban, university academic center ICU from the emergency department between November 2016 and March 2017. Exclusion criteria included age less than 18 years, patients with renal transplant, known acute kidney injury, creatinine greater than 1.5 mg/dl, and history of acute liver failure, cirrhosis or congestive heart failure. Patients meeting inclusion criteria had a urine sample collected within 4 hours of ICU admission for Nephrocheck® testing. Patients were risk stratified for propensity to develop AKI based on the results of the Nephrocheck®. Based on regulatory approval data, Nephrocheck values of < 0.3 were considered low risk, levels 0.3-1.5 intermediate risk and levels > 1.5 high risk for AKI development based on prior studies [5,8]. The Nephrocheck® kits were provided to our institution by Astute Medical, Inc. (San Diego, California). Due to logistical constraints, our laboratory agreed to perform the test between 8 am to 4 pm Monday to Friday. Because the Nephrocheck® kit was FDA approved, and resulted in no risk to the patient, the IRB granted waiver of consent since this test has already passed regulatory evaluation.
The clinical team was notified of the initial Nephrocheck® test result and renal protective guidelines were recommended based on the risk group (Figure 1). The ICU physicians received in-service education regarding acute kidney injury and renal protective strategies which constituted elements of the nephroprotective bundle. Elements of the nephroprotective bundle included avoidance of nephrotoxins, monitoring urine input and output with a Foley catheter, keeping mean arterial pressure > 65 mmHg, and utilizing fluids other than normal saline for hydration/resuscitation to reduce hyperchloremia. Additionally, in the high-risk group, a nephrology consultation as well as hemodynamic monitoring (such as an arterial line or Cheetah Nicom® bioimpedance monitor for noninvasive measurement of stroke volume and cardiac output) were advised. The ICU team was encouraged, but not mandated, to follow the guidelines. A second urine sample was collected at 24 hours for patients whose initial Nephrocheck® value was ≥ 0.3.
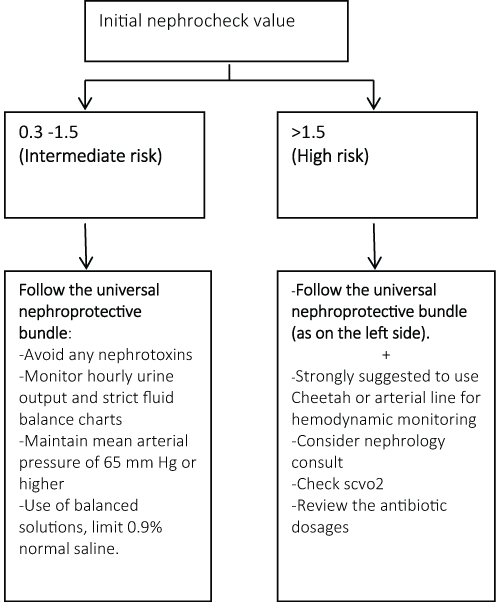 Figure 1: Elements of nephroprotective bundle based on risk of AKI.
View Figure 1
Figure 1: Elements of nephroprotective bundle based on risk of AKI.
View Figure 1
A retrospective chart review was performed to collect baseline demographic information including age, sex, Sequential Organ Failure Assessment (SOFA) score on admission, nephrotoxic drug exposure (angiotensin converting enzymes (ACE)-inhibitors, intravenous contrast, or nonsteroidal anti-inflammatory drugs (NSAIDs)), daily creatinine, ICU length of stay and hospital length of stay. Information was also collected on hypotension, use of vasopressors and blood pressure (BP) monitoring in the first 24 hours and use of renal replacement therapy (RRT) during first 7 days. AKI was defined as an increase in serum creatinine by ≥ 0.3 mg/dl (≥ 26.5 µmol/l) within 48 hours or an increase in serum creatinine to ≥ 1.5 times baseline within the previous 7 days as per KDIGO clinical practice guidelines [9].
Categorical variables were reported as numbers (percentages) and continuous variables as means (standard deviation). We used a non-parametric Mann Whitney U test for continuous variables and student t-test for categorical variables. We conducted univariate analysis of variance to study association of initial Nephrocheck® value with development of AKI. For analysis, the study population was divided into three groups-those with initial Nephrocheck® values < 0.3, initial Nephrocheck® value ≥ 0.3 with a decrease in second value at 24 hours, and initial Nephrocheck® ≥ 0.3 with an increase in second Nephrocheck® value at 24 hours. The three groups were also compared for AKI, outcomes including ICU length of stay and hospital length of stay.
Forty-six patients were screened during the enrollment period. Twenty-one patients were enrolled based on inclusion and exclusion criteria. One patient's urine sample was inadvertently discarded; therefore, 20 patients were enrolled with an average age 59 (range 35-96). Enrollment chart is shown in Figure 2. Initial Nephrocheck® values ranged from 0.04-1.52. Eleven (60%) patients had an initial Nephrocheck® value 0.3-1.5 (intermediate risk of AKI) and two (10%) patients had an initial value ≥ 1.5 (high risk of AKI). Of these 13 patients, 5 had a decrease in the second Nephrocheck® value and 6 had an increase in the second Nephrocheck® value. Two patients did not have a second Nephrocheck® value measured due to logistical issues. Table 1 shows a comparison of baseline characteristics and outcomes in patients with initial Nephrocheck® value < 3 vs. Nephrocheck® value ≥ 0.3.
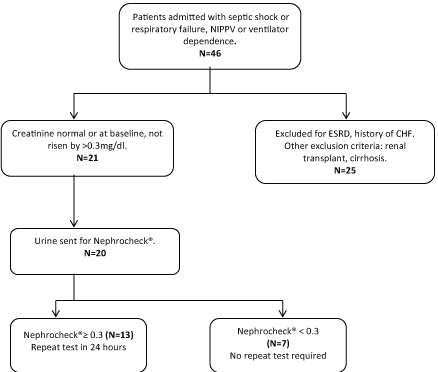 Figure 2: Enrollment flowchart.
View Figure 2
Figure 2: Enrollment flowchart.
View Figure 2
Table 1: Comparison of patients based on initial Nephrocheck® value. View Table 1
Six (30%) patients developed AKI during their hospital stay. All 6 had an initial Nephrocheck® value of ≥ 0.3 with an increase in the second Nephrocheck® value at 24 hours. Fifty percent (n = 3) patients had a creatinine increase on Day 2, while in the other 3 patients the Nephrocheck® elevation preceded the creatinine elevation. None of the patients with an initial value < 0.3 developed AKI.
An initial Nephrocheck® value ≥ 0.3 was significantly associated with development of AKI (p = 0.03). However, patients with initial values of ≥ 0.3 who had a decrease in the subsequent value at 24 hours did not develop AKI and therefore behaved similarly to patients with a low initial Nephrocheck® value of < 0.3. Figure 3 shows the creatinine trends in the first 7 days in the groups with initial Nephrocheck® value < 0.3, initial Nephrocheck® value ≥ 0.3 with a decrease in second value at 24 hours, and initial Nephrocheck® ≥ 0.3 with an increase in second Nephrocheck® value at 24 hours.
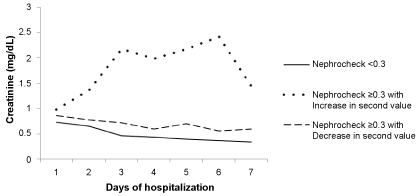 Figure 3: Comparison of outcomes of serum creatinine varies in subgroups based on initial and subsequent Nephrocheck® value.
View Figure 3
Figure 3: Comparison of outcomes of serum creatinine varies in subgroups based on initial and subsequent Nephrocheck® value.
View Figure 3
Only one patient required RRT in the first week of ICU stay (initial value was 1.1 and repeat at 24 hours was 2.7).
Nephrotoxins were successfully avoided in all patients after ICU admission. Three patients received intravenous contrast before their ICU admission. Only one of them had an initial elevated Nephrocheck® value of 0.38, which decreased to 0.28, and none of the three developed AKI. Three patients were on ACE inhibitors and they were stopped in all three patients. Similarly, NSAIDs were stopped on 2 patients.
An arterial line was used for blood pressure monitoring in 45% of patients (43% in Nephrocheck® value < 0.3 and 46% in Nephrocheck® value ≥ 0.3). Non-invasive monitoring of cardiac output using Cheetah NICOM™ bioreactance technology was used in the two patients with Nephrocheck® value ≥ 1.5. Nine patients (45%) had prolonged hypotension in the first 24 hours and that number was not significantly different in patients with initial values < 0.3 and ≥ 0.3 (43% vs. 46%, p = 0.8). History of hypertension, NSAID use, vasopressor use, intravenous contrast use, antibiotic dose adjustment, mode of blood pressure monitoring in the first 24 hours were not associated with changes in second Nephrocheck® value or the development of AKI (Table 2).
Table 2: Comparison of patients characteristics and outcomes based on trend of Nephrocheck® values. View Table 2
An initial Nephrocheck® value ≥ 0.3 had a trend in higher ICU length of stay (9.0 vs. 4.8 days, p = 0.07), but not hospital length of stay (12.4 vs. 12.2 days, p = 0.96) as compared those with Nephrocheck® value < 0.3 (Table 1).
In this study, the trend of the Nephrocheck® value was more significant than the initial value in the development of AKI. While none of the patients with a low value developed AKI, all patients who had an initially elevated value > 0.3 and an increase in value within 24 hours developed AKI. Interestingly, patients who had elevated initial Nephrocheck® values with a decrease in Nephrocheck® value at 24 hours did not develop AKI and therefore avoided AKI similarly to patients who had low initial Nephrocheck®.
It has been proposed that, in the face of injury, renal tubular cells use protective mechanisms for energy conservation such as cell cycle arrest to avoid replicating injured cells. Recent investigations have found a correlation between the presence of these biomarkers, and development of acute kidney injury [10]. The Nephrocheck® test evaluates two specific, early biomarkers: Insulin-like growth factor binding protein 7 (IGFBP7) and tissue inhibitor of metalloproteinases-2 (TIMP-2), which are involved in cell cycle arrest and can be found in the urine. Kashani, et al. found that risk of developing AKI, as defined by KDIGO, significantly increased when the Nephrocheck® level was > 0.3, and almost quintupled for levels > 2.0 [11]. These cut-offs were verified in the Opal Study [8] and the Topaz study [5].
In addition, patients that developed AKI had elevated Nephrocheck® levels before a rise in creatinine in approximately 50% of patients. This observation further emphasized the role of Nephrocheck® as a marker for earlier recognition of renal tubular injury compared to elevation of creatinine or oliguria and can enable earlier renal protective strategies such as optimization of hemodynamics, avoidance of nephrotoxins such as intravenous contrast and chloride-rich fluids, discontinuation of diuretics and ACE-inhibitors as appropriate, re-dosing of drugs such as antibiotics. This study was not intended to see to what degree these interventions mitigated AKI and therefore no conclusions can be made regarding the effectiveness of these interventions.
Thirty percent of the patients in this study admitted with sepsis or respiratory failure with normal renal function at admission developed AKI. Sepsis has been noted to be a common contributor for the development of acute kidney injury in ICU patients [3,4]. Dellepiane, et al. explored the mechanisms by which sepsis causes AKI, and found that it was multifactorial, with changes in renal microvasculature and perfusion, pathogen particle interaction with renal cells, and cytokine storm all contributed to development of AKI in sepsis [12].
Jeganathan, et al. found that there was a discrepancy in mortality rates (both ICU mortality and hospital mortality) between critically ill patients with ESRD and critically ill patients with new AKI; the overall mortality rates were significantly higher for patients classified with new acute kidney injury compared to patients without kidney injury and patients with ESRD [13]. These findings suggest that the increase in mortality in acute kidney injury is not caused solely by a loss in renal function, but is likely the result of a complex interplay of a variety of contributing factors, including the clinical circumstances causing the AKI and the immune response of the body to these circumstances [13].
This study has several limitations, the most important being the limited sample size. Size limitation was due to number of patients already presenting to the emergency department already with AKI. The sample size limited the implications of the conclusion to being hypothesis generating. Also, the nephroprotective bundle was only a suggestion and not a mandated protocol. Therefore, the degree to which specific interventions limited AKI could not be assessed. It is suspected that due to the pre-study education process conducted, there may have been cross-contamination of some of the interventions across all groups and initiation of measures due to the study being conducted (the "Hawthorne effect.").
The Nephrocheck® test on ICU admission and at 24 hours in critically ill patients at risk for AKI is an accurate early indicator of development of AKI. Further studies are needed to determine if nephroprotective interventions can mitigate this risk if instituted before occurrence of traditional markers of injury such as oliguria and elevation of serum creatinine.
Astute Medical, Inc. (San Diego, California) provided The Nephrocheck® kits to our institution. Astute Medical was not involved with study design, enrollment, or analysis.