We aimed to investigate whether a Blood Flow Restriction Training (BFR) prior to a High Intensity Interval Training (HIIT) leads to a higher increase in physical performance than a sole HIIT.
24 male participants (18 to 30 years) were randomly assigned to a BFR+HIIT or HIIT group which performed 3 times per week over 4 weeks a HIIT on bicycle-ergometers. Prior to each training session of the HIIT, both groups carried out 4 sets of deep squats without additional load but the BFR+HIIT group performed this exercise under BFR conditions. Immediately before and after the intervention period, the endurance capacity and strength capacity was assessed by a spiroergometry (parameter: maximal power, maximal oxygen consumption per kilogram body weight [VO2max/kg]) and a maximal strength test for the legs [1RM] as well as the Counter Movement Jump [CMJ]. The Growth Hormone (GH) and Insulin-like Growth Factor 1 (IGF-1) were measured prior to and after the first and last intervention. Blood lactate concentrations [La] were assessed immediately before and after deep squats as well as after the HIIT during the sixth training session.
The maximal power increased significantly in both groups (BFR+HIIT: + 9.5%, p < 0.001; HIIT: + 4.5%, p = 0.038) but to a higher extent in the BFR+HIIT group (interaction effect: F = 4.80, p = 0.039, ηp2 = 0.179). The VO2max/kg did not change due to the intervention of the BFR+HIIT and HIIT group. Regarding the 1RM and CMJ, a significant improvement in 1RM (p = 0.005) was observed only in the HIIT group. Both, GH and IGF-1 increased significantly immediately after the interventions, but without differences between groups (no interaction effect). La increased during the intervention in a comparable manner among both groups.
The combined intervention efficiently improves the maximal power in context of endurance capacity. Here, the underlying mechanisms have to be clarified in future investigations.
Blood flow restriction, Growth hormone, Insulin-like growth factor 1, Strength performance, Endurance performance
In the context of endurance training, High-Intensity Interval Training (HIIT) is a training method that involves repeated high to maximal physical exertions over a short time, interrupted by periods of rest or low-intensity exercise [1,2]. To design a HIIT, several exercise variables are available (e.g. intensity, duration and number of performed intervals, duration of recovery phases, design of recovery phases [active vs. passive]). These exercise variables can be designed in many different ways which, in turn, could provoke diverse physiological stimuli [3,4]. Therefore, the effectiveness of a HIIT with respect to physical performance improvement depends on the design of the exercise variables [3,5], making each HIIT unique [6].
Even though the underlying signal pathways are not yet known, hypoxia, metabolic stress, and mechanical stress, are crucial stimuli evoked by a HIIT [7-9] which activate mitochondrial, angiogenetic and cardiac adaptations [3-5,10,11]. Here, the magnitude of the stimuli depends, among other things, on the design of exercise variables mentioned above.
With the exception of mechanical stress, hypoxia and metabolic stress can also be evoked by Blood Flow Restriction Training (BFR) [12,13]. BFR describes a training method where the blood flow is restricted on the limbs of the body with elastic straps or blood pressure cuffs [14]. Special inflatable cuffs with pressure sensors named KAATSU-cuffs are also used [15]. BFR aims to evoke a local hypoxia which create metabolic stress in the restricted muscles [12]. Metabolic stress is the result of a high activity of anaerobic glycolysis for the resynthesis of Adenosine Triphosphate (ATP) and is characterized by the subsequent buildup of metabolites such as lactate, hydrogen ion, inorganic phosphate, and creatine. In turn these lead to an increase in hormones (e.g. testosteron, growth hormone [GH], insulin-like growth factor 1 [IGF-1]), myokines (e.g. interleukin 6 [IL-6]), reactive oxygen species (ROS), cellular swelling, and muscle fibre recruitment [16,17]. These mediators trigger signaling pathways that evoke adaptation processes enhancing physical performance (e.g., on mitochondrial and angiogenetic level) [16]. To increase the extent of metabolic stress during BFR, low-intensity exercises (strength or endurance training) at a high volume are performed under these conditions [13,14,18].
Because of the mentioned comparable physiological stimuli, we assume that a successive application of these two types of interventions may catalyze adaptation effects enhancing physical performance more than a sole HIIT.
This assumption is reinforced by the investigation by Taylor, et al. [9]. Taylor, et al. [9] combined a HIIT with a postexercise BFR (HIIT+BFR) and showed that the hypoxia-inducible factor 1α (HIF-1α) and the maximal oxygen uptake (VO2max) increase to a higher extent than HIIT alone. The HIF 1α is a hypoxia-induced master regulator adapting the oxygen homeostasis which supports the transport, allocation, and utilization of oxygen in the organism [19]. The HIF-1α is also the origin for many molecular signaling pathways because of the regulation of a wide variety of transcriptional factors [20]. For example, the peroxisome proliferator-activated receptor gamma coactivator 1-alpha (PGC-1α) and the vascular endothelial growth factor (VEGF) increase due to the expression of the HIF-1α [21-23]. While the PGC-1α is the master regulator of mitochondrial biogenesis [24], the VEGF is crucial for angiogenetic adaptations [25]. Both, the PGC-1α and the VEGF increased in the investigation by Taylor, et al. [9] in the group conducting a HIIT+BFR as well as a sole HIIT. However, the enhanced HIF-1α in the HIIT+BFR as compared to the HIIT suggests that the combined intervention may have a superior physiological stimulus.
Based on the presented theoretical background and the insights of the investigation by Taylor, et al. [9], the purpose of this study was to investigate the effects of a HIIT in combination with BFR (using KAATSU-cuffs) in comparison to a sole HIIT on physical performance. Therefore, we have investigated a combined intervention which comprises a BFR for the legs immediately before a HIIT. It is to be assumed that this intervention leads to higher metabolic stress, which could catalyze adaption processes in this context. To clarify the extent of metabolic stress, the accumulation of blood lactate concentrations (La) during the intervention as well as acute and basal changes of the GH and IGF-1 have been measured. BFR and/or HIIT can evoke adaptations for different subsystems that are crucial for physical performance (e.g. cardiopulmonary, metabolic, and muscular systems). The aerobic, anaerobic, and maximal endurance capacities as well as the strength capacity of the legs were examined.
24 young male participants (age: 18 to 30 years) with no known cardiovascular or musculoskeletal disorders were enrolled in this study. Prior to beginning the study, all participants were provided with information about the purpose of the investigation and the study design before signing a written informed consent (according to the Declaration of Helsinki). Thereafter, they were randomly (permuted block randomization, proportion 1:1, block size = 4) assigned in two groups: BFR+HIIT and HIIT.
The groups BFR+HIIT and HIIT performed a HIIT-intervention for four weeks, three times per week (Monday, Wednesday, Friday). Immediately prior to each HIIT-intervention, four sets of deep squats without additional load were performed by both groups. The BFR+HIIT group conducted the deep squats under BFR conditions.
Within one week before (pre) and after (post) of the four-week intervention, the endurance capacity was tested using a spiroergometry on a bicycle-ergometer. Strength capacity of the legs was tested with a counter movement jump (CMJ; [26]) and the one repetition maximum for the exercise leg press (1RM; [27]). The GH and IGF-1 were analysed immediately before and after the first (T1,T2) and last (T3,T4) intervention to quantify acute (T1 to T2 and T3 to T4) and basal (T1 to T3) changes. During the sixth intervention, the La were measured immediately before (pre) and after the BFR/squat (post BFR/squat) and after the HIIT (post HIIT). The group allocation of the participants was blinded for all investigators who conducted the pre- and post-tests. The data analysis was also blinded.
We made sure to adhere to the key considerations to conduct BFR described by Nakajima, et al. [28]. We used special inflatable KAATSU-cuffs (flexible pressurizing cuffs with pressure sensors, cuff width 5 cm; see [15]) which were applied proximally on the legs with an initial pressure of 49 ± 5 mmHg (the initial pressure results from the initial tightness of the cuffs when these were applied manually on the limb and fixed with the velcro; the mean value of initial pressure was calculated after the intervention period) [29]. Next, the individual optimal pressure (IOP) of the KAATSU-cuffs was obtained by using an alternate inflation and deflation of the cuffs for 20 and 10 seconds. To identify the IOP, we incrementally increased the pressure on the KAATSU-cuffs until a capillary refill time of 3 seconds for the legs was reached (tested on the vastus medialis of the M. quadriceps femoris). The mean IOP here was 273 ± 33 mmHg (mean IOP of all participants over the four weeks; the mean value was calculated after the intervention period). The procedure of alternate inflation and deflation of the cuffs is called KAATSU-cycle: start pressure: 140 mmHg; incremental pressure: 20 mmHg; number of cycles: at least 7 cycles (cf. [30,31]). This procedure was completed prior to each intervention to determine the IOP for every intervention for the BFR. The IOP allows arterial blood flow but not venous blood flow which leads to a venous blood pooling [32]. In order to control the initial pressure and IOP as well as the inflation and deflation of the cuffs, the special device KAATSU-Master was used (see [15]); regarding the described procedure to determine the IOP and conduct the KAATSU-cycle see the KAATSU user manual [33]. Immediately after completing the KAATSU-cycle, the participants of the BFR+HIIT group performed the four sets of deep squats with the KAATSU-cuffs. Thereafter, the KAATSU-cuffs were removed and the HIIT was started.
The exercise variables for performing deep squats are aligned to the recommendations for conducting a BFR by KAATSU GLOBAL INC., see [33]. In this regard, it is recommended to perform BFR-exercises with ≥ 3 sets, low loads, a high number of repetitions, and short inter-set resting periods to ensure an effective stimulus of BFR for muscular adaptations leading to an increased physical performance [17,33]. The evaluation of an effective load will be reached by the observation of muscular failures or technical failures at the end of the sets. If one or both of these crucial factors are achieved, there will be a reasonable strain (load) for the given exercises. Here, the load can be applied by the body weight, resistance bands, weights (like dumbbells or a barbell), etc. [33].
In the current study, the participants of both groups performed four sets of deep squats without additional load (body weight) with 40, 25, 20 and 15 repetitions. The decreasing number of repetitions per set with a high number of repetitions in the first set is based on the practical hints for conduction a BFR by Abe and Sato [34], Scott, et al. [35], and the above-mentioned recommendations of KAATSU GLOBAL INC. [33]. The inter-set resting periods was 20 seconds. We have observed that all participants reached muscular or technical failures at the end of the third and/or fourth set of deep squats. Therefore, no additional load was needed. In order to ensure that all participants showed comparable movement velocities, we utilized a metronome with 50 bpm (2 beats per repetition, 1 beat for the eccentric and concentric phase, respectively).
After having completed the deep squats, the participants started with the HIIT. This was carried out on bicycle-ergometers (KardiomedBike, Proxomed®, Germany) and consisted of three intervals each lasting four minutes with a resting period of one minute. The intervals were performed with an intensity which was adjusted to the second ventilatory threshold plus five percent (BFR+HIIT HR: 168 ± 14 min-1 [90.5 ± 7.0% HRmax]; HIIT HR: 163 ± 15 min-1 [87.2 ± 8.2% HRmax]), with heart rate (HR) as the control parameter (measured by the heart rate monitor FT7, Polar®, Finland). This intensity was chosen because of the criterion that a HIIT must be performed at an intensity greater than the anaerobic threshold [1]. The cycling cadence was predetermined from 80 to 90 rpm (according to the cadence which was used in the pre-test of the spiroergometry, see below).
In order to check the effectiveness of the used intervention on the endurance capacity as well as to identify the individual training intensity for the HIIT, the participants were asked to conduct an incremental load test (initial load 50 W, 50 W was added every 3 minute, cycling cadence was 80 to 90 rpm) on a bicycle (Xrcise Cycle Med, Cardiowise, Germany) in combination with a spirometry (MetaMax®3B, CORTEX, Germany). The test was performed until physical exhaustion under control of vital parameters by a medical doctor (e.g. heart rate, blood pressure).
The first and second ventilatory threshold (VT1, VT2) were determined by the V-slope method with the parameters oxygen uptake (VO2) vs. carbon dioxide production (VCO2) and minute ventilation (VE) vs. carbon dioxide production (VCO2) with regard to Meyer, et al. [36].
The CMJ was performed to examine neuromuscular changes of the legs caused by the intervention [37]. Therefore, a contact mat (SpeedCourt®, Global Speed, Germany) was used and three CMJ with a resting phase of two minutes were performed. For the pre-post comparison, the main values of the height of the three CMJ were calculated.
The 1RM was determined using the multiple repetition maximum test as described by Reynolds, et al. [38]. The test was assessed with the exercise dynamic leg press.
Blood samples were collected by a medical doctor at the above-mentioned time points (T1, T2, T3, T4) from a superficial forearm vein under stasis conditions. Here, a 5 ml blood sample was drawn to determine GH and IGF-1, respectively. The blood samples were analyzed in a local medical laboratory.
La was measured on the ear lobe of the participants to the time points as mentioned in the study design. The samples were analysed with the measuring device Super GL3 by HITADO (Germany; measuring error < 1.5% according to the manufacturer’s information).
The data were tested for normal distribution using the Kolmogorov-Smirnov-Test. For normally distributed data, the interaction effect between the groups over the intervention time was checked with a two-way ANOVA with repeated measures (factors: time x group). Thereafter, differences between measurement time points within a group (time effect) and differences between groups during a measurement time point (group effect) were analysed with a dependent and independent t-test. A one-way ANOVA with repeated measures was performed with a post-hoc test with Bonferroni correction to identify time effects between the measurement time points regarding the La. All analyses were done using the statistical computer program SPSS Statistics 24 (IBM®, Germany) and the level of significance was set to α = 5%. In conjunction with the interaction effects, main time effects, and main group effects, the effect size partial eta squared (ηp2) were reported [39].
In each of the groups (BFR+HIIT and HIIT) 12 participants with comparable characteristics were included: BFR+HIIT: age: 23.4 ± 3.3 years, weight: 84.1 ± 10.9 kg, height: 182.0 ± 4.6 cm, VO2max: 47.6 ± 8.5 ml/kg/min; HIIT: age: 22.6 ± 2.9 years, weight: 77.4 ± 9.8 kg, height: 180.2 ± 5.4 cm, VO2max: 48.8 ± 4.7 ml/kg/min).
Outcome values regarding the endurance capacity are shown in Table 1. The participants of both groups did not differ in their maximum endurance capacity (referring to the pre-test) regarding the maximal power and the maximum oxygen consumption per kilogram body weight (VO2/kg). Therefore, the groups can be considered homogeneous at the beginning of the intervention.
Table 1: Mean values (standard deviation) of parameters of endurance and strength performance collected in the pre- and post-test in the BFR+HIIT group and HIIT group. View Table 1
After the four weeks of intervention, we determined a significant increase in the maximal power in both groups with the increase in the BFR+HIIT group being approximately twice as high as in the HIIT group (see interaction effect in Table 1). Furthermore, after the intervention period, the power was also higher during the VT1 and VT2 in both groups. But in the BFR+HIIT group, the increase in power during the VT1 was much higher than in the HIIT (see Table 1). These results did not become statistically significant but for the BFR+HIIT group, a tendency (0.100 > p > 0.050) was observed. Moreover, the improvements can be considered practically relevant. Regarding the development in power of training intensity during each week of the intervention, the increases were considerably greater in the BFR+HIIT group (see Figure 1). While the BFR+HIIT group was able to enhance their power with constant HR (referring to the VT2 + 5%, see methods) to + 8.5% (1. to 2. week, p < 0.001), + 8.9% (2. to 3. week, p < 0.001) and + 4.0% (3. to 4. week, p = 0.001) as well as overall to + 23.7% (1. to 4. week, p < 0.001), the enhancement of the power in the HIIT group was only + 5.3% (1. to 2. week, p = 0.049), + 5.2% (2. to 3. week, p = 0.023) and + 3.5% (3. to 4. week, p = 0.210) as well as overall + 14.7% (1. to 4. week, p < 0.001). The different developments between the groups have just marginally failed a significant interaction effect (F = 2.65, p = 0.071, ηp2 = 0.076).
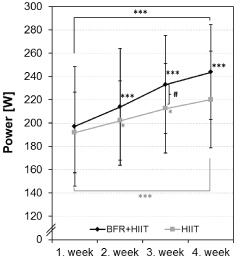 Figure 1: Mean values of the training intensity (power) during the High-Intensity Interval Training (HIIT) of the BFR+HIIT group and HIIT group over the four weeks of intervention. The training intensity was adjusted to the heart rate on the second ventilatory threshold plus five percent which were determined prior to the intervention period (time effects, significant different to the prior week: *p ≤ 0.050; **p ≤ 0.010; ***p ≤ 0.001; group effects, significant difference between groups BFR+HIIT and HIIT: #p ≤ 0.050; ##p ≤ 0.010; ###p ≤ 0.001). View Figure 1
Figure 1: Mean values of the training intensity (power) during the High-Intensity Interval Training (HIIT) of the BFR+HIIT group and HIIT group over the four weeks of intervention. The training intensity was adjusted to the heart rate on the second ventilatory threshold plus five percent which were determined prior to the intervention period (time effects, significant different to the prior week: *p ≤ 0.050; **p ≤ 0.010; ***p ≤ 0.001; group effects, significant difference between groups BFR+HIIT and HIIT: #p ≤ 0.050; ##p ≤ 0.010; ###p ≤ 0.001). View Figure 1
Except for a significantly lower HR at the VT1 of the BFR+HIIT group and the significantly lower maximal HR of the HIIT group, there were no significant changes in the HR or VO2/kg during maximal or submaximal workload (VT1, VT2) in both groups.
The values of the CMJ and the 1RM are shown in Table 2. With the exception of a significant increase in the 1RM in the group HIIT, no interaction effects, time effects, or group effects have arisen regarding the 1RM and CMJ for either group.
Table 2: Mean values (standard deviation) of the growth hormone and the insulin-like growth factor 1 immediately bevor and after the first (T1, T2) and last (T3, T4) intervention in the BFR+HIIT group and HIIT group. View Table 2
Outcome values of the GH and IGF-1 are shown in Table 2. Both, in the first (T1 to T2) and the last intervention (T3 to T4), the GH and IGF-1 increased significantly (acute reaction, see time effects in Table 2). There is a greater increment (absolute and relative) in IGF-1 in the BFR+HIIT group but no significant differences (but a tendency) were observed between groups (see interaction effects in Table 2).
In both groups, the basal levels of the GH have not been changed by the intervention (T1 to T3, time effects: BFR+HIIT p = 0.750; HIIT p = 0.122; interaction effect: F = 1.91, p = 0.181, ηp2 = 0.080). For the IGF-1, we observed a significant decrease in the basal levels (T1 to T3, time effects: BFR+HIIT p = 0.018; HIIT p = 0.006) but no difference between the groups was found (T1 to T3, interaction effect: F = 0.03, p = 0.861, ηp2 = 0.001).
Within the respective interventions accumulated the La in a comparable manner: BFR+HIIT: pre 0.99 ± 0.22 mmol/l, post BFR/squat 5.17 ± 1.54 mmol/l, post HIIT 7.72 ± 2.45 mmol/l; HIIT: pre 1.05 ± 0.34 mmol/l, post squat 4.08 ± 1.32 mmol/l, post HIIT 8.06 ± 2.18 mmol/l (Interaction effect: F = 1.44, p = 0.248; ηp2 = 0.062). The increase in the La over the three measurement time points was significant (main time effects: BFR+HIIT: F = 54.39, p < 0.001, ηp2 = 0.832; HIIT: F = 66.79, p < 0.001; ηp2 = 0.859). Between the groups, no differences were observed (group effects: pre p = 0.630; post BFR/squat p = 0.077; post HIIT p = 0.719).
We conducted this investigation assuming that a combined intervention with a BFR prior to a HIIT would lead to a higher metabolic stress as compared to a sole HIIT whereby adaptation processes will be catalysed and physical performance enhanced. Based on the results presented above, our main findings are: i) The combined intervention (BFR+HIIT) does not lead to a higher metabolic stress than a sole HIIT regarding the La as well as the acute changes of the GH and the IGF-1, ii) BFR+HIIT leads, in comparison to a sole HIIT, to an improvement that is twice as high in physical performance regarding the maximum power on a bicycle ergometer, and iii) The strength performance of the legs (vertical jump high in CMJ, 1RM for leg press) is not affected by a BFR+HIIT.
Regarding main finding i): These results differ from the general assumption that low-intensity strength exercises with BFR lead to a superior accumulation of La and therefore to a higher metabolic stress than strength exercises without BFR [12,40]. It could be that our chosen measurement time points caused the comparable La. It is known that only after 3 to 8 minutes, exercise peak values of La do occur [41]. Therefore, the time interval between BFR (and perhaps also HIIT) and the measurement of the La may have been too short to quantify differences (which however would have been quantifiable at a later point in time). However, it can also be assumed that the conditions of BFR (pressure, type of cuff) [42] in combination with the exercise variables (number of repetitions, resting periods, number of sets, load) did not lead to a superior metabolic stress (even though we have followed the recommendations of KAATSU GLOBAL INC. regarding the conditions of BFR and the exercise variables, see [33]).
In addition to our main finding ii), we observed that power on the bicycle was also improved at the VT1 and VT2 (no significant, but tendential time effects, respectively). Since higher metabolic stress did not occur, other physiological stimuli must have been responsible for adaptation processes that have increased physical performance. Here, we can discuss the following two adaptation processes only speculatively: Firstly, based on the theoretical background outlined in the introduction section, it can be supposed that the BFR may have provoked a hypoxia in the muscles of the legs which temporarily enhanced the HIF-1α. In consequence, HIF-1α-associated adaptation processes, like mitochondrial biogenesis or angiogenesis, were evoked. Due to these adaptations, the metabolism might have been improved which, again, led to a higher physical performance during the bicycle test. Secondly, BFR can increase the stimulation of group III and IV afferents which enhance the recruitment of fast twitch fibres [43]. Because of the repeated application of the BFR, it could be that the corticomotor excitability increases and as a consequence, the force capacity of the neuromuscular system could be positively influenced [13]. Neuromuscular adaptations may explain the continuous increase in power of training intensity during each week of the intervention as well as the increased power observed in the post-test during the incremental load test (compared to the pre-test) in the BFR+HIIT group. Furthermore, it is known that fast twitch fibres have a predominant energy metabolism with glycogen consumption [44]. The enhanced recruitment of fast twitch fibres may have led to higher glycogen depletion in the BFR+HIIT group. The repeated glycogen depletion has the potential to modulate numerous adaptive processes in the skeletal muscle, like mitochondrial biogenesis [45]. We suppose that due to hypoxic and/or neuromuscular stimuli, the mentioned subsequent signalling pathways were activated, and the metabolic and/or neuromuscular adaptations might be the reason for the improved endurance capacity during the incremental load test on the bicycle. However, the aforementioned adaptation processes would still have to be elucidated.
Our main finding iii) seems to be unusual for of two reasons: first, improvements in strength would have been expected at least as a result of BFR [46] and second, neuromuscular improvements would have been expected to enhance strength capacity as above assumed. Regarding the strength improvements, it is possible that the interference between the signaling pathways of a resistance training (BFR) and endurance training (HIIT) are the reason for the absence of performance increases [47]. With respect to the jumping performance, there are also investigations which did not observe enhanced jumping performance although, simultaneously, the 1RM was enhanced after a BFR over 10 weeks (twice a week) [48]. Here, a lack of neuromuscular adaptations was discussed to be the cause. This standpoint would argue against our statement assuming that neuromuscular adaptations might have occurred which improved the endurance capacity. However, improvements in physical performance caused by enhanced strength through neuromuscular adaptations are heterogeneous and depend for instance on the type of exercise, load, and velocity [49,50]. With this in mind, the intervention in this study may have led to specific (neuromuscular) adaptations of the muscles as result of the type of exercise which was performed during the intervention (performance on the bicycle). These adaptions do not seem to have had a transfer effect on the exercise characteristic of the 1RM and CMJ. The significant improvement in the 1RM in the HIIT group that is almost twice as much higher than in the BFR+HIIT group (+ 7.3% vs. 4.0%) is surprising to us and we cannot explain this performance enhancement.
Perhaps the above mentioned or other/further adaptation processes are the reasons explaining that the coupled intervention BFR+HIIT is more efficient than the HIIT regarding the improvement of the power on the bicycle. The high efficiency of this coupled intervention becomes clear when the effects will be compared with those of other HIIT protocols in the literature: After only 4 weeks of BFR+HIIT, we observed that the endurance capacity (with respect to the maximal power: + 10%) had been increased as much as other researchers achieved after 12 weeks of sole HIIT (see Sylta, et al. [51]). In order to apply specific stimuli aiming to efficiently increase endurance capacity, the use of this coupled intervention may thus be of particular interest with respect to programming and periodization of training [52]. For this, however, adaptation processes of BFR+HIIT must be investigated in more profound detail. Furthermore, the use of BFR immediately prior to a HIIT must be examined in conjunction with different protocols of HIIT because modified exercise variables of HIIT might elicit various adaptations.
As strength of the investigated coupled training method BFR+HIIT, we would like to mention the practicability and effectiveness with regard to the improvement of the endurance performance. The additional use of a BFR (according to KAATSU GLOBAL INC., [33]) with a commonly used HIIT protocol took a little more time (approximately 10 Minutes) but has increased the endurance performance approximately twice as much as a sole HIIT.
However, the present study is not without limitations. Even though the exercise variables of the BFR are in line with the recommendations of KAATSU GLOBAL, there is currently no consensus about the exercise variables of a BFR (such as optimal cuff pressure, cuff material, cuff width; see [42,53,54]). Therefore, the effects investigated may not be reached with other BFR methods/protocols. Furthermore, we were not able to elucidate the underpinning physiological adaptation processes in conjunction with the coupled training method. With regard to these limitations, further investigations are needed to focus on clarifying i) The efficient design of excise variables regarding BFR in context of this coupled intervention approach and ii) Adaptation processes that lead to performance improvements.
In summary, this is the first study which investigated an intervention consisting of a BFR immediately prior to a HIIT (BFR+HIIT) in comparison to a sole HIIT with the aim to improve physical performance. We observed that the coupled intervention does not lead to superior metabolic stress (evaluated on the basis of the accumulation of the blood lactate concentration, growth factor, and the insulin-like growth factor 1) than a sole HIIT. The BFR+HIIT and the sole HIIT increase the maximal power on a bicycle. But BFR+HIIT enhances the maximal power approximately twice as much as a HIIT. Moreover, there was enhanced power during the ventilatory threshold 1 and 2 with a practical relevance (whereas the significance level was only just failed).
Although the underlying mechanisms of adaptations in response to the intervention BFR+HIIT are not yet fully understood, this study provides first insights suggesting that this type of intervention may be valuable for an enhancement of physical performance in an effective way. Therefore, further investigations which focus the elucidation of underlying mechanisms of this coupled intervention are recommended.
AT, MB, MK, TB and LS conceived and designed research; AT, MB and MK performed experiments; AT, MB and TB analyzed data; AT, MB, and TB interpreted results of experiments; AT prepared figures; AT drafted manuscript; AT, MB, MK, TB and LS edited and revised manuscript; AT, MB, MK, TB and LS approved final version of manuscript.
No external financial support.