The effects of 7 days of "Dry" water Immersion (DI) with Neuromuscular Electrical Stimulation (NMES) on the function and architecture of the human triceps surae muscle were investigation in six young men.
During DI performed NMES muscle of both lower extremities. Architectural properties of the triceps surae muscle were determined in vivo. The ankle was positioned at 15° dorsiflexion (-15°), 0, +15, +30° plantar flexion, with the knee set at 90°. At each position, longitudinal ultrasonic images of the Medial (MG) and Lateral (LG) Gastrocnemius and Soleus (SOL) muscles were obtained while the subject was relaxed (passive) and performed 50% of the maximal voluntary plantar flexion (active), from which fascicle lengths (Lf) and angles (Θf) with respect to the aponeuroses were determined.
The maximal plantar flexion torque was increased by 11.3% after DI with by NMES-training. In the passive condition, Lf changed from 36, 47, 39 to 27, 31, 28 mm; Θ changed from 31, 20, 23 to 49, 29, 34° for MG, LG, and SOL, respectively. After DI in the passive condition, Lf decreased by 16, 37, 24; Θf increased by 38, 35, 34% for MG, LG, SOL, respectively.
Results suggest that DI decreased architectural the triceps surae muscle and these changes might protect from a larger loss of muscle force.
"Dry" water immersion, Neuromuscular electrical stimulation, Ultrasonography, Muscle architecture, Triceps surae muscle, Muscle contraction
A number of studies have indicated that sudden exposure to microgravity environment causes a decrease in the tone of the skeletal muscles [1,2], reduction of muscle strength [3-5], perceptual and coordination disorders in the neuromuscular systems [3,6], shift of the spinal reflex mechanisms [7] and degradation of joint position sense [8,9]. It is accepted that the major factor responsible for all of these changes is the sudden elimination of the proprioceptive information from the muscle and tendon in response to absence of load-bearing. Accordingly, the anti-gravitational or the postural muscle should be the principal target for the action of unloading.
Among the various methods used to model the effects of weightlessness "Dry" water immersion [10] is the gold standard to simulate the neuromuscular changes induced during short-term space flight [6,7,11-19]. It is well that unloading of the musculoskeletal system of humans and lower mammals due to actual or simulated spaceflight associated with extensive loss of muscle mass in the extensors and the least loss in the flexors [20,21] and lead to muscle wasting and weakness [22,23]. These differences are based on the distribution of muscle fiber types. In experiment show that weightlessness simulated by water immersion changed the recruitment order of motor units during isometric contraction in the hip flexor muscles [6,15], а significant reduction in muscle fiber conduction velocity [11,12]. Moreover, shown that weightlessness simulated by water immersion significantly decrease in conduction velocity of potentials on slow motor units (muscle fiber) without changes in fast motor units [11,12]. Principal cause of these changes can be sharp decrease in muscular activity. It is known that weightlessness simulated causes development of changes basically in muscular fibres of tonic type [24].
For instance, it has been shown that immersion for 7 day significantly reduces isometric Maximal Voluntary Contraction (MVC) by ~40% in the ankle plantar flexors (Triceps Surae-TS) [16,17,25] and isokinetic concentric by ~20% in the knee extensors [13,14] and by ~34% in the ankle plantar flexors [23,24] and by ~20% in the ankle dorsi flexion [13]. The magnitude of these changes is similar to that observed in spaceflights studies of similar duration, in which MVC (maximal torque) reductions of 30% and 20%, respectively, have been found in the ankle plantar flexors, and the ankle dorsi flexors [13] and by ~11% in the knee extensors [26].
Gravitational loading appears to be necessary for the maintenance of human lower limb skeletal muscle force [4,27-30]. The muscle groups most affected by exposure to microgravity appear to be the antigravity extensors of the knee and ankle [20,31]. Among these, the plantar flexors seem to be the most affected [20,31] likely due to their greater mechanical loading under normal gravitational conditions. Most notable after exposure to microgravity is a disproportionate loss of force as compared to that of muscle size [20,32] indicating that factors other than atrophy contribute to muscle weakness.
However, it should be noted that in all these researches it is ignored changes in architecture of a muscle. This is despite the fact that most muscles of the human body are pennate [33]. The internal architecture of a muscle is an important determinant of its functional characteristics [34].
Studies simulating microgravity have shown that exercise countermeasures can attenuate, but not completely prevent the loss of muscle mass and force [25,31,32]. Moreover the limitation for active muscle training during a long-term space mission in term of time and space required the consideration of automatic support. EMG amplitude decreased in the SOL, and increased in the MG and VL [35,36]. EMG examinations have revealed a significant shift toward a higher frequency spectrum, which means that a greater reduction in slow-twitch muscle fiber (type I) was found compared to fast-twitch fibers (type II). Thus adaptations observed post-simulation were different from those observed post flight, indicating that there was a response to the microgravity environment. Peak MG tendon force amplitudes were approximately two times greater post-compared with pre-flight or pre-simulation. Adaptations in tendon force and EMG amplitude ratios indicate that the nervous system undergoes a reorganization of the recruitment patterns biased toward an increased recruitment of fast (type II) versus slow (type I) motor units and flexor versus extensor muscles. In this connection long-term Neuromuscular Electrical Stimulation (NMES) can be one of means of preventive maintenance of slow-twitch muscle fibers.
NMES produces skeletal muscle contractions as results of the percutaneous stimulation of peripheral nerves. Clinically, the use of NMES has been shown to potentially improve or compensate for disadvantages in disabled or chronic patients with physical inactivity. In fact, NMES of skeletal muscles mау also increase the strength and endurance of their paralyzed muscles during daily activity [37].
Therefore, the purpose the purpose of the present study was to investigate the internal architecture of the triceps surae muscle [Medial (GM) and Lateral (LG) Gastrocnemius and Soleus (SOL) muscles] in relation to the functional characteristics of the plantar flexors after 7 days of «Dry» water Immersion (DI) with use exercise countermeasures (NMES-training).
Six healthy males aged 20 to 24 years (22.3 ± 0.6) volunteered for the study. Their average height and mass were 1.78 ± 0.4 m, and 78.3 ± 3.4 kg (means ± SD), respectively.
Selection of subjects was based on a screening evaluation that consisted of a detailed medical history, physical examination [1], complete blood count, urinalysis, resting and cycle ergometer electrocardiogram, and a panel of blood chemistry analysis, which included fasting blood glucose, blood urea nitrogen, creatinine, lactic dehydrogenase, bilirubin, uric acid, and cholesterol. All of the subjects were evaluated clinically and considered to be in good physical condition. No subject was taking medication at the time of the study, and all subjects were nonsmokers.
None of them had а habit of exercise a regular basis. Prior to the experiment, details and possible risks of the protocols were explained to the subjects, and written informed consent was obtained from each of them. The experimental protocol was approved be the Russian National Committee on Bioethics of the Russian Academy of Sciences and was in compliance with the principles set forth in the Declaration of Helsinki.
In the week preceding the DI period the participants were asked to visit the laboratory on several occasions, to become familiar with testing procedures. All baseline data were collected in the week before starting the DI. Subsequent measurements were performed immediately after the end of the DI.
On day 0, all participants completed the evaluation session (pre-session) (Figure 1), which consisted of a full set of tests, including isometric maximal effort and the assessment relationships between joint angles and muscle architecture (lengths and angles of fascicles) of human Triceps Surae muscles (TS) in vivo in passive (relaxed) and active (contracting) conditions. Then, the NMES group underwent the NMES training program conducted one times per week for 1 week (Figure 1). Then subjects were placed in an immersion bath. Each subject served as his own control.
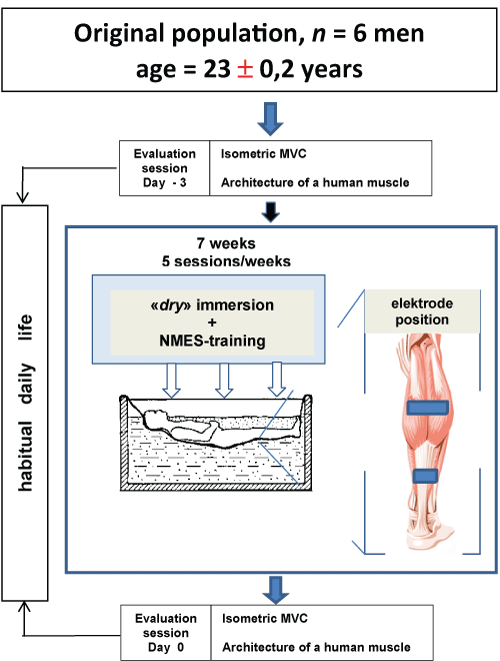 Figure 1: Experimental design. All of the enrolled participants were older than 20 years and have never used NMES. Seven days separated the two evaluation sessions. The main objectives of the evaluation sessions were the assessment of some intrinsic muscle and architecture properties during DI as well as the properties during a NMES task. NMES-training of muscles of the examinee was carried out directly in a bath. The intensity level stimulation is determined by a threshold of bearableness of subjects. NMES-training continued for six days, during which daily five days on end (from Monday to Friday inclusive) including one day of rest (Saturday). During DI, subjects executed а NMES-training during 3 hours per day with 1 s « оn » and 2 s « off » with a frequency of 25 Hz and amplitude of stimulus from 0 up to 45 V for training. View Figure 1
Figure 1: Experimental design. All of the enrolled participants were older than 20 years and have never used NMES. Seven days separated the two evaluation sessions. The main objectives of the evaluation sessions were the assessment of some intrinsic muscle and architecture properties during DI as well as the properties during a NMES task. NMES-training of muscles of the examinee was carried out directly in a bath. The intensity level stimulation is determined by a threshold of bearableness of subjects. NMES-training continued for six days, during which daily five days on end (from Monday to Friday inclusive) including one day of rest (Saturday). During DI, subjects executed а NMES-training during 3 hours per day with 1 s « оn » and 2 s « off » with a frequency of 25 Hz and amplitude of stimulus from 0 up to 45 V for training. View Figure 1
DI was used to simulate microgravity as described bу Shulzhenko and Vil-Villiams [10]. Each subject was positioned horizontally in а special bath оn fabric film that separated him from the water (Figure 1). During the DI, the subjects remained in a horizontal position (an angle which make the body and horizontal line, e.g. 5° head-up position) continuously for all including excretory function and eating. The water temperature was constant (33.4 ℃) and maintained automatically at this level throughout the experiment. The duration of the DI was 7 days.
А nursing staff was present for subjects transportation, maintenance of hygiene including toilet and shower, provision of food and medical care, as well as support of subjects needs within the constraints of the protocol. Тhe subjects were supervised 24 h⋅d-1.
Тhе subjects sat оn а couch with the back supported and the lower limbs fully extended. Аll measurements were carried out on the right leg, dominant in all the subjects, with the foot positioned at 90° relative to а footplate (Figure 2A). The foot of the limb studied was relatively rigidly fixed on а special platform of the isokinetic dynamometer («Biodex», USA). All measurements were carried out with the knee joint flexed at 60°. Subjects performed a series а standard warm-up and three to five submaximal of isometric plantar flexion contractions on an isokinetic dynamometer at ankle angles of 0° (neutral anatomic position: the footplate of the dynamometer perpendicular to the longitudinal axis of the tibia). Subjects performed of the plantar flexors following instructions to "push down as if you were pressing a gas pedal, increase force to a maximum" with the subsequent measurement of the maximal torque moment (Maximum Voluntary Contraction - MVC). Each subject performed between two and four MVCs. There was a 1-min rest between the set. In each set, the contractions were performed at a rate of 0.5 s-1. Subjects were given visual feedback of the target and elicited force on a computer screen. The MVC was determined as the highest value of voluntary force recorded during the entire contraction.
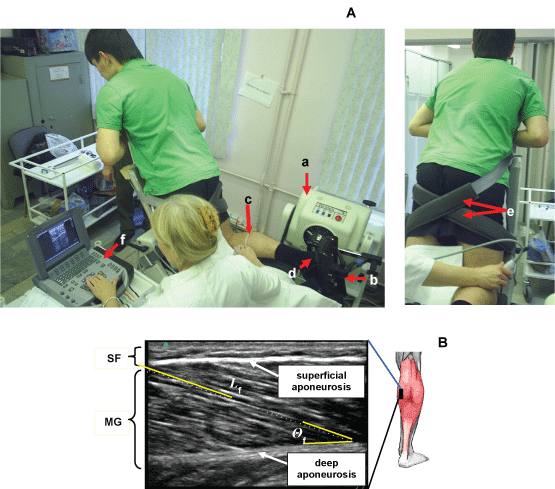 Figure 2: Experimental set-up. A) Position of the subject оn the dynamometer. Subject performing a muscle function test using isokinetic dynamometer and ultrasound scanning MG and LG muscles. Тhe ankle and the knee of the tested leg are fixed at 90° (neutral anatomic position) and 60°, respectively. a) dynamometer; b) dynamometer footplate; c) marker (arrow) was placed between the skin and the ultrasonic probe as the landmark to confirm that the probe did not move during measurements; d) ankle joint and dynamometer footplate ахis; e) velcro straps for fixating the thigh; f) ultrasonic system. Position of ultrasonic transducer at research of MG (left panel) and LG (right panel) muscle. B) Ultrasonic images of longitudinal sectional of medial head of the Gastrocnemius Muscles (MG). Ultrasonic transducer was placed on skin over the muscle at 30% (MG) distance between the popliteal crease and the center of the lateral malleolus. Fascicle length was determined as length of a line drawn along ultrasonic echo parallel to fascicle. Fascicle angles was determined as angle between echoes obtained from fascicles and deep ароneurosis in ultrasonic image. The superimposed white line at the топ indicates the path of a fascicle between the superficial and deep aponeuroses. Θf, the angle of pennation; SF, subcutaneous fat; GM, medial head of the gastrocnemius muscle. View Figure 2
Figure 2: Experimental set-up. A) Position of the subject оn the dynamometer. Subject performing a muscle function test using isokinetic dynamometer and ultrasound scanning MG and LG muscles. Тhe ankle and the knee of the tested leg are fixed at 90° (neutral anatomic position) and 60°, respectively. a) dynamometer; b) dynamometer footplate; c) marker (arrow) was placed between the skin and the ultrasonic probe as the landmark to confirm that the probe did not move during measurements; d) ankle joint and dynamometer footplate ахis; e) velcro straps for fixating the thigh; f) ultrasonic system. Position of ultrasonic transducer at research of MG (left panel) and LG (right panel) muscle. B) Ultrasonic images of longitudinal sectional of medial head of the Gastrocnemius Muscles (MG). Ultrasonic transducer was placed on skin over the muscle at 30% (MG) distance between the popliteal crease and the center of the lateral malleolus. Fascicle length was determined as length of a line drawn along ultrasonic echo parallel to fascicle. Fascicle angles was determined as angle between echoes obtained from fascicles and deep ароneurosis in ultrasonic image. The superimposed white line at the топ indicates the path of a fascicle between the superficial and deep aponeuroses. Θf, the angle of pennation; SF, subcutaneous fat; GM, medial head of the gastrocnemius muscle. View Figure 2
Each subject's right foot was firmly attached to an isokinetic dynamometer, and the lower leg was fixed to a test bench. The ankle joint was fixed at 15° dorsi flexion (-15°) and 0 (neutral anatomic position), +15, and +30° plantar flexion. The knee joint was positioned at 60°. Thus the following measurements were performed in 4 conditions. In each condition, the subject was asked to relax the plantar flexor muscles (passive condition), and passive plantar flexion torque was recorded from the output of the dynamometer by a computer.
After performance in the passive condition, the subject was perform measurement 50% of the MVC (static contractions; active condition) was calculated for neutral ankle position (0°), and they were encouraged to hold contraction for about 2-3 s.
Fascicular lengths and pennation angles of human TS were measured in vivo from sonographs taken during rest (passive) and active conditions. A real-time B mode ultrasound apparatus («SonoSite MicroMaxx», USA) with a 7.5 MHz linear-array probe, and length of a scanning surface 60 mm and thickness of 10 mm was used to obtain sagittal images of the TS. In each position, longitudinal ultrasonic images of the triceps surae [Medial (MG) and Lateral (LG) Gastrocnemius and Soleus (SOL) muscles] were obtained at the proximal levels 30% (MG and LG) and 50% (SOL) of the distance between the popliteal crease and the center of the lateral malleolus. Images were recorded at 50% shin length (90° flexion at the hip and knee joint), according to procedures previously described [38,39]. The marker was a 1.5-mm copper wire encased in a soft plastic material and placed on the surface along the lower leg circumference at a fixed distance (Figure 2A). The axially oriented transducer was placed perpendicularly to the muscle examined to ensure close contact and was transposed from the central to the lateral position along the marker placed on the muscle surface. During scanning, the pressure of the transducer on the skin was minimized to prevent muscle compression.
Each level is where the anatomic cross-sectional area of the respective muscle is maximal [40]. At that level, mediolateral widths of MG and LG were determined over the skin surface, and the position of one-half of the width was used as a measurement site for each muscle. For SOl, the position of the greatest thickness in the lateral half of the muscle was measured at the level mentioned above. The echoes from interspaces of fascicles and from the superficial and deep aponeuroses were visualized. By visualizing the fascicles along their lengths from the superficial to the deep aponeuroses, one can be convinced that the plane of the ultrasonogram is parallel to the fascicles [41]; otherwise, the fascicle length would be overestimated and the fascicle angle would be underestimated [42]. The echoes from interspaces of the fascicles were sometimes imaged more clearly along the length of fascicles when the plane was changed slightly diagonally to the longitudinal line of each muscle. The fascicles were somewhat curvilinear in all muscles (particularly MG) at shorter lengths. The length of a fascicle was always measured along its path, with the curvature, if present, taken into consideration. For the fascicle angle, a line was drawn tangentially to the fascicle at the contacting point onto the aponeurosis. The angle made by the line and aponeurosis was measured as the fascicle angle. Some authors have approximated a fascicle as a straight line between its origin and insertion to determine fascicle angles [43]. The probe was coated with, а water-soluble transmission gel to provide acoustic contact without depressing the dermal surface.
The Length of fascicles (Lf) across the deep and superficial aponeurosis was measured as a straight line [44] (Figure 2B).
The fascicle pennation angle (Θf) was measured from the angles between the echo of the deep aponeurosis of each muscle and interspaces among the fascicles of that muscle [38,44] (Figure 2B).
In the present study, ultrasonic measurement was repeated three times for each individual and averaged values were used. The coefficients of variation of three measurements were in the range of 0-2%. All ultrasonic images were processed with use of the software package «Dr. ReallyVision» (Ltd. «Alliance - Holding», Russia).
The distance between aponeuroses (muscle thickness) was estimated from the fascicle length and pennation angle using the following equation:
Muscle thickness = Lf x sin α [1,44]
Where Lf, and α is the pennation angle of each muscle determined by ultrasound.
Shorter fascicle lengths and steeper fascicle angles in the active compared with the rest (passive) conditions show internal shortening of fascicles by contraction (ΔLmuscle). The ΔLmuscle was estimated by the following formula, i.e.
ΔLmuscle = Lr⋅cos Θr – La∙cos Θa [45]
Where Lr and La - are fascicle lengths in rest (passive) and active conditions (50% MVC);
Θr and Θa - are fascicle angles in rest (passive) and active conditions, respectively.
Neuromuscular Electrical Stimulation (NMES) is applied to 4 muscle groups of both lower extremities. "Dry" electrodes (Ltd. «Axelgaard», USA) are placed оn the skin above the quadriceps femoris muscles, the hamstrings, the tibialis anterior, the peroneal, and the Triceps Surae muscles (TS). Тhe synchronous stimulation of antagonistic muscle groups prevents unwanted joint movements. The electrical stimulus was provided by the «STIMUL LF-1» stimulator (Russia). The technical equipment consists of electrode trousers carrying stimulation electrodes for the 12 channels, and 2 interconnected 6 channel stimulators carried оn a belt.
During DI, subjects executed а NMES-training during 3 hours per day with 1 s « оn » and 2 s « off » with a frequency of 25 Hz and amplitude of stimulus from 0 up to 45 V for training. Used biphasic rectangular by 1 ms pulse width. Low frequency stimulation was used to actuate the slow-twitch fibers. After initialization procedure, the system begins automatic training. NMES-training of muscles of the examinee was carried out directly in a bath. The intensity level stimulation is determined by a threshold of bearableness of subjects.
NMES-training continued for six days, during which daily five days on end (from Monday to Friday inclusive) including one day of rest (Saturday). Duration of NMES-training was 3 hours/day. Each subjects instructed "increase amplitude stimulation pulse during training". Dynamics of changes stimulus pulse of everyone subjects during daily training shown Figure 3.
 Figure 3: Dynamics of change of amplitude stimulus pulses during training. View Figure 3
Figure 3: Dynamics of change of amplitude stimulus pulses during training. View Figure 3
Contractile properties and muscle architecture of the TS were tested twice: Before and after DI. The test protocol was identical for both pre- and post-DI tests.
Data are presented as the mean values ± Standard Error of the mean (± SE). Differences in pennation angles, fibre lengths and thicknesses between rest and 50% MVC and between different ankle angles were tested using two-way analysis of variance tests. Tukey's test was used to determine significant difference between mean values. One-way Analysis of Variance (ANOVA) was used for comparison of muscle thickness, pennation angles, and fibre lengths. A level of p < 0.05 was selected to indicate statistical significance.
The individual data of the maximal torque developed by muscles-extensor foot are shown in Figure 4. Maximal plantar flexion torque increased on the average by 10.5% (148.2 ± 6.9 vs. 163.8 ± 5.9 N) after DI with application by NMES-training, corresponding by five subjects and one has decreased for 9.6% (155 vs. 140 N; p > 0.05).
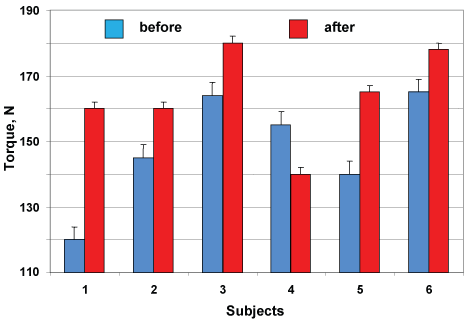 Figure 4: Changes in maximal plantar flexion torque of individual subjects after DI with NMES training. View Figure 4
Figure 4: Changes in maximal plantar flexion torque of individual subjects after DI with NMES training. View Figure 4
Thicknesses of MG, LG and SOL at rest (~18, 16 and 15 mm, respectively) did not change significantly in response to changes in muscle length resulting from changes in ankle joint angle (Figure 5).
 Figure 5: Medial (MG), and Lateral (LG), and Soleus (SOL) muscles thickness as a function of changes ion ankle at rest. View Figure 5
Figure 5: Medial (MG), and Lateral (LG), and Soleus (SOL) muscles thickness as a function of changes ion ankle at rest. View Figure 5
In all three muscles at rest, were ankle angle dependent (Table 1). In all three muscles, as ankle angle increased from -15 to +30°, the Θf increased in MG from 31 ± 2.8 to 49 ± 1.7° (58%, p < 0.01), in LG from 20 ± 2.1 to 28.5 ± 1.6° (43%, p < 0.05) and in SOL from 22.8 ± 1.4 to 34 ± 2.2° (49%, p < 0.01), respectively (Figure 6). In all three muscles, as ankle angle increased from -15 to +30°, Lf decreased in MG from 36 ± 1.2 to 27 ± 2.1 mm (25%, p < 0.01), in LG from 46.8 ± 0.6 to 31.2 ± 1.9 mm (33%, p < 0.01) and in SOL from 39.2 ± 1.2 to 28.2 ± 2.0 mm (28%, p < 0.01), respectively (Figure 6).
 Figure 6: Changes in the triceps surae complex architecture. Medial (MG), and Lateral (LG) Gastrocnemius, and Soleus (SOL) muscles fascicle length (L) and pennation ankle (Θ) as a function of changes joint ankle at rest. Values presented are
means ± SD. View Figure 6
Figure 6: Changes in the triceps surae complex architecture. Medial (MG), and Lateral (LG) Gastrocnemius, and Soleus (SOL) muscles fascicle length (L) and pennation ankle (Θ) as a function of changes joint ankle at rest. Values presented are
means ± SD. View Figure 6
Table 1: Fascicle angles and fascicle lengths of MG, LG, and SOL. View Table 1
In all three muscles after DI with NMES training, Θf and Lf were ankle angle dependent (Table 1). In all three muscles, as ankle angle increased from -15 to +30°, the Θf increased: in MG from 26 ± 2.8 to 36 ± 2.8° (38%, p < 0.05), in LG from 15 ± 1.4 to 20.2 ± 1.0° (35%, p < 0.05) and in SOL from 18.5 ± 1.6 to 24.8 ± 1.7° (34%, p < 0.01), respectively (Figure 6).
In all three muscles, as ankle angle increased from -15 to +30°, Lf decreased: in MG from 30.2 ± 1.7 to 25.5 ± 1.9 mm (16%), in LG from 40.2 ± 0.6 to 25.2 ± 2.9 mm (37%, p < 0.01) and in SOL from 29.2 ± 2.9 to 22.2 ± 2.1 mm (24%, p < 0.05), respectively (Figure 6).
The muscle fibres Θf and Lf as a function at 90° joint ankle, аге shown from rest to 50% MVC in Figure 7. Θf and Lf decreased as a function of contraction intensity in all three muscles. Θf in MG, LG and SOL gradually decreased from 49 ± 1.4 to 40.5 ± 2.4° (17%, p < 0.01), from 26.2 ± 1.8 to 20.5 ± 1.9° (22%, p < 0.05) and from 36 ± 5.3 to 29.8 ± 3.9° (17%), respectively. Lf in MG, LG and SOL decreased gradually from 26.3 ± 2.6 to 21.5 ± 1.9 mm (18%), from 36 ± 2.9 to 28.2 ± 2.2 mm (22%, p < 0.05) and from 32.2 ± 2.2 to 25.5 ± 2.1 mm (21%, p < 0.05), respectively.
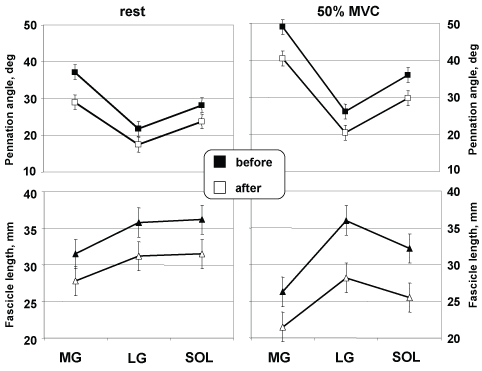 Figure 7: Architectural characteristics during graded isometric force. Changes at rest and during 50% MVC at the neutral position in MG, and LG, and SOL. View Figure 7
Figure 7: Architectural characteristics during graded isometric force. Changes at rest and during 50% MVC at the neutral position in MG, and LG, and SOL. View Figure 7
А comparison оf the mean values of Θf, Lf, and thickness, at rest and at 50% MVC аге presented in Table 2. As shown in this table, in the transition from rest to 50% MVC at the neutral ankle position (0°), the thickness of MG decreased (no significant difference) at about 5 mm but the thicknesses of LG and SOL decreased gradually from 13 to 9 mm (31%, p < 0.01) and from 17 to 13 mm (no significant difference), respectively.
Table 2: Pennation angle (Θ), fibre length (L), and thickness (H) at rest and at the 50% MVC before and after 6-d DI with long-term electromyostimulation trainings. View Table 2
Estimated by after DI with NMES training, Θf during 50% of isometric plantar flexion moment differed by 8.9° (17.3%, p < 0.05), 5.7° (21.8%, p < 0.05), and 6.2° (17.2%, p < 0.05) from the corresponding actual Θf in MG, LG and SOL, respectively (Figure 7, top, right panel). In MG, Θf values were higher than the corresponding actual values at contraction intensities by 50% of MVC but in LG and SOL values were systematically lower than the corresponding actual values.
Estimated by after DI with NMES training, Lf during 50% of isometric plantar flexion moment differed by 4.8 mm (18.2%, p < 0.01), 7.8 mm (21.7%, p < 0.01) and 6.7 mm (20.8%, p < 0.01) from the corresponding actual fibre lengths in MG, LG and SOL, respectively (Figure 7, bottom, right panel).
This study describes, for the first time, the architecture of the human TS [Medial (MG) and Lateral (LG) Gastrocnemius and Soleus (SOL) muscles] in vivo, both at rest and during graded (50% MVC) isometric plantar flexions.
The results obtained in vivo indicate that human MG, LG, and SOL architecture drastically changes both as a function of ankle joint angle at rest and as a function of the force developed during isometric contractions at a fixed joint angle. At rest, when changing the ankle joint angle from -15 to +30°, MG pennation angle increased from 31 to 49°, LG - from 20 to 28.5°, and SOL - from 22.8 to 34°; fibre length decreased from 35.5 to 26.8 mm, LG - from 46.8 to 31.2 mm, and SOL - from 39.2 to 28.2 mm. These results indicate that fibre length and pennation angle of the human TS cannot be assumed to remain constant with changing muscle length [3,46,47]. The decrease in fibre length and increase in pennation angle with increasing muscle length may be ascribed the taking up of the slack characterizing these structures [3]. In the present study, the decrease in fibre length (cf. above) occurring from -15 to +30° of passive plantar flexion also suggests that muscle fibres became progressively slack with increasing ankle joint angles. This observation is consistent with the findings of Ichinose, et al. [48]. These authors observed that when the knee is fully extended, muscle fibres are remarkably slack, for they decrease by about 35% in length when contracting only by 10% of the MVC. In the present study, the decrease in fibre length (cf. Table 1) occurring from -15 to +30° of passive plantar flexion also suggests that muscle fibres became progressively slack with increasing ankle joint angles.
The present study showed that from rest to 50% MVC, MG pennation angle increased from 37.2 to 49.0°, whereas fibre length decreased from 31.5 to 26.9 mm; LG - 21.8 to 26.2°, whereas fibre length slightly increased from 35.8 to 36.0 mm; SOL - 28.2 to 36.0 whereas fibre length decreased from 36.2 to 32.2 mm with no significant change in the distance between the aponeuroses. This finding agrees with the predictions of Gans and Bock [34].
The present study aimed to elucidate the effects of chronic unloading on the mechanical properties of human muscle and to examine the potential preventive effects of NMES-training performed during the period of unloading on mechanical properties. Our findings show that of unloading resulted in a reduced structural and increased contractile properties of human muscle, and, although the exercise regimen (NMES-training) performed did attenuate these detrimental effects, it did not completely prevent them. The present study may be considered unique in terms of the duration of unloading and use NMES-training; many studies have investigated physiological adaptations to longer periods of unloading [49,50].
A number of studies have documented that the microgravity environment encountered during spaceflight or simulated by using models of weightlessness induces alterations in skeletal muscle function [1,19,29,31,51]. In the absence of weight-bearing activity, strength loss is the most evident consequence of atrophy. These alterations are also accompanied by changes in the mechanical properties of muscle in humans by un weighting such as immersion [14,16,17,25] and bed rest [25,28,30,32], and spaceflight [29,52]. According to Kirenskaya, et al. [15], and Sugajim, et al. [6] weightlessness induced by DI gave rise to characteristic changes in the recruitment order of motor units during voluntary isometric contraction. The order is not completely fixed, being variable under different conditions. Of the factors controlling the order, the proprioceptive input to the moto neurons is known to be most important, especially to the voluntary muscle contraction. Weightlessness releases the musculature from its weight-bearing task and should reduce the proprioceptive inputs from tendon and muscle spindles.
The major findings of this study were that, isometric maximal voluntary torque by the plantar flexor muscles increased. Previous studies have documented decrease of the contractile properties of skeletal muscles during DI [14,16,17,25]. The present exercise training resulted small increased (~11%) in maximal voluntary plantar flexion torque whereas absence of preventive actions results in decrease in MVC more than on 40% [13,14,16,17,19,25,53] and in Po more than on 30% [16,17,19].
However efficacy of NMES-training for increased the contractile properties of skeletal muscles during un weighting has been suggested in previous studies [4,54,55] NMES may lead to a reorganization of brain structures and descending motor outputs. It is known that afferent stimulation implicated in a focal increase of motor cortical excitability of the trained muscle [5]. In a recent study, Khaslavskaia and Sinkjaer [56] demonstrated that motor evoked potentials of the muscle (tibialis anterior) elicited after the training were increased in conditions rest by 38%. Thus, increasing cortical excitability of the human TS, stimulating CPN further at voluntary activation of the human TS would facilitate cortical activity, as well as it is shown in this study (cf. Figure 3 and Figure 4).
The insignificant increase in force of contraction in the present study can be assumed it is defined by slack intensity impulses.
It is well known that the smaller motoneurons innervating muscles are more readily activated than the larger cells innervating units, as the strength of the contraction increases progressively. The smaller units consist of slow twitch muscle fibres (type I) and the larger units consist of fast twitch fibres (type II). In submaximal voluntary contractions, type I fibres the motor units are activated by the synaptic current impinging on the motor neuron. The situation is completely different in contractions triggered by NMES, because the muscle fibres of the motor units are activated by an electric current which is applied extracellular to the nerve endings, and larger cells with lower axonal input resistance are more excitable [57,58]. In fact, when the stimulus is applied from outside the cell, the electric current must first enter through the membrane before it depolarizes the cell, but the extracellular medium shunts the current, and the smaller motor units will not be activated during submaximal NMES because of their higher axonal input resistance. Therefore, the smaller motor units do not adapt to training with submaximal NMES. However when use electrical stimulation high training intensity, larger force NMES-training to be more efficient exercise [59]. In present study average intensity impulses during training was essentially insufficient for activation of small motor units (e.g. subject 4 vs. 1, see Figure 3).
The increase in the maximal torque was accompanied by changes of internal architecture the MG, LG and SOL which have been in part prevented by preventive exercises (NMES-training). Both fascicle length and pennation angle were reduced after DI with NMES-training, this strongly suggests a loss of both in-series and in-parallel sarcomeres, respectively. The functional consequence of the decreased fascicle length was a reduced shortening during contraction. The loss of in-series sarcomeres would mean that this is likely to have implications both on the force length and force velocity relationships of the muscle. The observation of a smaller pennation angle during contraction after DI with NMES training will partially compensate for the loss of force, because of a more efficient force transmission to the tendon. The reduced initial resting Θ probably, grows out reduction decreased tendon stiffness or of the muscle-tendon complex that finds confirmation in substantial growth ΔL muscle of LG (with 0.9 up to 3.3 mm after DI) during contraction. This observation is consistent with the findings of Kubo, et al. [30].
Moreover, reduction of number consistently connected sarcomeres allows assuming, that the size of developed reduction of a fibre will be reduced. This supervision will be coordinated to the results received earlier in conditions immobilization of limb [27,60-62].
Smaller pennation angle an inclination of a fibre during reduction of a muscle after DI with use of NMES-training, apparently, in part compensates loss of force which is constant "satellite" of gravitational unloading muscular, the device [16-19,25,29,51-53] because of more effective transfer of the force developed by fibres to a sinew. Reduced initial resting pennation angle an inclination of a fibre, probably, grows out reduction of rigidity of a sinew or muscle-tendon complex that finds confirmation in substantial growth ΔL muscle of LG (with 0.9 up to 3.3 mm after DI) during reduction of a muscle and proves to be true earlier received data [30].
Muscle thickness of LG, and MG significantly decreased after DI with NMES-training in the trained limb. This result undermines the contention that the degree of muscle atrophy is related to the relative amount of slow twitch fibers within а muscle, since LG and MG has relatively higher percentage of fast twitch fibers [2,63]. А decrease in muscle thickness of LG, MG, in the trained limb appears contradictory considering that the trained limb did exercises during DI. The reason for this is not clear, but it clearly points to the fact that specific training is required for the maintenance of the contractile properties and architecture of skeletal muscles during DI.
The increase in the maximal voluntary torque after DI with NMES training allows to assume, that NMES training, apparently, promotes increase stream muscular afferentation [64] in conditions of his deficiency at gravitational unloading the muscular device caused long immersion that can promote also to the certain role in maintenance and normalization of activity of control systems by any movements (by a principle of a feedback). Tetanic electrical stimulation applied over human muscle generates contractions by depolarizing motor axons beneath the stimulating electrodes. However, the simultaneous depolarization of sensory axons can also contribute to the contractions by the synaptic recruitment of spinal motoneurons. Upon entering the spinal cord, the sensory volley recruits spinal motoneurons, leading to the development of central torque. This recruitment is consistent with the development of persistent inward currents in spinal motoneurons or inter neurons [65-67]. Persistent inward currents lead to sustained depolarizations (plateau potentials), and it is becoming increasingly clear that they play an important role in regulating cell firing in normal [67-69]. Maximizing this central contribution may be beneficial for increased muscle force (by a principle of a feedback).
Conclusions, from the present results, follows, first, that the architecture different lead the triceps surae muscle considerably differs, reflecting, probably, their functional roles, second, various changes fibre length and pennation angle between different muscles, probably, are connected to distinctions in ability to develop force and elastic characteristics of sinews or muscle-tendon complex and, at last, in the third, NMES training has preventive an effect on stimulated*muscles: in part reduces loss of force of reduction of the muscles, the caused long unloading. The received data, allow concluding, that use of NMES trained renders the expressed preventive action, essentially reduces depth and rate of atrophic processes in muscles.
Present results suggest that the structural adaptations to immersion (unloading) likely to contribute to a reduced force loss. On this background used NMES training of muscles in conditions of unloading allows to increase contractile function.
The author is particularly grateful to M. Kuz'mina, M.D., Ph.D. (Clinical Hospital N 1 President Medical Center, Moscow, Russia) for the executed ultrasonic researches. Special gratitude the author expresses Mrs V. Kovalenko, Head of Moscow Representation «Dalco International» (Moscow, Russia) for providing the ultrasound apparatus «SonoSite MicroMaxx» (USA).
He thank Dr. I. Berezhinski, Ph.D., Head of Moscow Ltd. «Alliance - Holding» for development of the software, and Mr. N. Khimoroda for assistance with the isokinetic dynamometer «Biodex Systems 4 PRO».
At last, the author expresses gratitude to all the volunteers which participated in this research.
This study was supported by Grant the Ministry of Education and Science of the Russian Federation (number RFMEFI61317X0074).
The author declares that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.