Intraepithelial Lesions (IELs) are focal outgrowths of the Mammary Gland (MG), some of which are considered early precursors of tumors. The early detection of IELs has improved the survival rate in women. IELs in female canines have been proposed as a comparative model of IELs in humans.
The main aim of this work was to histologically characterize IELs in the MGs of intact female dogs with no signs of mammary disease.
Biopsies of the right inguinal MG were obtained from 37 dogs under anesthesia, and the MGs from another 13 female dogs were collected at necropsy (108 total samples).
MG samples obtained via biopsy did not show any IELs. Among the MGs collected at necropsy, 19 (6/13 dogs) had IELs. The most frequent IELs were adenosis (14 MGs from 4 dogs) and Carcinoma In Situ (CIS) (12 MGs from 3 dogs). Based on the results, we propose that benign and malignant complex lesions be included as IELs in canines. A main finding was that CIS appears to arise spontaneously in multiple lobes/lobules without any evidence of atypical IELs, that is, CIS lesions were malignant from the onset and did not appear to require any other IEL as a precursor. Conclusion: This study demonstrates that female dogs with healthy mammary glands from Bogota have a high rate of IELs in their MGs and that many of the IELs have a malignant character from the onset. Our findings support a new hypothesis for mammary gland carcinogenesis in dogs in accordance with recent theories of the origin of breast carcinomas in humans, i.e., the theory of the Sick Lobe (SL) and biological timing, highlighting the similarities between these pathologies in both species. The importance of myoepithelial cells in the early evolution of IELs is also highlighted in the present work.
Canine, Fusiform cells, Intraepithelial lesions, Immunohistochemistry, Mammary gland, Mammary carcinoma, Myoepithelial cells
Human mammary cancer has reached alarming levels and is considered a public health problem in Colombia and worldwide [1,2]. Mammary neoplasia is one of the most frequent types of cancer, with an incidence of 1.15 million new cases globally in 2002, or 23% of all malignant tumors. In Latin America and the Caribbean, 300,000 women die yearly due to this disease (83 deaths per day) [3].
In female canines, mammary tumors are considered one of the most important cancers due to their high prevalence [4-6] and represent nearly 20% of all tumors in this species [7]. A new classification for this type of neoplasia in female canines was recently released and is very similar to that established for human females, with one important difference being the existence of complex tumors (with epithelial and myoepithelial components) in dogs. Most mammary neoplasias in canines are malignant.
Compared with canines that undergo ovariectomy, non-ovariectomized dogs have a three- to seven-fold increased risk of mammary tumors, and ovariectomy has been shown to be effective in preventing the onset of mammary tumor [8]. After two and a half years of age, surgery has little or no effect on tumor presentation [8]. However, some studies suggest that there may be some protection conferred by sterilization between the fourth and sixth estrus [9]. Since mammary carcinoma occurs in almost all mammalian species, an analysis and comparison of the natural occurrence of this disease in different species will improve our understanding of how this type of tumor develops in humans [10].
Factors including hormone levels, genetic predisposition, diet, environmental factors and hormone therapy have been suspected as risk factors for the development of canine mammary gland tumors, but a definitive cause(s) has not been definitively demonstrated [4,8].
The "theory of the sick lobe" posits that Ductal Carcinoma In Situ (DCIS), Lobular Carcinoma In Situ (LCIS) and invasive breast cancer are diseases of the lobe in which genetic factors predispose the duct to cancer, and environmental factors promote the development of the disease. This theory suggests that individual lobes can have large areas of premalignant cells that can ultimately develop into cancer even after cancerous portions of the lobe have been removed [11].
Most invasive breast cancers are thought to evolve from early breast lesions such as atypical hyperplasia and Carcinoma In Situ (CIS) [12]. These lesions are increasingly diagnosed because of increased public awareness and routine mammographic screening [13]. Similarities in the early development of the most common carcinomatous lesions between human and canine species have recently been demonstrated. These early lesions are called Intraepithelial Lesions (lELs) [10], intraepithelial ductal and lobular neoplasia or "borderline breast diseases" [14,15]. The formation of some mammary carcinomas may involve several stages (IELs) prior to manifesting clinically, and some of these IELs are morphologic risk factors or precursors of neoplastic pathologies [12,16]. Because of a lack of clear criteria for diagnosing lesions and abnormal growths of the ductal or acinar epithelium with no evidence of basement membrane rupture (also called IELs) in canines [10], researchers from various regions of the world have used criteria established in human mammary pathology [17]. These criteria are based on the fact that these IELs can originate from the lobules or the ducts of the MG [16]. IELs are categorized as adenosis, sclerosing adenosis, intraductal papilloma, sclerosing papilloma, ductal hyperplasia, Atypical Ductal Hyperplasia (ADH), Ductal Carcinoma In Situ (DCIS; high, intermediate, and low grade) [17] and columnar cell lesions of the canine mammary gland [18].
This is the first of two reports documenting the early development of IELs in female canines. In this report, the epithelial and myoepithelial components of IELs are described, analyzed and compared with similar lesions in women. We propose a new hypothesis regarding the origin of these carcinomatous lesions in canines, as has recently been done for these lesions in humans [11,19-21]. Moreover, in this study, changes in canine myoepithelial cells similar to those in human IELs were found, and new IELs not reported in the veterinary literature are described.
A descriptive and semiquantitative study was undertaken of non-invasive and invasive intraductal and acinar epithelial lesions, denoted IELs [10,17,22]. Fifty female canines were included and represented the breeds most commonly affected by mammary tumors. The following breeds were assessed at our laboratory of Veterinary Pathology, National University of Colombia (PVL, UN): Mixed, French Poodle, German Shepherd and Labrador [23]. The animals were reproductively active (intact), had more than one estrus and had no evidence of clinical mammary disease. The physiologic status of the MG was morphologically determined according to the criteria defined by Rehm, et al. [24] Orfanou, et al. [25] and Santos, et al. [26]. The approach was used because the densities of estrogen and progesterone receptors in the mammary epithelial cells vary among the stages of the estrus cycle, which influences the activity (growth or involution) of the gland. For example, it has been described that in canine diestrus, increasing progesterone levels induce the synthesis of mammary gland Growth Hormone (GH) and Growth Hormone Receptor (GHR), which promote mammary gland proliferation [24]. Therefore, this variation must be accounted for during the histological interpretation and counting of IELs to prevent the classification of normal hyperplasia as pathological hyperplasia [27].
The samples originated from two sources: 1) A biopsy sample from the right inguinal MG was collected under general anesthesia from individuals undergoing ovary-hysterectomy at the Zoonosis Center of Bogotá City (ZC) (post-chirurgic controls were the responsibility of the ZC). Biopsy was performed by incising the skin over the nipple, which is then grasped and separated bluntly from the surrounding parenchyma using mosquito hemostats or scissors. Once exposed, the mammary gland is sampled gently by cutting with scissors. Following control of hemorrhage, the skin is closed with simple interrupted sutures. The size of the MG varied between 1 and 2 cm3. 2) Visible MGs were collected from females that had been submitted to our necropsy service or to euthanasia at the ZC for reasons other than mammary disease. All of the procedures were approved by the Bioethics Unit of the Veterinary and Zootechnology School of the National University of Colombia (2011), and a procedure favored by the concept of the Professional Ethics Tribunal COMVEZCOL was utilized (2012). Chirurgic procedures were performed by qualified veterinarians.
A single incisional biopsy was taken from the right inguinal MG from thirty-seven females undergoing ovary-hysterectomy. From the 13 cadavers (6 from the PVL, UN and 7 from ZC) 108 MGs were obtained. A total of 145 MG samples made up the study sample.
All of the samples were processed following routine histology: 3.7% formalin fixation, paraffin embedding, sectioning into 3-4 µm sections and Hematoxylin-Eosin (H&E) staining.
Immunohistochemical analysis was performed with antibodies directed against Smooth-Muscle Actin (SMA) using a polymer-based non-avidin-biotin-peroxidase system Dako EnVision™ + Dual Link System-HRP (DakoCytomation, Glostrup, Denmark) method with a primary antibody mouse monoclonal for anti-actin, muscle-specific (HHF35) BioGenex (San Ramon, CA, USA) at a dilution of 1:50 according to the manufacturer's instructions [28].
The criteria proposed by Antuofermo, et al. [10] for diagnosing IELs in canines were partially followed in this work (Table 1). These criteria were established by consulting medical pathologists, the literature regarding canine mammary pathologies [29], and recent textbooks on woman breast pathologies [11,30-32]. The classification of Ferreira, et al. [18] was followed for the columnar lesions. For carcinomas, the grade was primarily established by nuclear pleomorphism and the presence of mitosis: Low grade (1), intermediate grade (2) or high grade (3) [10,17,32,33]. Only one H&E slide per gland was used for counting the number of IELs. A ductal or lobular lesion was considered an IEL when it was separated from another by interlobular connective tissue.
Table 1: Type of mammary IELs and corresponding relative risks for invasive carcinoma. Atypical Ductal Hyperplasia (ADH), Atypical Lobular Hyperplasia (ALH). View Table 1
A descriptive analysis of the frequencies of the various IELs was performed. Due to the distribution of the study population, i.e., two different populations, a separated point prevalence of the IELs was determined for each experimental group [34]. The prevalence of IELs in the biopsied canines was 0% (0/37), whereas the prevalence of IELs in the canines submitted to necropsy was 46.15% (6/13).
Forty-seven of the females were mixed breed; in addition, there was one Labrador, one Poodle and one German Shepherd. IELs were found in six of 13 cadavers (five mixed breed and one German Shepherd). No IELs were observed in the animals from which biopsies were taken.
Regarding the reproductive status of the affected animals, two were in early diestrus, one was in late diestrus (pregnant), one was in estrus, one was in early anestrus and one was in late anestrus.
Only 19 of 108 (17.6%) sampled MGs had IELs and/or Invasive Carcinomas (ICA) in the six females that showed any of these lesions. A total of 50 IELs and 7 ICA were diagnosed. A total of 64% of the IELs were low risk, and the remaining 36% were high risk. However, when including the ICA, 43.8% were high risk and 56% were low risk. High-risk lesions, whether invasive or not, affected 4 (66.7%) of the 6 females. Eighty-four percent of the affected MGs were from the inguinal and abdominal regions, whereas only 16% were from the thoracic region (Table 2).
Table 2: A: Adenosis; UDH: Usual Ductal Hyperplasia; CCL: Columnar Cell Lesions; IP: Intraductal Papilloma; CH: Complex Hyperplasia; ALH: Atypical Lobular Hyperplasia; CIS: Carcinoma In Situ; CC: Complex Carcinoma; ICA: Invasive Carcinoma. Individual glands presented multifocal and multiple lesions of both the same type and of different types. View Table 2
Only the frequency was investigated for IELs already described in the veterinary literature [10,17,29,35].
Fourteen adenosis, five Usual Ductal Hyperplasia (UDH), four columnar lesions (all within DCIS), four intraductal papillomas and one atypical lobular hyperplasia were found; their cytology and architecture agree with the reported characteristics [10].
Complex hyperplasia and complex carcinoma, which have equivalents in mammary tumors but have not been described as IELs or hyperplasia/dysplasia in the veterinary literature, were included in this work [29].
Four lesions were diagnosed as CH. These followed the classical morphology described for these lesions, i.e., proliferating epithelial and myoepithelial cells.
In 12 (63%, 12/19) MGs, two types of CIS were diagnosed: Ductal carcinoma in situ and lobular carcinoma in situ. They were counted in each gland, and the DCIS lesions were subdivided according to morphologic pattern: Cribriform, papillary, micropapillary, solid and comedo-type necrosis. Each CIS was separated by interlobular connective tissue.
In addition to the morphologic characteristics and frequency of DCIS or LCIS, the nuclear grade for each lesion was established. As shown in Table 3, 26% of CIS lesions had high nuclear grade and 18(42.9%) were grade 2.
Table 3: Distribution of different types of DCIS and LCIS. View Table 3
The term DCIS encompasses a heterogeneous group of neoplastic epithelial cells within the mammary duct system with an intact basement membrane and the presence of a myoepithelial cell layer; they do not have the ability to metastasize but possess a risk for progression to invasive cancer [30,36,37]. These lesions differ with regard to their mode of presentation and histopathological features. The ductal carcinoma in situ has traditionally been classified according to its architectural pattern of growth in cribriform, papillary, micropapillary, solid, and comedo carcinoma, the latter being characterized by the presence of central necrosis. Graduation is established mainly by nuclear pleomorphism and/or presence of necrosis in a system of three degrees (low grade or 1 degree, intermediate or 2 and high grade or 3) [38].
Papillary DCIS: Seven carcinomas were classified as Papillary DCIS. These carcinomas are within a dilated duct and frequently arise from one or more locations on the wall of the ductal epithelium, supported by a branching fibrovascular core. Most of the nuclear grades of these lesions were intermediate (n = 5) or high (n = 2).
Micropapillary DCIS: Eight lesions were classified as Micropapillary DCIS; within a duct, these lesions are primarily multifocal, but in contrast with the papillary DCIS, they lack a branching fibrovascular core. Most nuclear grades were low (63%) or intermediate (25%) (Figure 1). One of these lesions showed squamous differentiation (layers of keratin with undifferentiated cells).
Solid DCIS: Five lesions were considered as solid DCIS; among them, 40% were high nuclear grade, 40% were intermediate grade and 20% were low grade. These lesions consist of a duct filled with an uninterrupted mass of epithelial cells.
Comedo DCIS: Five lesions were diagnosed as Comedo DCIS. The nuclear grade was III in 100% of these lesions. This lesion is characterized by the presence of necrosis in the central mass of epithelial cells.
Seventeen lesions were diagnosed as LCIS. They involve the alveoli and the ductules in a lobule (i.e., they have a lobule-centric distribution) and are characterized by a proliferation of cells with typical features of carcinoma, including a higher density of cells than in normal acini, atypia and lack of polarity of the cells. They show different levels of nuclear degree and typically a lack of polarity; eccentric nuclei as well as large nucleoli were included. Distention of the ductules is common but not necessary for the diagnosis.
These lesions primarily affected the acini and terminal duct lobular unit of the MG. Most of the lesions had a nuclear grade from low to intermediate, and only one case had a high nuclear grade.
Six lesions were considered as complex carcinoma (multifocal proliferations of malignant epithelial cells accompanied by benign myoepithelial cells). The malignant phenotype could be demonstrated only at high magnification in the epithelium.
Seven lesions were included in the ICA group. The lesions were small and could still be viewed in a 40x field; they exhibited various architectonic patterns and formed ducts or solid areas and comedo-necrosis. The cells demonstrated moderate to severe pleomorphism, and those that invaded the stroma had a fusiform appearance (Figure 2). This fusiform morphology was apparent not only for invading stroma but also for cells that projected into the duct lumen (Figure 3 and Figure 4), and even some of them return to the alveolar wall (Figure 5). The nuclear pleomorphism was moderate to severe, with clumped (vesicular) chromatin and many mitoses, some of which were aberrant. Multifocal necrosis and an infiltrate of lymphocytes plasma cells and macrophages accompanied these lesions.
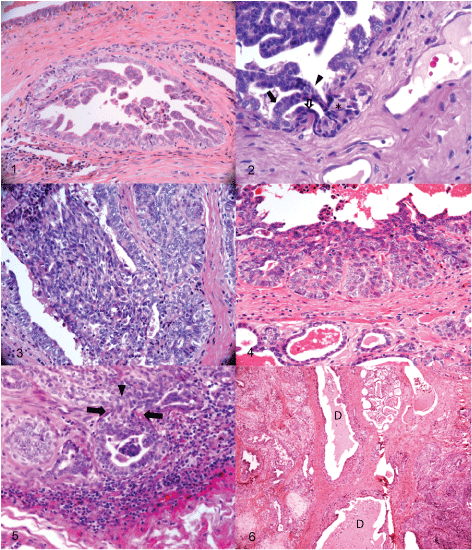
Figure 1: Mammary gland, dog, intermediate grade Micropapillary DCIS. A mild lymphocytic infiltrate is observed immediately below. H&E 200x.
Figure 2: Mammary gland, dog. Invasive carcinoma. Two bands of neoplastic cells project into the duct´s lumen (arrow), they fuse to each other (arrowhead) before returning to the duct´s wall, where they break the basal membrane and invade the stroma. Some nuclei adopt a fusiform appearance (open arrow). Another band of fusiform cells accompany the invading cells at the periphery (asterisks). H&E 400x.
Figure 3: Mammary gland, dog, High-grade DCIS. Fusiform cells project into the duct lumen. H&E 20x.
Figure 4: Mammary gland, dog, high-grade DCIS. The uppermost layer of the epithelium adopts a fusiform aspect and appears to form a single layer joining five active proliferating projections of the epithelium; the innermost layer of cells has large nuclei with prominent nucleoli and some with loose chromatin (euchromatin). The acini at the bottom show a normal appearance. H&E 20x.
Figure 5: Mammary gland, dog. Invasive carcinoma. The epithelial cells within a projection of a tubule adopt a fusiform appearance when they project into the stroma (arrowhead between arrows). Lymphocytic infiltrate surrounds the lesion. H&E 400x.
Figure 6: Mammary gland, dog. Complex carcinoma on the left and LCIS on the right; they are separated only by the ducts. Different subtypes of carcinomas can arise within the same “sick lobe”. H&E 4x.
View Figure 1-6The most relevant finding of this work regards the presence of DCIS or LCIS in all of the dogs with carcinomatous lesions. These lesions arise de novo in a few or many ducts or lobules simultaneously (synchronously or asynchronously) without any evidence of a precedent IEL, i.e., within or in direct contact with morphologically normal acini or ducts (Figure 6 and Figure 7). A lymphoplasmocitary and macrophage response accompanies most of these lesions (Figure 8).
Most ICA showed multifocal breaches of the nearby myoepithelial cells; however, not all demonstrated this change. Some ICA retained a complete myoepithelial layer; additionally, in some or multiple places, normal ducts or acini showed evidence of slight breaches in the myoepithelial layer (Figure 9, Figure 10 and Figure 11).
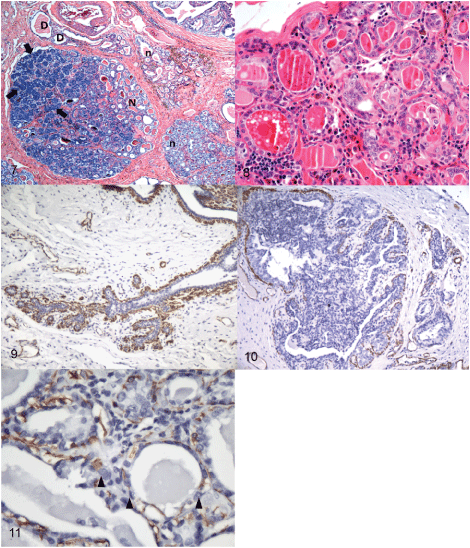
Figure 7: Mammary gland, dog, intermediate grade LCIS "sick lobe”. Note higher cellular and nuclear densities (deeper basophilia) of carcinomatous acini (primarily in the uppermost left quadrant (arrows)) adjacent to some apparently normal acini (N); other lobules with normal appearance (n); at first view, there is no compromise of the intralobular ducts (D). Note the "spontaneous" origin of the carcinomatous region with no other intervening IELs. H&E 40x.
Figure 8: Mammary gland, dog, intermediate grade LCIS, "sick-lobe". Apparently healthy acini admixed with other malignant appearing acini; no other intervening IELs are evident between the “normal” and malignant areas. Cells and nuclei at the malignant region are irregularly oriented, nuclei are more numerous per unit of area, the chromatin is very active, and prominent nucleoli are visible. Infiltrating lymphocytes are evident at the “advance front” of the lesion. H&E 200x.
Figure 9: Mammary gland, dog. Normal myoepithelial cells surrounding the terminal duct-lobular unit. IHC, myosin-heavy chain, 100x.
Figure 10: Mammary gland, dog, papillary DCIS. Extensive areas of discontinuous myoepithelial cell layer surround and are within a papillary DCIS. IHC, myosin-heavy chain, 100x.
Figure 11: Mammary gland, dog, low grade LCIS. Multiple breaches of the myoepithelial cell layer (arrowheads). IHC, myosin-heavy chain, 400x.
View Figure 7-11No IELs were detected in female dogs treated at the ZC, from which only a biopsy of the right inguinal gland was sampled. This site is reported to be the most frequent site for mammary tumors in this species. This observation is supported by the results of the necropsy group of females, in which the highest frequency of IELs was observed in the inguinal MG (Table 3). The present results suggest that incisional biopsy is unsuitable for the diagnosis or research of early lesions in canines.
Furthermore, as deduced from the morphological diversity of IELs in this work and from their multifocality within and between MGs, excisional biopsies will face the same diagnostic inaccuracies as the incisional technique. To reduce sampling error, it may be appropriate to recommend that future research utilizes live animals and clinical studies and that samples for histology are always accompanied with diagnostic images [13,39].
The prevalence of IELs in necropsied females was 46.15%, which is similar to that reported by Warner [40] and Mouser, et al. [17] Four (66.7%) of the 6 necropsied individuals with IELs had at least one problematic lesion since its beginning; this percentage is notably higher than the 52% reported by Mouser, et al. [17] Interestingly, with only one less ICA than CIS, there was a nearly even ratio (0.87:1) of these two lesions in female number 6 (Table 2). The potential implications of this finding are discussed below. If all the lesions with a malignant but non-invasive appearance and those declared overtly malignant are grouped together, the percentage reaches 44% (CIS, 21%; Ca complexes, 11%; ICA, 12%), i.e., 66% of the population would be almost 50% likely to have a compromising injury from the start.
The IELs in this study occurred more frequently in abdominal and inguinal regions than in thoracic glands. This is consistent with what is known for malign and benign clinical tumors [41] and confirms the good correlation of the localization of IELs and tumors with the prevalence percentage of both groups of pathologies. Similar results were documented by Mouser, et al. [17].
We found more intermediate (42.9%) and high-risk (26.2%) IELs than low-risk lesions. These data are in accordance with those reported by various authors for clinically evident malignant tumors. Approximately 40% to 50% of these tumors were malignant [8]; however, our findings are in contrast with those of Mouser, et al. [17] who reported a greater percentage of low-risk than high-risk lesions. This difference between studies might be attributable to the different breeds included in each study. In Mouser, et al, [17] most of the females were coonhound and foxhound breeds, for which no high frequency of MG tumors has been documented; in our case, we selected breeds that in our experience and in international studies are considered highly susceptible to mammary neoplasia [8,9,23]. Our results suggest that in addition to breed, the morphology and nuclear grade of the IELs might be a prognostic factor for the development of tumors. In addition, our results are consistent with those of Antuofermo, et al. [10] and Mouser, et al. [17] corroborating that an MG can bear more than one type of IEL (Table 2) and emphasizing the utility of incisional or excisional biopsies as the only diagnostic approach in clinical cases [42].
The susceptibility of mixed-breed females with mammary neoplasia was supported in this study. Ten individuals with this condition had the largest number of IELs, which is in contrast with other studies in which pure-bred canines appeared to be more predisposed to these pathologies [9]. A higher frequency of these neoplasms has been documented for pure breeds, such as Shih Tzu, which develop malignant mammary tumors at young ages and overexpress BRCA1, a gene involved in the carcinogenesis of mammary tumors. Maltese, Yorkshire Terrier and Poodle breeds also develop mammary cancer but do not overexpress BRCA1 [43].
There is considerable disagreement regarding the classification of human IELs [22,27,30]. There is even less consensus in veterinary medicine, and although IELs are reported and classified as in human medicine, few studies have thoroughly investigated these lesions in dogs or compared them with those of humans [13,17,44]. Additionally, with the exception of the works by Mouser, et al. [17] (in which the study animals had no clinical signs of mammary tumors) and Mohammed, et al. [13] (in which some females did not have mammary tumors), previous studies on IELs have included animals with clinical mammary tumors [10,18]. This complicates comparisons.
Based on our results, we suggest that the definition of IEL expression in canines should be revised, split or expanded. We propose that the following criteria be included for assessing and classifying these lesions: 1) At least two categories of IELs should be established: "low-risk" (including adenosis, UDH, ADH, papillary, and CH IELs) and "high-risk" (such as CIS IELs, which should include complex CIS because this lesion is more common in canine than human mammary carcinoma). This proposal is supported by others who claim that the low-risk epithelial arrangements do not necessarily manifest clinically and do not always progress to cancer [10]. 2) The criteria should specify the histogenetic origin of the canine mammary epithelial lesion [45] (duct or lobule). This will enable investigations into differences regarding the pattern of invasion and prognostic value of tumor cells during tumor progression [11]. 3) The use of 'intraepithelial' means that these pathologies are independent of their immediate environment; however, as recent research demonstrates, the stroma has a more significant role in the evolution of these lesions than previously acknowledged [20,46-54]. Our own research in canines clearly shows that these pathologies are accompanied by significant (and to some extent are dependent on) changes starting in early development not only of myoepithelial cells but also their surrounding microenvironment, which includes the basal membrane, the amount of connective tissue and the infiltration of leukocytes [19,55-67].
Adenosis was the most prevalent lesion in this study (25%). However, only one female (canine 22) had this as a unique lesion. In another individual (canine 6), in addition to abundant adenosis, high-risk IELs as well as ICA were present.
Mouser, et al. [17] found higher frequencies of UDH (45.4%), DCIS (29.6%), ADH (13%), and complex adenoma (11.1%) IELs than observed here; in the present study, adenosis was the most prevalent (25%) type of IEL, and CIS (21%), complex carcinoma (11%) and ICA (12%) were also frequently observed. Mouser, et al. [17] analyzed 209 lesions, with UDH lesions comprising 45.4%, much higher than the 9% recorded in this study. The differences might be due to differences in the classification criteria used in the two works. In our case, the physiological stages of the canine estrus (reproductive cycle), such as proestrus, estrus, and diestrus, as proposed by Sorenmo, et al. [9] were taken into account. However, Mouser, et al. [17] used inclusion criteria of human breast pathologies and thus considered UDH when more than two cell layers were present; [38] according to those criteria, the primary criterion for classifying a lesion as UDH is several epithelial cell layers with regular proliferation within the duct lumen without any other sign of abnormal cell growth [9,24-26]. Under our criteria, this would not be considered a pathologic change.
The presence of columnar lesions in canine mammary glands is described within ICA (28.4%), DCIS (29.8%), benign tumors (31.4%) and normal MGs (10.4%). These lesions are pathologically and immunephenotypicaly similar to those in human breast [18]. In our study, all columnar lesions (7%) were contained within CIS; in women, the columnar injuries are considered to be markers for the presence of atypia, with a two-fold increased risk of progressing to a breast cancer [30].
The study by Mouser, et al. [17] was aimed at detecting IELs, and the animals were selected without clinical mammary signs. However, they diagnosed several "benign tumors", such as simple adenoma, complex adenoma, mixed benign tumor, basaloid adenoma, ductal papilloma and sclerosing papilloma, among which only ductal papilloma was found in the present study. We are not sure why these authors ascribed the term "benign tumors" to lesions that are not clinically manifested because that was their primary criterion for defining IELs. According to our (and others') understanding of IELs, in addition to the continuity of the basal membrane [10,15,68], they should not exhibit any clinical signs.
No ADH was observed in this work, and only one ALH was observed. In contrast, Antuofermo, et al. [10] and Mouser, et al. [17] reported these injuries in 4.67 and 13% of cases, respectively. Our results suggest that unlike IELs in women (in whom ADH is considered a step in the evolution to malignant lesions), IELs in canines do not evolve from benign to malignant stages. Several findings of this study lend support to the hypothesis that high-risk lesions have a malignant character from the very beginning, as also reported for women by Man, et al. [20]. For example, it is not plausible that highly prevalent lesions (such as CIS, which exhibit a variable morphology) can derive from a monomorphic atypical IEL (such as ADH, which had a very low prevalence in this study) and are not spatially related to high-risk lesions (CIS, ICA and Complex carcinoma).
This study categorizes CIS as 1) DCIS lesions that are derived from the ducts and 2) LCIS, in which the cells preferentially originate from the lobules. The latter can originate from the acini or the intralobular ductules because both structures are indistinguishable via H&E [2,30]. Important carcinogenic mechanism differences have been demonstrated for both types of CIS depending on their localization [11]. For instance, the lobular carcinoma does not express E cadherin; consequently, the malignant cells do not show cohesion and adopt a plasmacytoid aspect when invading, whereas the ductal carcinoma tend to form tubules when invading [30]. Our findings demonstrate that as in women, pre-invasive lesions of the MG in canines anatomically originate from the terminal duct-lobular unit; however, the immunohistochemical profiles of E cadherin and CK-34 beta E12 are required to confirm the 17 LCIS diagnosed in this work [2,69].
No clear pattern of prevalence of the nuclear grades emerges from the 3 works that specifically analyze IELs (Mouser, et al. [17], Antuofermo, et al. [10] and this work). Nonetheless, remarkably, most CIS in this study were of intermediate (42%) or high grade (27%); only 5.88% of the IELs diagnosed as LCIS were grade III, whereas the same grade for DCIS had a rate of 44%. It is possible that, as proposed by the theory of the "sick lobe" (see below), DCIS lesions are more malignant than LCIS lesions and consequently have a higher probability to progress to an ICA [14,21,70,71].
In female 6, there was a high coincidence between ICA and CIS (0.87:1). This finding lends support to the hypothesis that ICA in canine's progresses from lesions that have high-risk characteristics from inception, such as the DCIS grade III. A similar progression has been described for women [16].
In this study, we propose that IELs include pathologies of complex origin: Complex hyperplasia (7%), and complex carcinoma (11%). To our knowledge, these have not been described as such in canine IELs but have counterparts in clinically manifest neoplasia [29,72].
Female canine and human mammary carcinomas share epidemiological, clinical, biological and histological features and have been the subject of comparative research for decades [10,17]. However, comparative studies of these carcinomas during the first developmental stages have received less attention [10,73,74]. The detection of early lesions in MG has demonstrated its importance for preventing and diminishing the mortality of women as a result of mammary tumors. In this study, two similarities of canine IELs with the equivalent developmental stage in female breast cancer are proposed: 1) The origin of carcinomas in canines resembles that of human mammary carcinomas as described by the "sick lobe" theory [11], and 2) Consistent with human early CIS [28,67,73,75], more frequent multifocal ruptures of the myoepithelial layer were observed in canine CIS.
Perhaps the most relevant finding in this study is the presence of multilobar or multilobular in situ carcinomas (Figure 6 and Figure 7) involving several nearby or distant lobes (multilobar) or a single or multiple neighbor lobules (multilobular). In some cases, multilobar carcinomas belong to one or two different morphological categories (Figure 7 and Figure 8) that occupy most of the lobe, and no evidence of a relationship with any benign lesion is observable. Those growths appear to originate spontaneously de novo in a duct or in a lobule in which some "morphologically normal" ducts or acini persist. In our opinion, this morphology is akin to what is described by the carcinogenic theory of the "sick lobe" in the human breast. This postulates that the mammary carcinoma is a ductal/lobular disease in which unconnected tumoral foci appear simultaneously or asynchronously and develop within a unique diseased lobe/lobule [11].
The sick lobe is characterized by the presence of a large number of potentially malignant cells corresponding to mutant stem or progenitor cells dispersed unevenly within the sick lobe and that might undergo malignant transformation under the influence of exogenous and endogenous oncogenic stimuli [21]. This characteristic would make the lobe/lobule more susceptible to endo- or exogenous oncogenic stimuli than lobes or lobules of the same mamma that do not bear or have a lower number of potentially malignant cells [11,20]. One of the most important findings in the sick lobe is that these cells are malignant from the outset [11].
The sick lobe theory is supported by histopathological studies in women reporting that the majority of breast carcinomas are multifocal and of variable size [11]. These results are supported by modern diagnostic imaging techniques in which 40% of the disease is multifocal. The multifocality and size of carcinomas are considered important prognostic factors of the biological behavior of this disease in women [21,76]. Our results indicate that this process also likely occurs in female dogs and that similar studies on the biological behavior of these lesions should be performed in canines. A recent, predominantly epidemiologic report that included clinical and histopathologic data from 90 female canines with 296 tumors hypothesized those malignant mammary tumors could develop from pre-existing low-risk IELs and that the malignant tumors would be the final stage [77]. Although the authors of the report described that within the same MG there is substantial heterogeneity and that the benign and malignant lesions are multifocal and have variable size, they did not interpret the meaning of these findings. Similarly, Mouser [17] and Sorenmo [77] also reported the multifocality and simultaneous or asynchronous character of IELs and mammary tumors but did not offer any explanations for these changes.
Several additional changes that equate with those of woman breast carcinoma were observed in this work: First, as in female carcinoma [28], we observed a significant increase in the focal interruptions of surrounding myoepithelial cells at early stages in IELs and incipient invasive mammary carcinomas; moreover, these interruptions were more frequently associated with morphologically malignant acini or ducts (Figure 10 and Figure 11). Second, the invasion signs to the stroma show that the epithelial cells adopt a fusiform appearance as in human breast carcinomas [59]. Proliferative malignant cells in ICA in this study showed a similar morphology, and although most of them projected into the duct´s lumen unexpectedly, some some of them appeared to return to the wall of the duct, break the basal membrane and invade the stroma (Figure 5). We are not aware of any reports of such behavior of malign epithelial cells in canine mammary carcinoma. Third, invasive cells exhibit characteristically nuclear changes associated with malignity, such as a loss of polarity, a prominent nucleolus, an irregular shape, and megakaryosis [78]. Such changes were common in malignant epithelial cells in this study. These findings suggest that the linear model of tumor progression cannot be applied in every case [79] and that these spontaneous lesions are malignant from the beginning [19,20].
In conclusion, this study provides evidence that canine IELs presented in Bogota with significant frequency. Similarities in the morphology of these lesions with that reported in women were demonstrated. This study provides evidence that female dogs with healthy mammary glands from Bogota, have considerable numbers of IELs in their MGs and that many have a malignant character from onset. Additionally, the current classification of IELs in female dogs should be reviewed. Our findings provide evidence to propose a new hypothesis for mammary gland carcinogenesis in dogs in accordance with recent theories of the origin of breast carcinomas in humans, i.e., the theory of the SL and biological timing, highlighting the similarities in pathologies between these species. This study also provides evidence of the epithelial-mesenchymal transition of malignant cells at the invasive front of the ICA. The relevance of myoepithelial cells during the early development of IELs and ICA in canines was also demonstrated. This information argues for a method of local molecular characterization of these lesions in canines for comparative studies of canines and women.
We thank the Veterinary Pathobiology Group, Leonardo and Gilberto, for their tireless assistance.
This study was approved by the Institutional Animal Care and Use Committee of the Faculty of Veterinary Medicine and Zootechnology, National University of Colombia, Bogota.