Leiomyosarcomas (LMS) are rare tumors associated with poor outcomes. Treatment of the condition is challenging due to limited cases in the literature. Here, we present a case of a 46-year-old male who presented with left lower limb deep vein thrombosis, with an ultrasound scan revealing a right renal mass. MRI demonstrated a large, well-circumscribed lesion engulfing the middle and lower right renal poles associated with an invasion of the related part of the inferior vena cava (IVC), right renal vein and IVC tumor thrombus in the retrohepatic segment and left renal vein. The patient underwent right radical nephrectomy and IVC thrombectomy suprarenal and IVC resection infrarenal till both common iliac veins. Subsequent pathology was consistent with an LMS of the inferior vena cava. We document this case and discuss its implications.
Leiomyosarcoma, Inferior vena cava, Renal mass, Nephrectomy
Leiomyosarcomas (LMS) are rare tumors, accounting for just 6% of all sarcomas [1]. Currently, around600 cases of LMS of the inferior vena cava (IVC) have been reported worldwide [2,3]. Usually diagnosed in the 5th or 6th decade of life, LMS of the IVC is predominant among females, with a 5:1 to 3:1 ratio of females to males [4,5]. Here, we present a case of a 46-year-old male who was found to have a 14 cm right renal mass. Pathological findings following a right radical nephrectomy and IVC thrombectomy confirmed LMS of the IVC.
A 46-year-old man, previously healthy, presented to the surgical oncology outpatient clinic in the Kuwait Cancer Control Center in July 2021 with left lower limb deep vein thrombosis (DVT) and was admitted to the hospital. Incidental findings of the abdomen and pelvic ultrasound revealed right renal mass and DVT involving the IVC and bilateral common iliac and femoral veins.
MRI revealed a large, well-circumscribed lesion engulfing most of the middle and lower right renal poles associated with an invasion of the related part of the IVC (extending cranially and ending at the retrohepatic segment), right renal vein, and mid-length of the left renal vein (Figure 1). Subsequent signal void was appreciated in the lower segments of the IVC as well as the examined parts of bilateral common iliac veins. The tumor measured around 14.0 x 12.5 x 9.0 cm in maximum dimensions.
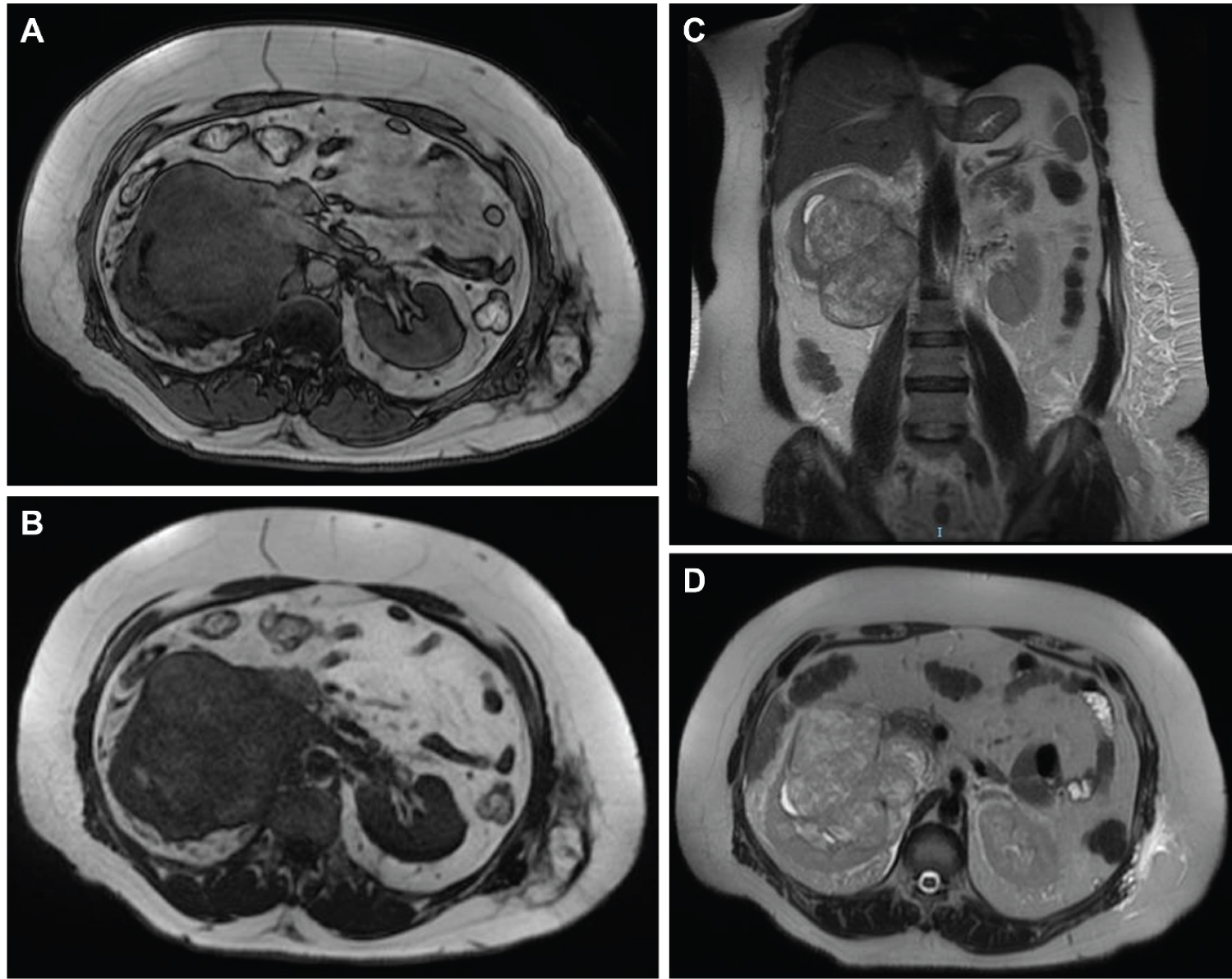 Figure 1: MRI urography. (A) Axial view 3D; (B) Axial view T1; (C) Axial view T2; (D) Coronal view T2. A well-circumscribed lesion engulfing most of the middle and lower right renal poles associated with an invasion of the related part of the IVC (extending cranially and ending at the retrohepatic segment), right renal vein, and mid-length of the left renal vein.
View Figure 1
Figure 1: MRI urography. (A) Axial view 3D; (B) Axial view T1; (C) Axial view T2; (D) Coronal view T2. A well-circumscribed lesion engulfing most of the middle and lower right renal poles associated with an invasion of the related part of the IVC (extending cranially and ending at the retrohepatic segment), right renal vein, and mid-length of the left renal vein.
View Figure 1
CT scan revealed a large irregular well-circumscribed lesion seen epic entered within the right lower renal pole invading the related part of the IVC (extending cranially and ending at the retrohepatic segment), right renal vein, and mid-length of the left renal vein (Figure 2), Subsequent filling defect is appreciated at the intrahepatic segment of the IVC, as well as the whole length of the IVC, whole length of the left common iliac, external iliac and demonstrated part of the femoral vein, and the proximal segment of the left common iliac and proximal part of left internal iliac veins. The upper border of the lesion was seen abutting the hepatic capsule at segment VI with no definite line of cleavage appreciated. The lesion was heterogeneously enhanced in the post-contrast images, measuring 9.7 x 12.5x13.5 cm.
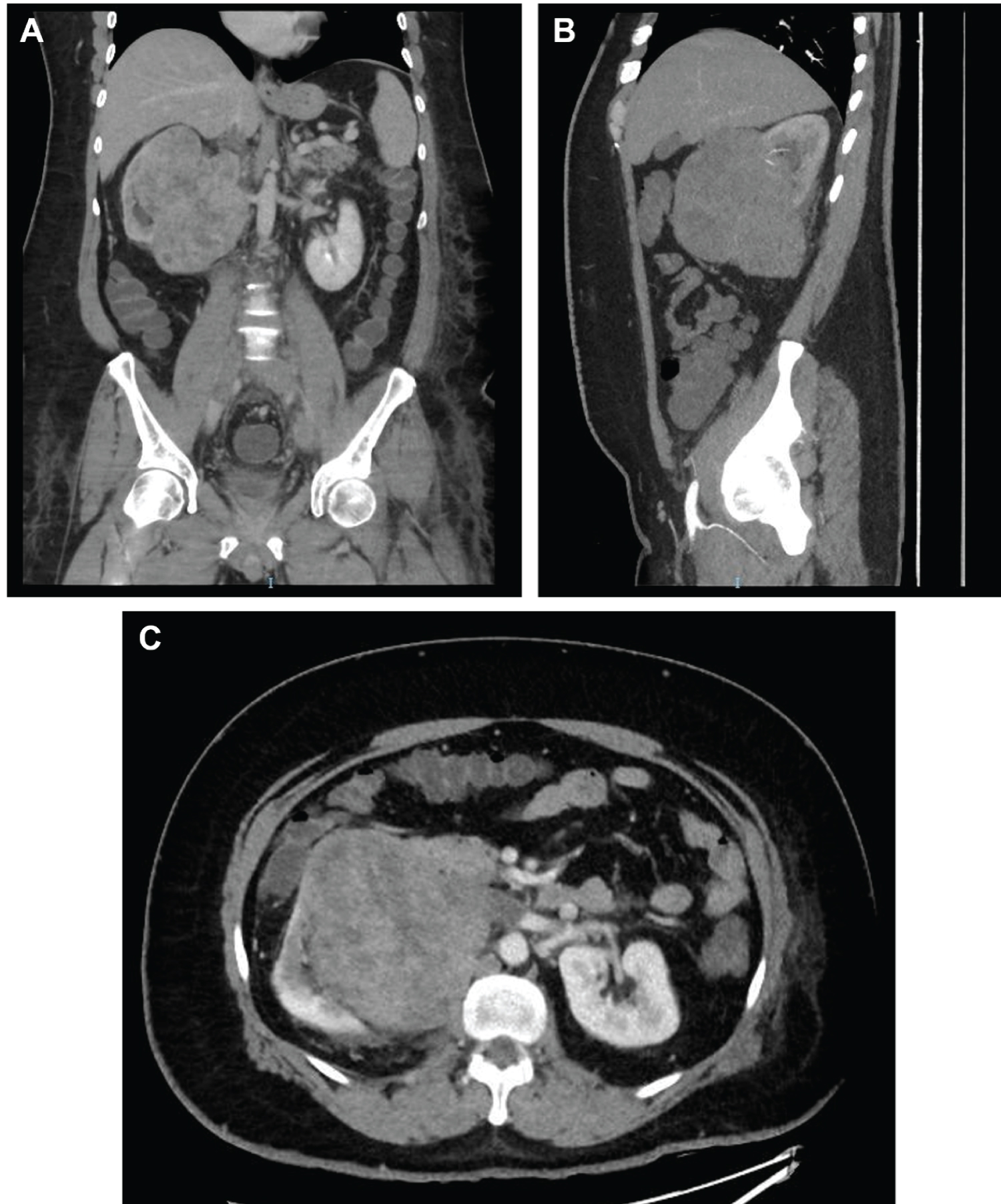 Figure 2: Computed tomography angiogram of the abdomen and pelvis. (A) Coronal venous phase; (B) Sagittal view (maximal intensity projection); (C) Venous phase; (D) Venous phase tumor thrombus (black arrow) in retrohepatic inferior vena cava (IVC) just below the confluence of the hepatic veins.
View Figure 2
Figure 2: Computed tomography angiogram of the abdomen and pelvis. (A) Coronal venous phase; (B) Sagittal view (maximal intensity projection); (C) Venous phase; (D) Venous phase tumor thrombus (black arrow) in retrohepatic inferior vena cava (IVC) just below the confluence of the hepatic veins.
View Figure 2
Renal cortical scintigraphy showed normal morphology of left kidney (62% function) and right kidney showed a large photon-deficient area occupying its lower portion partially extending to mid-portion and encroaching on the renal pelvis with upward lateral displacement of the kidney, the remaining part of the right kidney showed good cortical uptake (38% function).
The patient was kept on therapeutic Clexane 0.8 BD (Enoxaparin sodium) subcutaneously. The patient was prepared for elective surgery and underwent a right radical nephrectomy and IVC tumor thrombectomy in August 2021 (Figure 3).
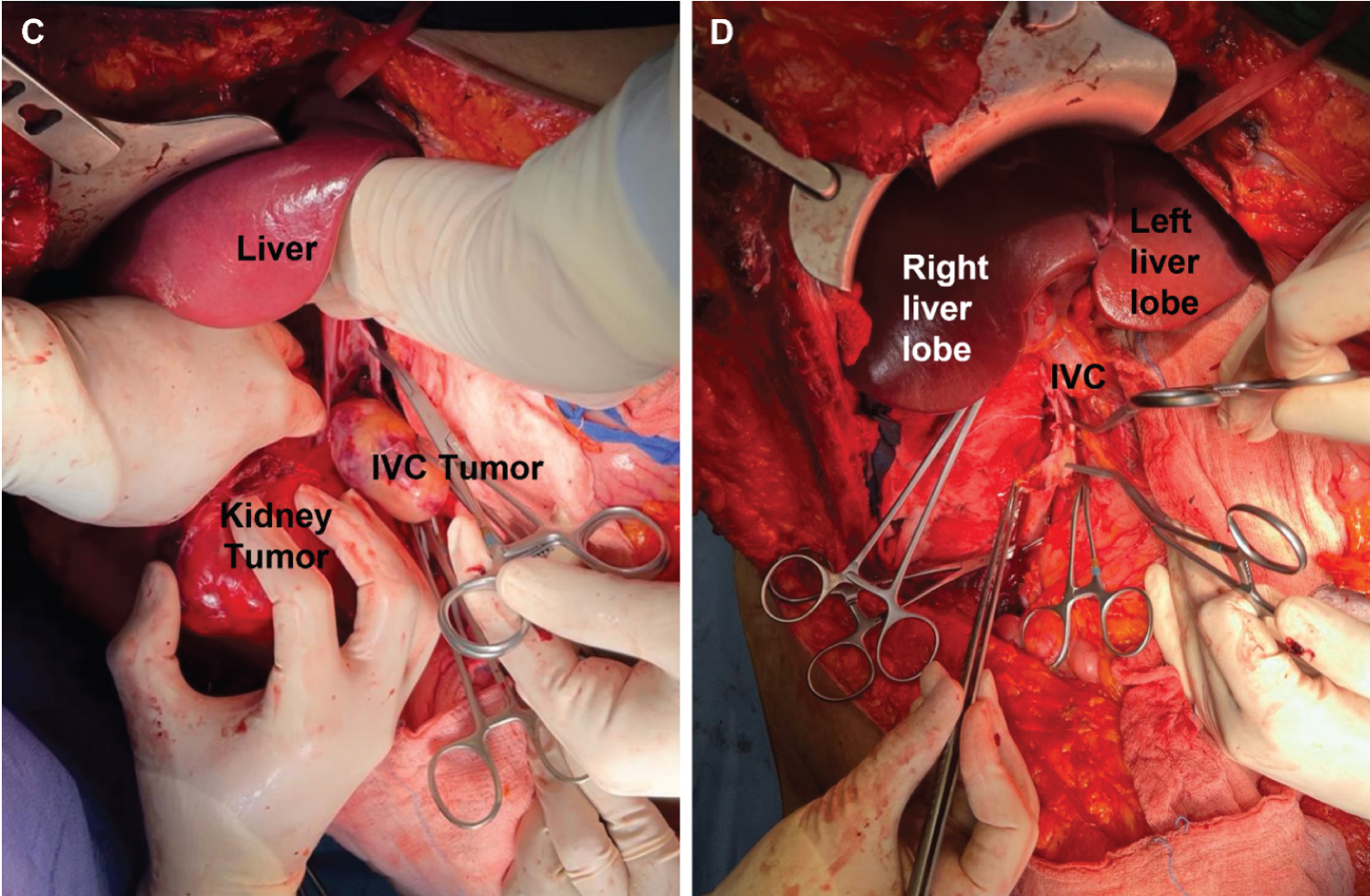 Figure 3: Intraoperative pictures of IVC leiomyosarcoma. (A-B) Exposure of the inferior vena cava (IVC) and kidney tumors. (C-D) Resected IVC tumor thrombus, veinotomy, and IVC reconstruction. A part of IVC resected, right renal vein, and IVC infrarenal bifurcation of the common iliac veins and till the level of the bifurcation of both internal and external iliac veins.
View Figure 3
Figure 3: Intraoperative pictures of IVC leiomyosarcoma. (A-B) Exposure of the inferior vena cava (IVC) and kidney tumors. (C-D) Resected IVC tumor thrombus, veinotomy, and IVC reconstruction. A part of IVC resected, right renal vein, and IVC infrarenal bifurcation of the common iliac veins and till the level of the bifurcation of both internal and external iliac veins.
View Figure 3
The surgical procedure and intraoperative findings were as follows. Under general anesthesia, the patient was placed in the supine position. After skin preparation and draping, a bilateral subcostal with upper midline extension incision the classic Mercedes incision was performed. Intraoperatively, a large right renal mass infiltrating the psoas muscle with adhesion to the 2nd and 3rd parts of the duodenum without infiltration was noted. No ascites or peritoneal disease were observed. Proximally, an IVC tumor thrombus extending from the retrohepatic part till below the hepatic veins (level III), with the distal part reaching the confluence of the common iliac veins, was noted. Para-aortic lymph, common iliac, and external iliac node enlargements were also noted. Next, the ascending colon was mobilized, and the duodenum identified. The liver was mobilized after reaching its ligaments, and the piggyback technique was done to explore the retrohepatic IVC. Following identification of the left renal vein, the IVC was dissected till the level of the hepatic veins, proximally and distally. After vascular control, the IVC was opened, and the tumor thrombus dissected proximally till the right hepatic vein and distally to the level of the left renal vein (extraction of the thrombus that was occluding the left renal vein). Radical nephrectomy was followed by dissection and ligation of the IVC under the level of entrance of left renal vein.
The specimen, with part of the IVC related to the specimen invaded by the tumor, was extracted. The thrombus inside the lower part of the infrarenal IVC was identified. The thrombus was dissected till the confluence of the common iliac vein and para-aortic and iliac lymph nodes. After dissection from the tumor thrombus, the left renal vein was ensured to be patent. A 3-0 prolene suture was used to close the superior part of the IVC and left renal (suprarenal part). Patient received 6 pints packed red blood cells (PRBCs), 4 pints of fresh frozen plasma, 7.5 IVF, and 200 mL albumin intraoperatively. His hemoglobin dropped from 12.4 g/dL preoperatively to 6.1 g/dL but increased to 9.9 g/dL after transfusion. He stayed in the intensive care unit for four days, where he received 3 PRBC. His hemoglobin was stable at 10.3 g/dL and his creatinine was elevated (between 120 to 140 μmol/L). He also had lower limb edema, presumably due to adequate collateral drainage. He was shifted to the surgical ward. The patient was discharged after 16 days without complications, stable hemoglobin (10.3 g/dL), and on therapeutic clexane.
On gross histopathology examination, grossly, a specimen of right radical nephrectomy was received, of the right kidney with an attached right adrenal gland, showing externally an adherent grey-white tumor thrombus in the inferior vena cava. On slicing, there was a grey, white firm mass involving the renal pelvis, measuring about 17 x 14 x 10 cm (Figure 4).
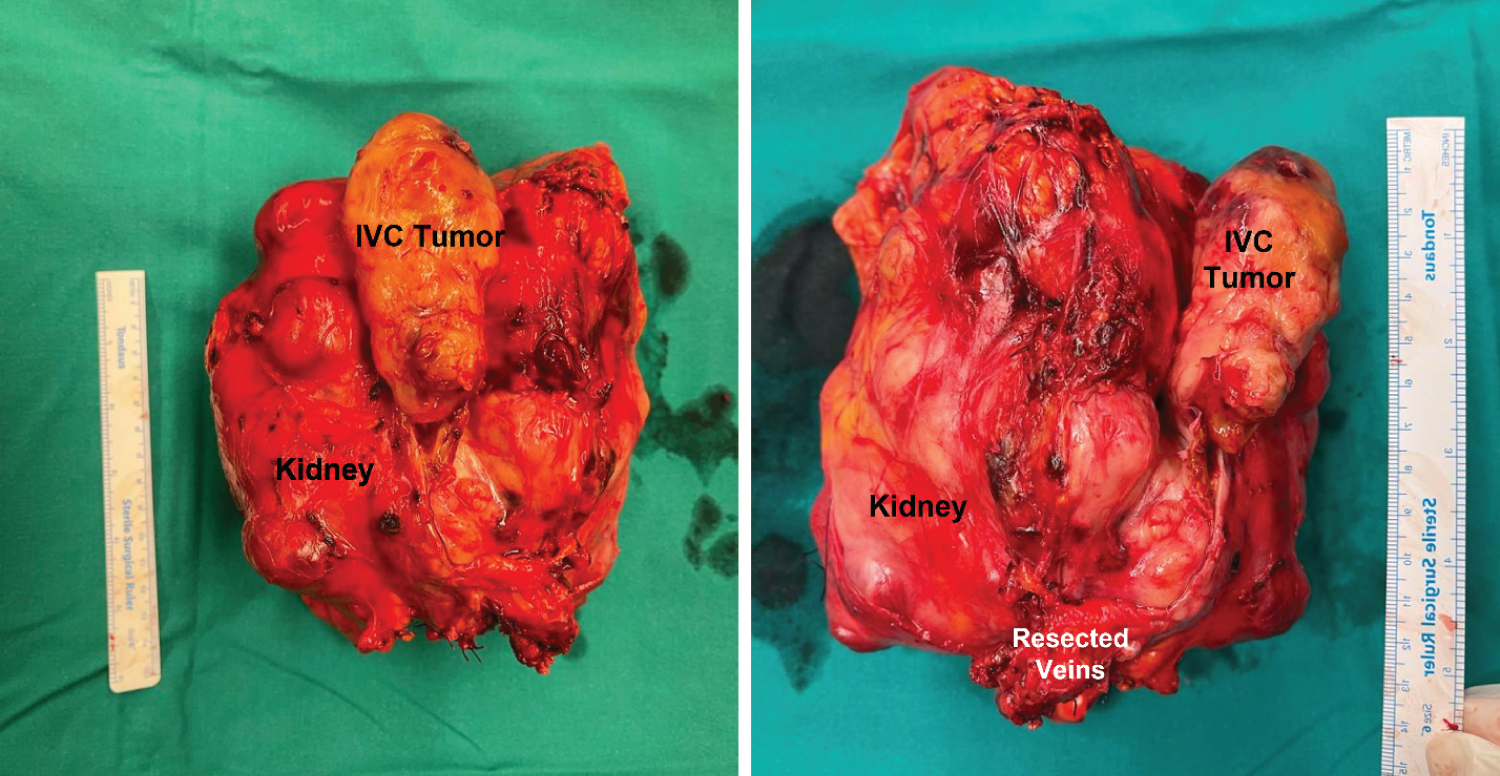 Figure 4: (A-B) Resected inferior vena cava (IVC) segment and kidney with the tumor thrombus following resection.
View Figure 4
Figure 4: (A-B) Resected inferior vena cava (IVC) segment and kidney with the tumor thrombus following resection.
View Figure 4
Microscopic examination revealed a spindle cell sarcoma, composed of spindled cells having cigar-shaped nuclei, scattered mitoses (up to 10-19/10 HPF) and foci of necrosis (about 40%; Figure 5). Focally, the tumor was seen arising from the IVC wall, along with adjacent areas of thrombus formation. Tumor thrombus was also seen in the right internal and external iliac veins. Immunohistochemically, the spindled tumor cells were positive for vimentin, H-caldesmon, focally for SMA, CD34 and desmin, while they were negative for CD117, DOG-1, S100, pan CK, renal cell carcinoma antigen (Figure 6). The overall features were of a spindle cell sarcoma, compatible with leiomyosarcoma, most probably arising from the IVC wall.
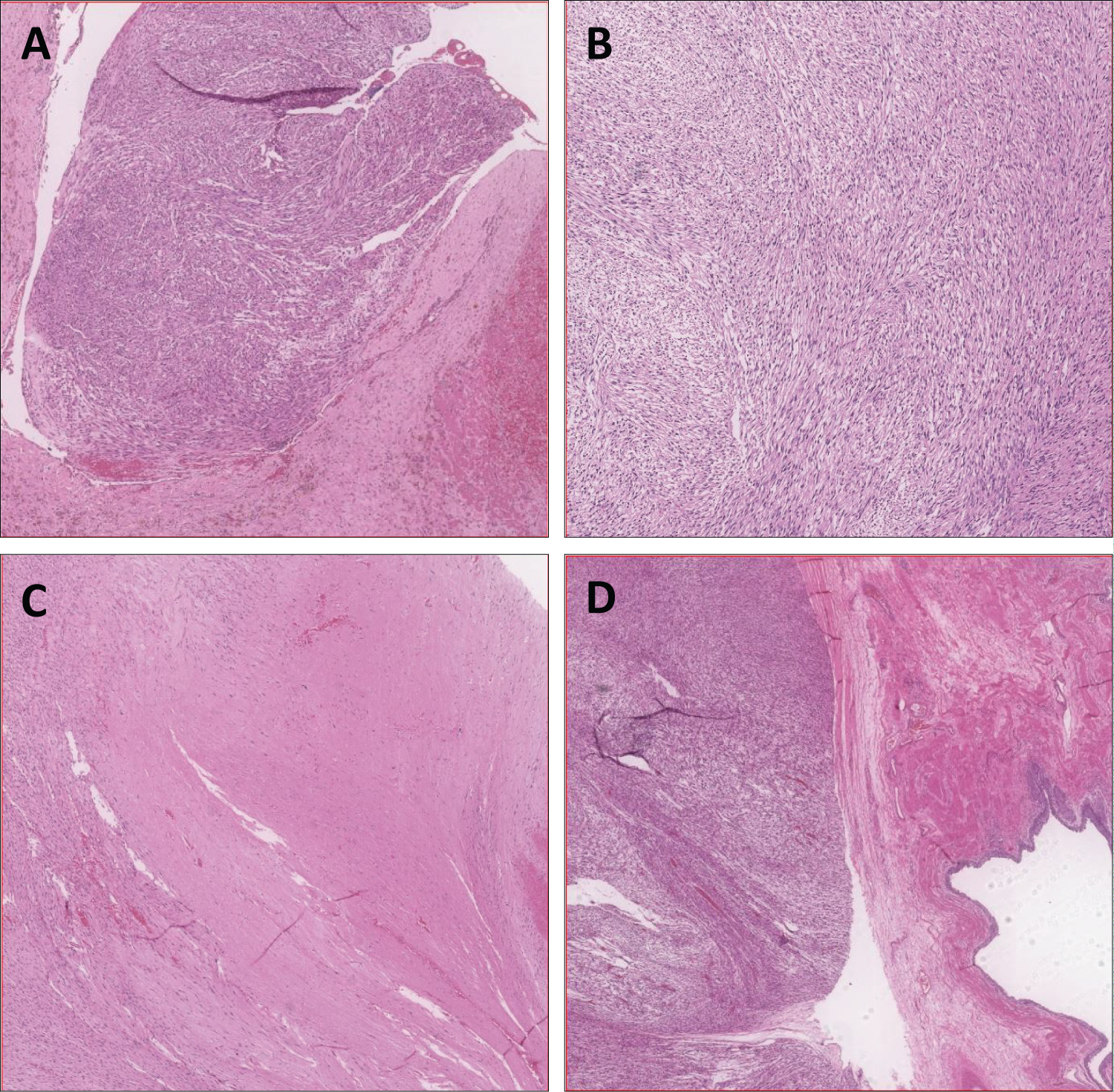 Figure 5: (A) Tumor thrombus with vessel wall; (B) Tumor, high power view showing spindle cells arranged in fascicles;
(C) Tumor with foci of necrosis; (D) Tumor with renal pelvis
View Figure 5
Figure 5: (A) Tumor thrombus with vessel wall; (B) Tumor, high power view showing spindle cells arranged in fascicles;
(C) Tumor with foci of necrosis; (D) Tumor with renal pelvis
View Figure 5
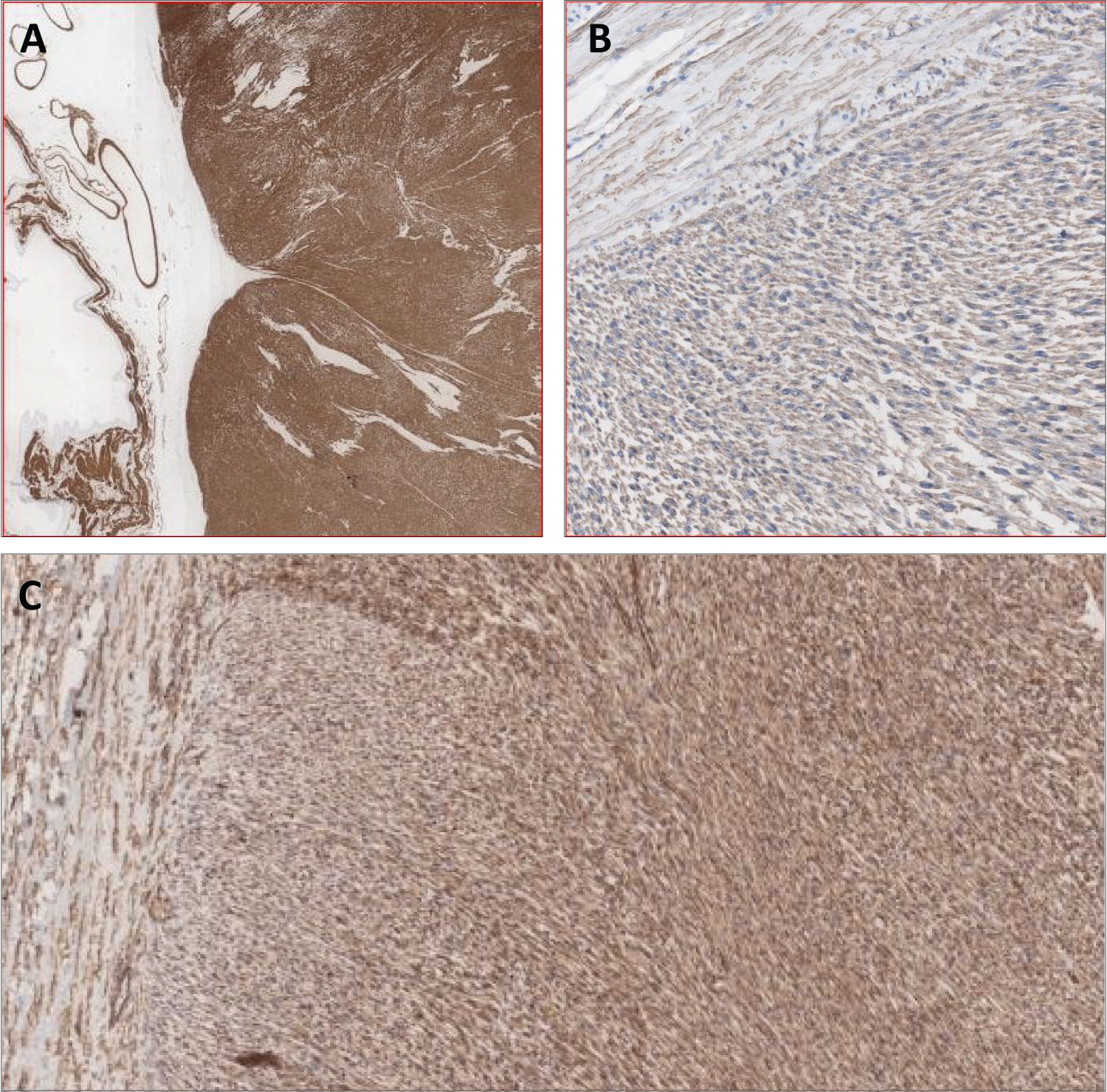 Figure 6: (A) H-caldesmon (strongly positive); (B) SMA (focal weak positive); (C) CD34.
View Figure 6
Figure 6: (A) H-caldesmon (strongly positive); (B) SMA (focal weak positive); (C) CD34.
View Figure 6
The case was discussed during the sarcoma multi-disciplinary team meeting and the consensus was for follow-up. Patient follow-up at three-to-six months post-surgery was uneventful.
Complicating diagnosis is the presence of non-specific symptoms, such as diffuse abdominal pain, vomiting, nausea and general malaise [4,7]. In more than 80% of symptomatic cases, the main reported symptom is lower back pain [4]. In the present case, the patient presented with left lower limb DVT. The non-specificity of the symptom presentation enhances the likelihood of misdiagnosis [8]. This contributes to the increased likelihood of diagnosis occurring in more advanced stages.
The most widely used classification system for IVC tumor thrombus in patients with locally advanced renal cell carcinoma is that that of the Mayo Clinic thrombus classification [9]. In Level I, the tumor thrombus is either at the entry at the renal vein or within the IVC, fewer than 2 cm from the confluence of the renal vein and IVC. Classification as Level II requires that the tumor thrombus extends within the IVC, 2 cm above the confluence of renal vein and IVC but is still below the hepatic vein. Involvement of the intrahepatic IVC leads to classification as Level III. Finally, Level IV classification requires extension either into the right atrium or above the diaphragm [9].
LMS can also be classified as one of three types according to locations in various segments of the IVC [10]. Segment I corresponds to the IVC below the entrance to the renal veins (infra-renal; Figure 7). Segment II, or the middle segment, which is the most commonly affected region [11], corresponds to the IVC located between the end of the hepatic veins and the renal veins. Finally, segment III, or the hepatic segment, corresponds to the IVC located between the right atrium and the hepatic veins. In the present case, the patient exhibited IVC LMS in the middle segment. Patients with localization in the middle segment often present with right upper quadrant pain and nephrotic syndrome due to renal vein drainage obstruction [8].
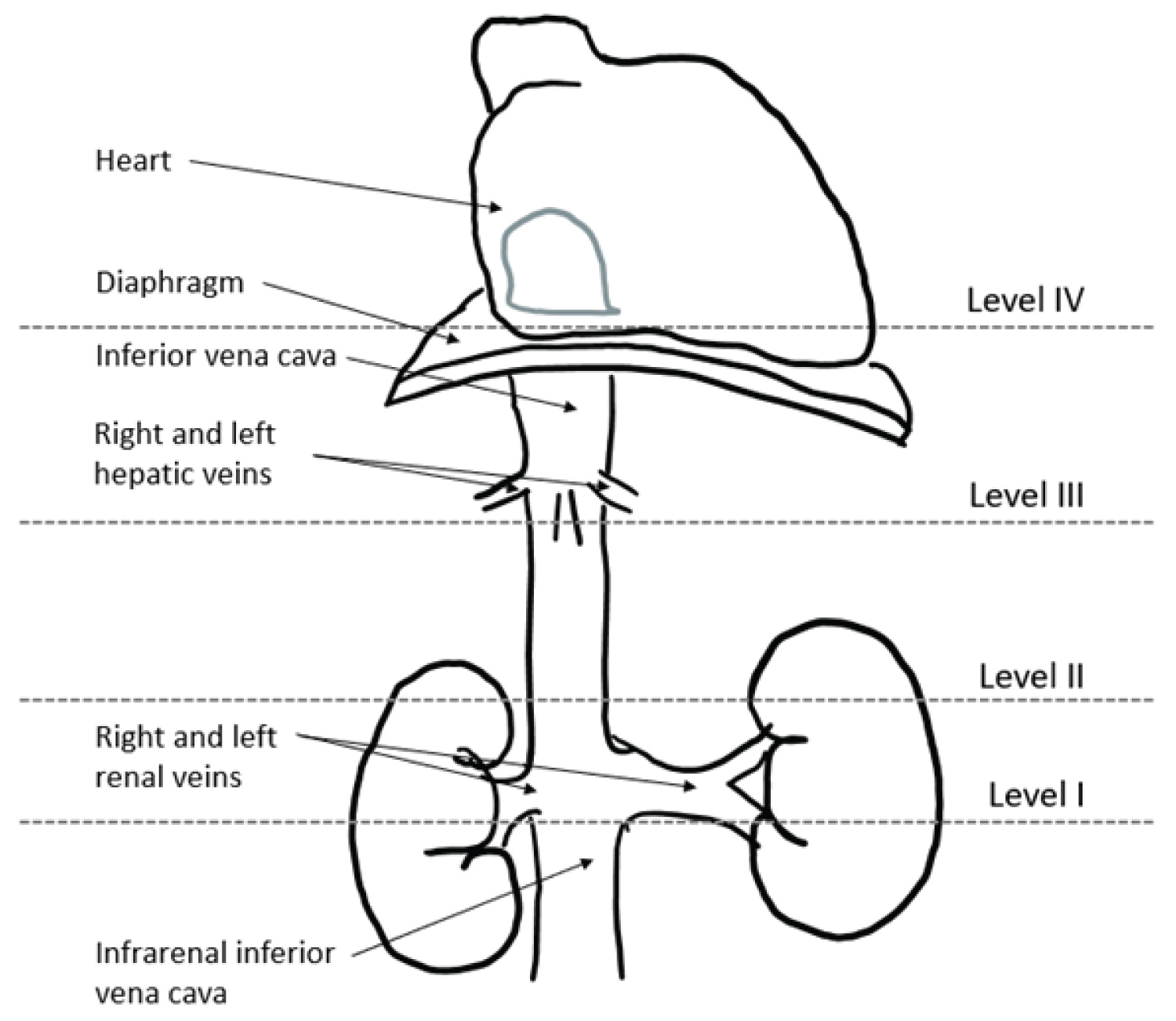 Figure 7: Classification of the inferior vena cava leiomyosarcoma according to (10).
View Figure 7
Figure 7: Classification of the inferior vena cava leiomyosarcoma according to (10).
View Figure 7
In addition, LMS of the IVC exhibits three main tumor growth patterns: extraluminal, which accounts for 62% of all cases; intraluminal (5%); and a combination of both (33%) [6]. Tumor growth is typically slow and lateralized to the right side due to limited resistance of fatty tissue [4]. Differentiated LMS of the IVC exhibits extraluminal growth that may confused with other retroperitoneal tumors [12].
Diagnosis is difficult, particularly in the presence of a predominant extraluminal component [1]. Due to the diagnosis rarity of LMS, no imaging protocols currently exist, and no studies have compared the accuracy of CT versus MRI in diagnosing LMS [8]. The role of MRI is thought to be limited to differentiating neoplastic invasion of thrombus [1] or as an adjunct in characterizing adjacent invasions [13]. By contrast, CT is helpful in deciphering the origin of the tumor as well as invasion and metastasis [8] suggest using non-contrast CT and intravenous contrast-enhanced CT images to aid with imaging in arterial, portal venous, and delayed phases. Characteristic findings on the CT include invasion and dilation of the lumen, as well as irregularly distanced IVC with complex, lobulated, and heterogenous soft tissue mass, are pathognomonic of LMS of the IVC [8,14]. According to Webb et al. [14], diagnostic features of LMS on a CT include the sign of imperceptible cava lumen, which is useful in identifying retroperitoneal mass. A biopsy is not required if the mass is removable [1]. In the present case, MRI urography, CT angiography, and renal cortical scintigraphy were used to aid in identifying and diagnosing the tumor.
Histological and immunohistochemical findings are used to make a definitive diagnosis of LMS [1]. These findings reveal elongated, spindle-shaped cells arranged in a bush and positive for alpha-smooth muscle actin, vimentin, and desmin [15]. In the current case, tumor cells may also be positive for CD34 (Bilgo et al., 2021) [1] and epithelial membrane antigens [16]. Immunohistochemical findings are negative for CD117 and the ki-67 proliferative index is low [17]. In the present case, tumor cells were positive for CD34 and H-caldesmon, and weakly positive for SMA. Some immunohistochemical markers have also been associated with survival and tumor recurrence. For example, high expression of cytoplasmic B-catenin and IGF-1R may be predictive of recurrence and poor survival [18].
In the present case, the patient underwent a right radical nephrectomy and IVC tumor thrombectomy on the assumption that it was a renal tumor. Due to the rarity of LMS of the IVC, information pertaining to effective treatment approaches is largely based on case studies as opposed to comprehensive systematic analyses. Consequently, there are no existing guidelines for management [8]. Because LMS of the IVC appears to be resistant to radiation and chemotherapy, though there have been reports of successful neoadjuvant chemotherapy and radiation before resection [19], radical en bloc resection with adequate margins is the main treatment approach [11,19,20]. Although an R0 excision with confirmation of venous return to concerned areas minimizes recurrence [21], complications following surgery are frequent, with up to 20% reporting lower extremity edema or renal failure [3]. As in the present case, patients often receive anticoagulation therapy, either prophylactically or long term [8].
Prognosis following surgery remains poor, with a 5-year survival rate of less than 50% and a 10-year survival rate of less than 30% [22]. Recurrence is a significant problem, with 25 months being the median time-to-recurrence [5,21]. In LMS of the IVC, hematogenous metastases are frequent and occur via the lymphatic system. Common metastatic locations include lung, liver, and lymph nodes. lung, though lymphatic dissemination is rare [13]. Predictors of poor short-term survival include level III tumor staging, macroscopically incomplete surgery, endoluminal tumor bud presence, right atrial extension of the tumor, and compromised liver function [23].
LMS of the IVC is a rare tumor that is predominant in women. Here, we presented a case of LMS of the IVC presenting as a renal mass in a 46-year-old man. Clinical symptoms were nonspecific. An MRI identified a 14 cm renal mass and invasion of the nearby IVC. Although management practices are not well delineated owing to the rarity of the condition, our patient was managed via right radical nephrectomy and IVC thrombectomy. The patient did not receive post-operative neoadjuvant therapy. Histopathology confirmed diagnosis of LMS of the IVC. Despite successful outcomes, ongoing follow-up is required as recurrence is frequent.
Ethical approval and consent to participate: Informed consent was obtained from subject.
Consent for publication: Consent for publication was obtained from subject.
Competing interests: The authors declare that they have no conflicts of interest.
No financial support.
No acknowledgements.