Drusen are pathognomonic of some ocular pathologies such as age-related macular degeneration (AMD), familial dominant drusen, best macular dystrophy (BMD) and optic nerve drusen. The presence of drusen in the peripheral retina may be an early sign of AMD. This article reports a rare case of peripheral retinal drusen occurring in a similar pattern in both eyes of a forty-year-old man with unaided visual acuity of 6/4 in each eye. Optical coherence tomography findings showed bumps between the retinal pigment epithelium and the Bruch's membrane in the peripheral retina of both eyes. In AMD, drusen are found on the macula and cause early onset of impaired central vision for near and distance, whereas peripheral retinal drusen are found in conditions such as familial dominant drusen involves the macula at a later stage. The distribution of the drusen in this case report is different from what is found in AMD and its variants or familial dominant drusen and BMD hence, these lesions may be congenital. The genetic basis of drusen in these pathologies has been studied and certain genes identified, therefore genetic testing is needed to fully describe the clinical relevance of the drusen found in this report to assess the risk of developing AMD.
Drusen, Retinal pigment epithelium, Genes, Age-related macular degeneration, Congenital, Glaucoma
Drusen are yellowish focal deposits found between the retinal pigment epithelium (RPE) and the Bruch's membrane [1]. A subtype of drusen known as subretinal drusenoid debris (SDD) is found in the subretinal space and forms sharp, broad elevations. SDD is found in geographic atrophy of dry AMD and type-3 neovascularization of wet AMD. They cause outer retinal atrophy leading to poor dark adaptation and poor contrast sensitivity [2,3]. Drusen are made up of carbohydrates, lipid zinc and about one hundred and fifty proteins [1,4].
Drusen can be described as hard, soft, cuticular, and subretinal drusenoid debris (SDD). Hard drusen have distinct borders and measure about 63 μm in diameter according to Wisconsin grading system. Soft drusen have a diameter between 63-125 μm and can be associated with choroidal neovascularization in advanced AMD. Cuticular drusen are multiple densely packed structures of about 50-75 μm in diameter. They contain the same materials as soft drusen and are found under the RPE. They attenuate blue light in their spectral profile [1,3].
The theories of formation of drusen include: Transformation theory, deposition theory and vascular theory. Donders postulated that the retinal pigment epithelium (RPE) is directly transformed to drusen. During the budding process of the RPE cells, some buds formed by RPE contains cytoplasm and plasma membrane, attaches the buds to the cytoplasm of the parent RPE cell which then penetrate into the Bruch's membrane. These buds later separate from the RPE cells and form disintegrations. Their disintegrations form drusen in the Bruch's membrane [3]. Muller suggested that drusen were exuded by RPE as waste products. The degeneration process of RPE cells leads to the formation of cellular debris that are deposited on the Bruch's membrane. These debris form inflammatory stimulus that in turn serve as a site for drusen formation [5]. Friedman, et al. in explaining the vascular theory of drusen formation postulated that drusen are formed from components of blood as a result of an unusual expulsion of waste products by the RPE cell. They noted that the choroidal collecting venules and drusen were closely associated to each other. As aging progresses, there is a reduction in the choroidal blood flow due to a reduction in the thickness of the choroids which then leads to senescence of RPE cells, accumulation of waste products which eventually leads to drusen formation [4]. Ocular conditions that cause drusen include: Age-related macular degeneration (AMD) and its variants, familial dominant drusen, Best vitteliform macular dystrophy and optic nerve drusen.
Age-related macular degeneration (AMD) is an age related maculopathy which causes central visual impairment that causes visual impairment and is the highest cause of irreversible blindness in the older population of the developed world [6]. The Complement Factor H (CFH) and Ala69Ser (LOC387715) are genes that have been found to be responsible for 50-75% of AMDs with the CFH gene being more important [7]. Drusen are seen as the earliest clinical feature of age-related macular degeneration (AMD). There are two types of AMDs; neovascular (wet) and non-neovascular (dry). Dry AMD has features which includes thinning of the macula and formation of tiny drusenoid deposits and causes gradual progressive loss of vision, whereas wet AMD has features of geographic atrophy (GA), pigment epithelial detachment which may be serous, haemorrhagic, fibrovascular or drusenoid and choroidal neovascularization which causes rapid visual loss due to choroidal neovascularization at the macula [8,9]. An early form of AMD known as age related maculopathy (ARM) is commoner among the younger age groups and typically presents with soft drusen found on the macula in both eyes. In ARM, vision is preserved till when it eventually progresses to AMD when the is deterioration of vision [10]. There are two main forms of early onset drusen typically presenting before the fourth decade of life; Doynes honeycomb retinal dystrophy (DHRD) and Malattia Leventinese (ML). Other variants of early onset drusen are Stargardt disease and Best disease. Drusen are also found in membranous proliferative glomerulonephritis type II [11].
Doynes honeycomb retinal dystrophy (DHRD) and Malattia Leventinese are inherited macula dystrophies characterized by the presence of drusen on the macula. In DHRD, the drusen are found around the optic nerve head while in ML, the drusen are arranged in a radial pattern from the fovea centralis [10]. The genetic defect in DHRD and MLis caused by a single autosomal dominant mutation in EFEMP1 (EGF-containing fibrillin-like extracellular matrix protein 1) gene. This gene encodes the protein fibulin-3 [12]. Patients may have a blurry vision at an early stage but advanced stages there is severe visual impairment [12,13].
Stargardt disease (STGD) is the highest occurring autosomal recessive macular dystrophy in children and young adults with a prevalence of about 1 in 8000-10,000. It is sometimes seen in late-adulthood. The clinical features of Stargardt disease is bronze beaten appearance of the macular that leads to a progressive bilateral visual impairment. It occurs as a result of mutations of the ABCA4 gene located on the chromosome 1. This gene codes for an ATP-binding cassette membrane protein, that helps in the transport of all trans-retinal aldehydes. The mainstay of management of this condition is the use of low vision aids although research is ongoing in the areas of gene therapy, stem cell therapy and the use of pharmacological agents [14,15].
Best disease or best macular dystrophy (BMD) is a macular degeneration caused by a mutation in the RPE gene BESTROPHIN (BEST1). This leads to the accumulation of lipofuscin-like material in the RPE, causing a degeneration of the RPE and photoreceptors. There is no medical or surgical management of Best disease, however, anti-VEGF therapy has shown to improve conditions [16,17]. The subtypes of Best disease are Best vitelliform macular dystrophy (BVMD), Adult vitelliform macular dystrophy (AVMD) and autosomal dominant vitreoretinochoroidopathy (ADVIRC). In these disease conditions, drusen are found on the macula and there is progressive loss of central vision. Also in ADVIRC the pathology may be associated with some developmental ocular anomalies such as nanophthalmos, microcornea, congenital cataracts and angle closure glaucoma. AVMD develops in the third and fourth decades of life [17].
Optic disc drusen (ODD) is a hereditary anomaly in which calcified, protein deposits are found in the optic nerve head. They are generally bilateral in about 70% of cases. These drusen usually do not cause impairment of vision but some patients may lose peripheral vision. Unlike drusen found in AMDs, they are mostly found in the younger population and are not related to aging. No treatment has been proven to alter ODD but lowering of the intraocular pressure which serves as a neuroprotective treatment and vasoactive therapy such as pentoxifylline has helped to manage this condition [18,19].
A forty-year-old banker who presented with difficulty reading text prints on his phone and laptop at work for about one year duration. His near vision gets clearer with the use of reading glasses or when the text size is large. There was no reduction in vision for distance or reduction in color vision. There was no known history of previous ocular pathology or surgery. There is a family history of glaucoma in the father who is on anti-glaucoma medication. However there is no family history of blindness. Unaided distance visual acuity was 6/4 in both eyes and near visual acuity was N8 at 33 cm which improved to N5 with a Pl/Add of +1.00D binocularly. Slit lamp biomicroscopic examination of the eyes revealed normal anterior segment, normal pupillary reaction bilaterally, intraocular pressure measured at 10 a.m. with a Goldmann applanation tonometer was 12 mmHg and 14 mmHg in the right and left eyes respectively. Examination of the fundus using a 90D handheld super field lens revealed pale disc, cup to disc ratio of 0.7 by 0.7, pink neuroretinal rim, normal macula and vessels in the right eye and similar findings in the left eye. These fundus findings are suspicious for glaucoma so the following investigations were requested: Gonioscopy, pachymetry, central visual field analysis and optical coherence tomography of the optic nerve. Gonioscopy showed open angles up to the ciliary body in all quadrants of both eyes and pachymetry revealed central thickness of 518 and 520 micrometers in the right and left eye respectively while central visual fields and optical coherence tomography were normal. He is being followed up for possible features of glaucomatous changes and presbyopic spectacles were prescribed and dispensed (Figure 1, Figure 2 and Figure 3).
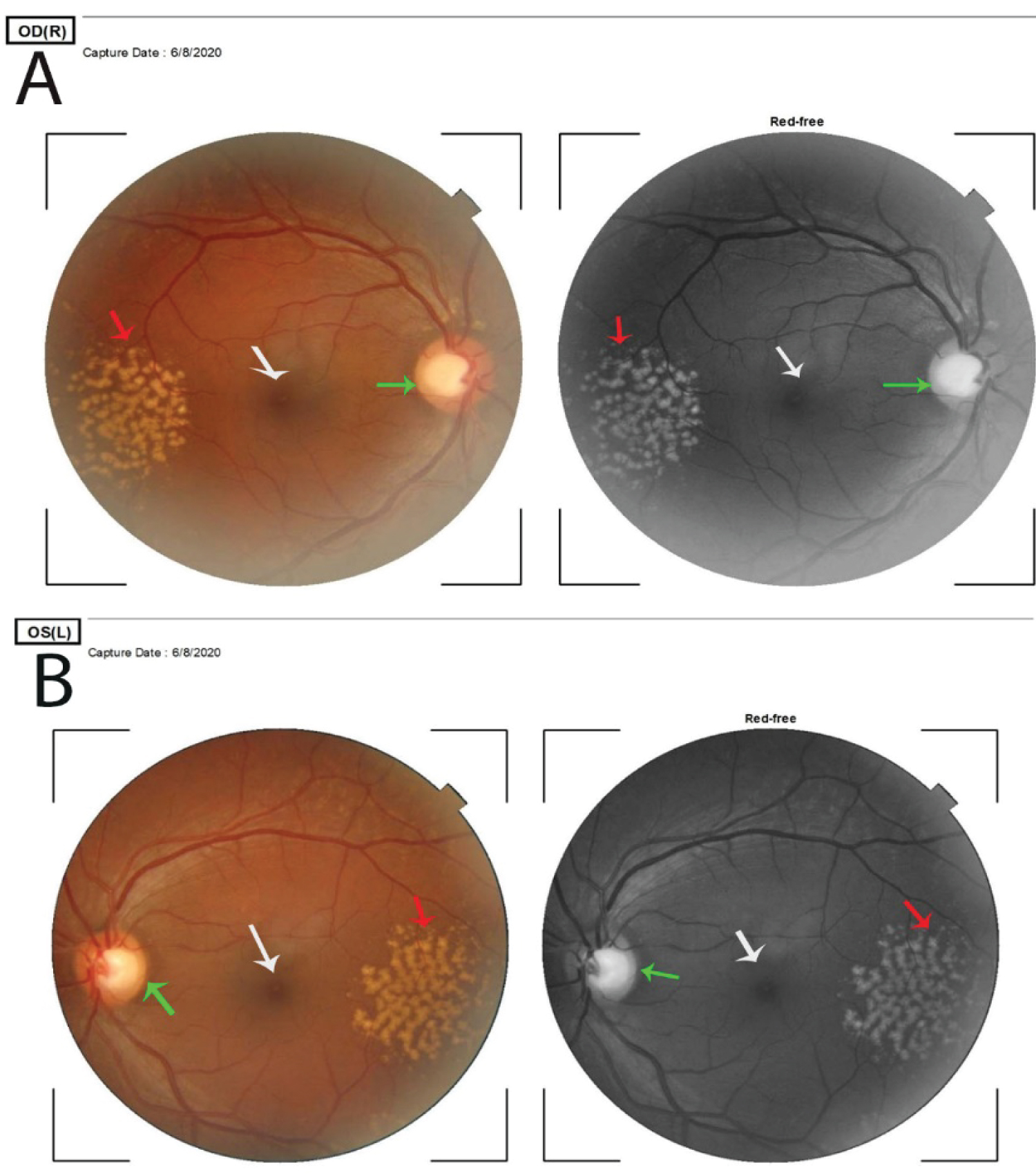 Figure 1: Pane A is the right eye and B the left eye.
View Figure 1
Figure 1: Pane A is the right eye and B the left eye.
View Figure 1
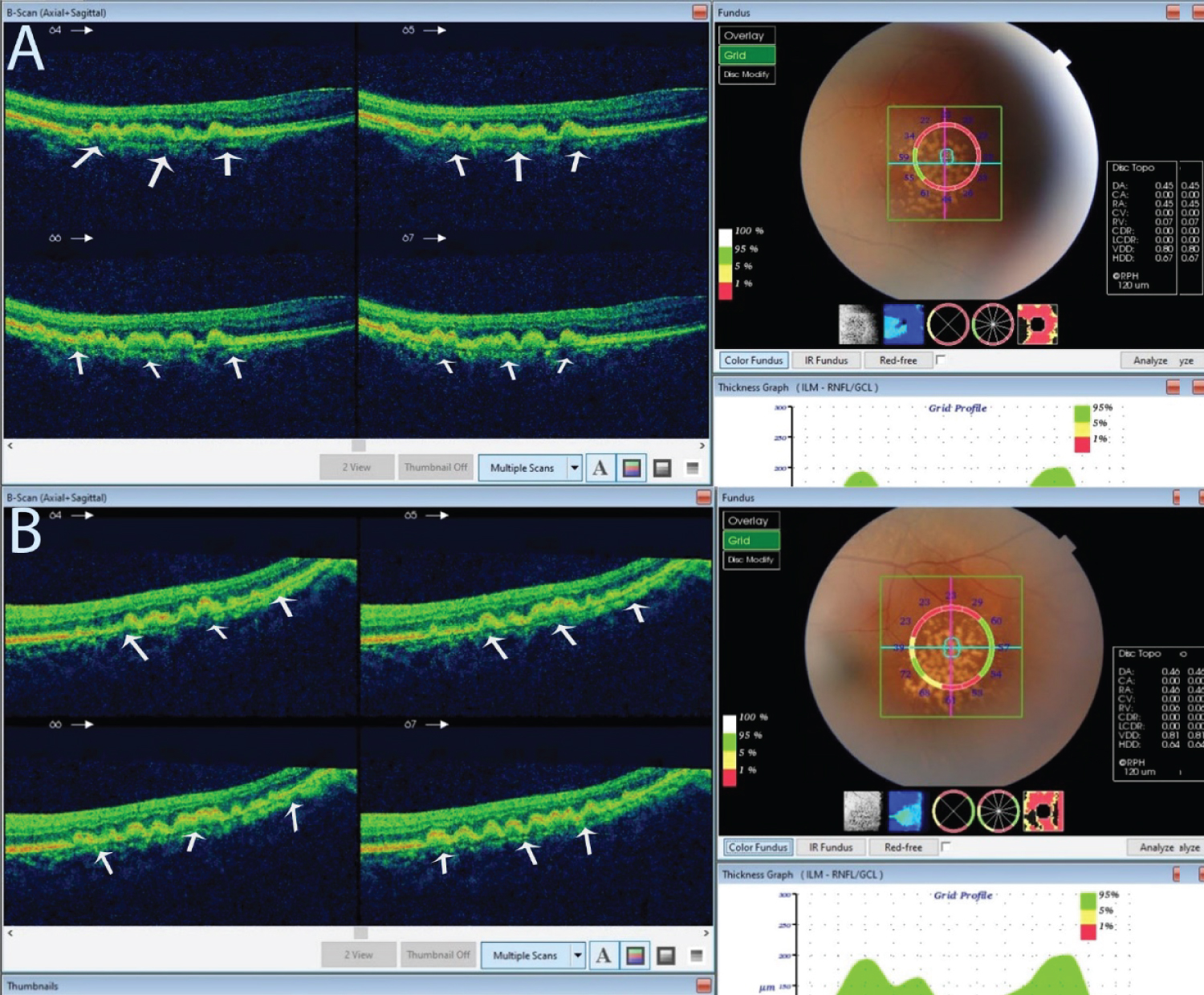 Figure 2: An optical coherence tomography of the lesion described in Figure 1.
View Figure 2
Figure 2: An optical coherence tomography of the lesion described in Figure 1.
View Figure 2
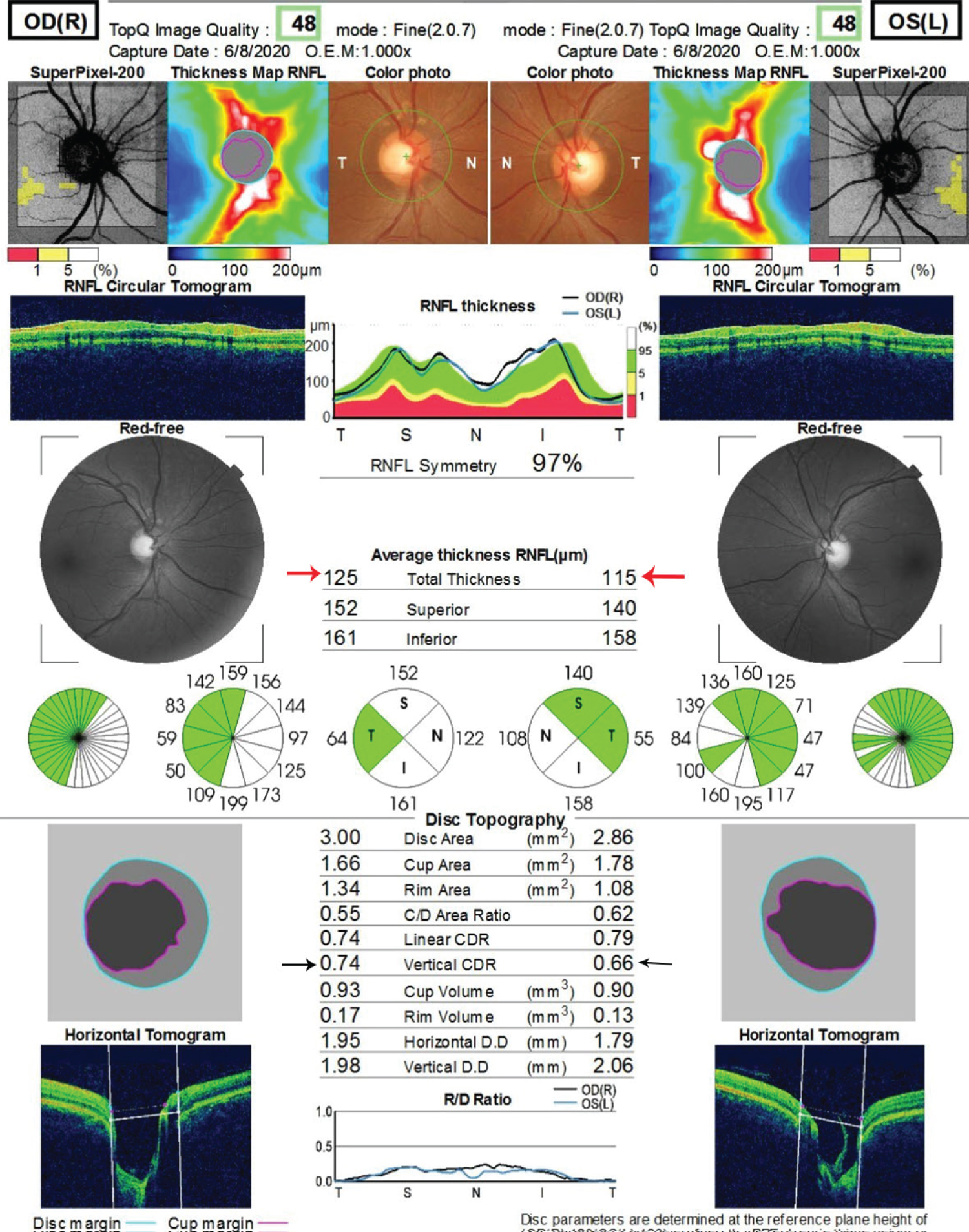 Figure 3: The optical coherence tomography of the optic nerve head of both eyes.
View Figure 3
Figure 3: The optical coherence tomography of the optic nerve head of both eyes.
View Figure 3
They show colored and red-free fundus photographs of both eyes. The macula appears normal in both eyes and is indicated by white arrows. There are bilateral symmetrical peripheral retinal drusen that are indicated by the red arrows. The diameter of the druse is wider than that of the retinal capillaries which indicates that they are soft drusen. The drusen measure about 5 × 4 mm in both eyes. The optic discs in both eyes are cupped and suspicious for glaucoma, indicated by the green arrows. The retinal vessels are normal.
Pane A is the right eye and B the left eye. Both show bumps between the retinal pigment epithelium and the Bruch's membrane. These bumps are drusen, indicated by the white arrows.
Average thickness of the RNFL in both eyes is within normal limit, indicated by the red arrows. The difference in the vertical cup to disc ratio in both eyes is less than 0.2. This is indicated by the black arrows.
Drusen are yellow deposits found under the RPE cells in age-related macular degeneration (AMD) which occur mostly in individuals aged 50-years and older. Drusen formed in AMDs and its variants are normally found around the macula and thus lead to central visual impairment. In younger age groups like in this case report, the presence of peripheral drusen will point to the differentials diagnosis of best macular dystrophy. In these differentials however, there is usually an involvement of the macula and subsequent loss of central vision in as the patient gets older. The index case did not have any deterioration of central vision.
In a study by Johanna, et al. [20] the relationship between peripheral retinal drusen and AMDs in family members and twin were studied and it was observed that peripheral retinal drusen may be found in patients with or without AMD. In subjects that developed AMD, there was a link to a subset of AMDs due to a genetic modification in the following gene loci; CFHY402H, CFHrs1410996, LOC387715A69S whereas in subjects who did not develop AMD, it was postulated that the drusen remained innocuous as long as the macula was not involved. It is possible that the subject of this report falls within the latter category. The association of peripheral retinal drusen to AMD is higher with soft drusen. Soft drusen generally measure more than 63 μm in diameter and have been linked to CFHY402H gene mutation that leads to AMD. The lesions in the index case have the appearance of soft peripheral drusen which may be linked to CFHY402H gene mutation. Small hard peripheral drusen are not associated with various known forms of AMD and may indicate other conditions such as familial dominant drusen [10,21]. The distribution of drusen in familial dominant drusen is more widespread compared to the organized symmetrical bilateral nature of the lesion described in this case report.
The morphology of the lesion in this report bears resemblance to the drusen found in best macular dystrophy (BMD) in which there is an initial deposit resembling an egg yolk. BMD is usually of juvenile onset and progresses from the previtelliform stage to a stage of choroidal neovascularization that ultimately leads to severe impairment of vision [17,22]. In comparison with the presentation of this index patient, there was no reduction of vision as the lesion did not involve the macula. Hence, the drusen in this case could be a congenital condition or an unspecified hereditary retinal dystrophy [12]. Genetic analysis (which is not readily available in our climes) will help to further describe the lesion to ascertain the risk of developing age related macular degeneration and prognostication of this condition.
Peripheral retinal drusen are harbingers of AMDs, the presence of which may be idiopathic. In this case report, the symmetrical nature of the lesion in both eyes of this patient is not the typical clinical feature found in AMD and its variants. More so, the age of the patient does not fall within the age group of individuals that would have age-related macular degeneration. These suggest that the lesion may be an innocuous congenital condition. There is need nonetheless to follow up and monitor this patient to see if he would develop age-related macular degeneration.
The authors are grateful to the management of Lily Hospitals Limited for providing a conducive atmosphere for working and learning. Professor Macdonald Idu reviewed this article, we appreciate his masterly guide. We are also thankful to Miss Onavwose Princess for providing technical assistance for this report.