Postoperative inflammatory cell deposition on the surface of intraocular lenses (IOLs) is a well-described entity and considered part of the normal postoperative course. This inflammatory response, which is usually a visually insignificant finding, typically begins shortly after surgery and peaks at 3 months.
We describe a case of a patient who developed late-onset intraocular lens (IOL) cellular precipitates 8 months after IOL placement. The patient had a history of clinically inactive toxoplasmosis and developed visually significant IOL precipitates 8 months after uncomplicated cataract surgery with a previously unremarkable postoperative course without other signs of ocular inflammation. The IOL deposits improved and resolved with use of topical steroids, which coincided with an improvement in visual acuity.
The results of this case suggest that a trial of topical steroid therapy should be considered in patients presenting with late-onset precipitates on the surface of IOLs, even in the absence of concomitant intraocular inflammation.
Postoperative deposition of inflammatory cells on the surface of intraocular lenses (IOLs) is a well-described phenomenon after cataract surgery and are typically of no clinical significance [1,2]. The cellular deposits, or precipitates, are thought to represent a normal component of the eye's foreign body response to the implanted IOL [3]. This inflammatory response consists of small, fibroblast-like cells early in the postoperative period (e.g. 1 month) and a giant cell deposition that begins early in the postoperative period and peaks at 3 months [4]. A study by Werner, et al. noted a variation in the magnitude and timing of the cellular inflammatory response dependent upon the various IOL materials [5,6]. Numerous risk factors have been identified for inflammatory giant-cell deposit (IGCD) formation including older generation silicone IOLs and presence of posterior synechiae.
Although rare, visually significant IOL changes are a concern for both surgeons and their patients as IOL removal or exchange can be a complex or potentially complicated procedure, particularly in patients with a history of uveitis [7]. We present a case of visually significant, localized cellular deposits on the surface of an IOL in patient with clinically inactive toxoplasmosis that presented late in the postoperative period and resolved with topical corticosteroids.
A 41-year-old man presented with a complaint of slowly worsening vision in his right eye (OD) 8 months after cataract surgery. His past ocular history was significant for uncomplicated cataract extraction with IOL placement in the capsular bag OD and bilateral congenital toxoplasmosis chorioretinitis on prophylactic double-strength trimethoprim/sulfamethoxazole 3 times per week. His chorioretinitis had been inactive for at least 9 years when he first established care at our institution, without clinical evidence of intraocular inflammation prior to surgery. Post-operatively his course was unremarkable with a BCVA of 20/30 and no signs of ocular inflammation at 1- and 3-months after surgery, off of topical corticosteroids. On the day of presentation, his ocular examination revealed a decrease in visual acuity to 20/40 OD. His vision remained stable at 20/80 in his left eye (OS). Intraocular pressure was normal in both eyes. Anterior segment exam of the right eye was notable for new multifocal, fibrotic appearing, white deposits on the anterior surface of the IOL that were diffusely distributed (Figure 1). Notably, there was no evidence of active inflammation of the anterior or posterior segment on examination (cell, flare, haze, active chorioretinitis). Anterior segment examination of the left eye was stable, notable for only mild cataract. Posterior segment examination demonstrated stable, inactive, macular involving chorioretinal scars in both eyes.
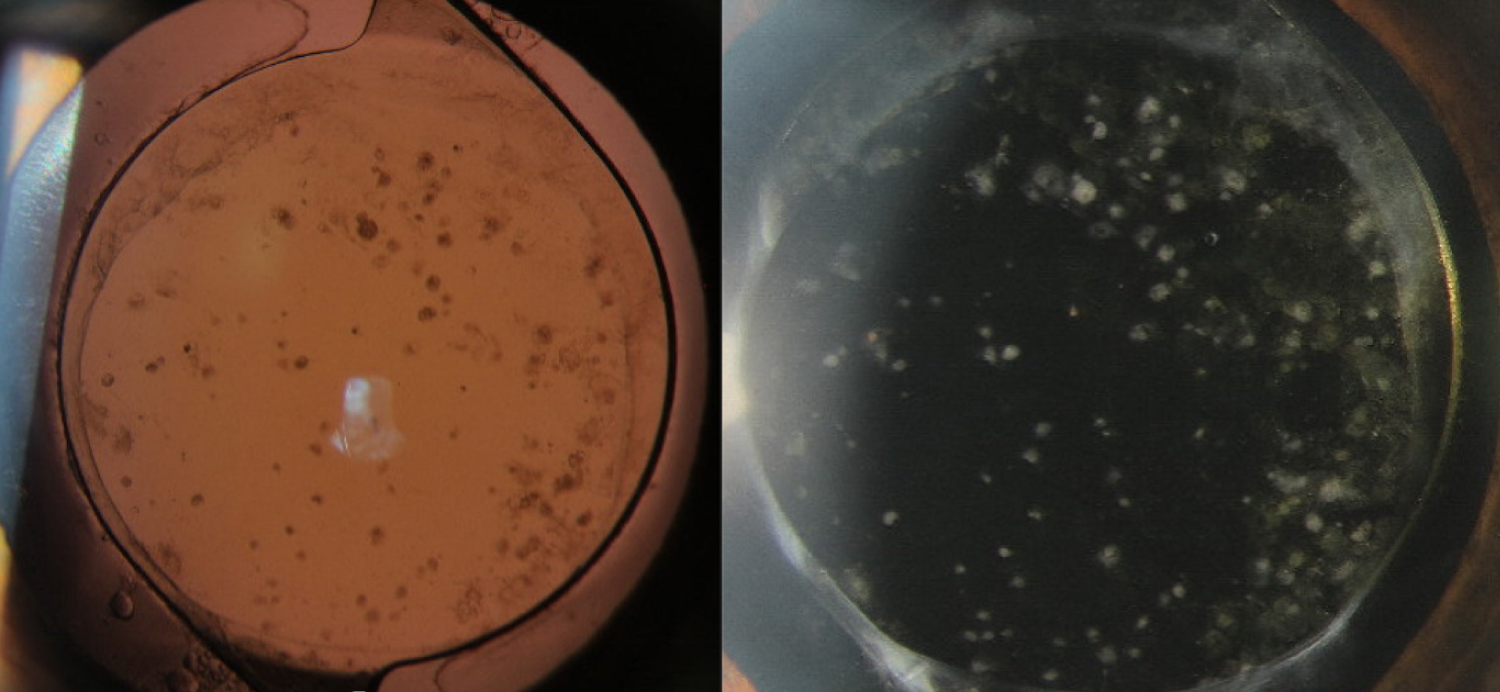 Figure 1: This figure demonstrates the cellular precipitates noted on examination on the anterior surface of the IOL after the patient presented with worsening vision in the affected eye.
View Figure 1
Figure 1: This figure demonstrates the cellular precipitates noted on examination on the anterior surface of the IOL after the patient presented with worsening vision in the affected eye.
View Figure 1
Given the patient's moderate visual impairment in the contralateral eye and symptomatic visual decline in his right eye with new inflammatory-appearing deposits on the lens, we elected to treat the deposits with topical steroids. The patient was started on prednisolone acetate 1.0% four times per day (QID) to the affected right eye and asked to return in 14 days. On follow-up examination, the deposits had markedly improved (Figure 2). The patient was continued on topical steroid drops QID in an effort to fully resolve the deposits. The patient followed up 4 weeks later and endorsed subjective improvement in vision. Examination noted a return of BCVA to 20/30. Moreover, the exam demonstrated continued improvement in the deposits on the surface of IOL (Figure 3). At the follow-up visit 6 months after initially presenting with the inflammatory cell deposits, the patient's IOL remained clear with stable BCVA maintained on a topical steroid once daily.
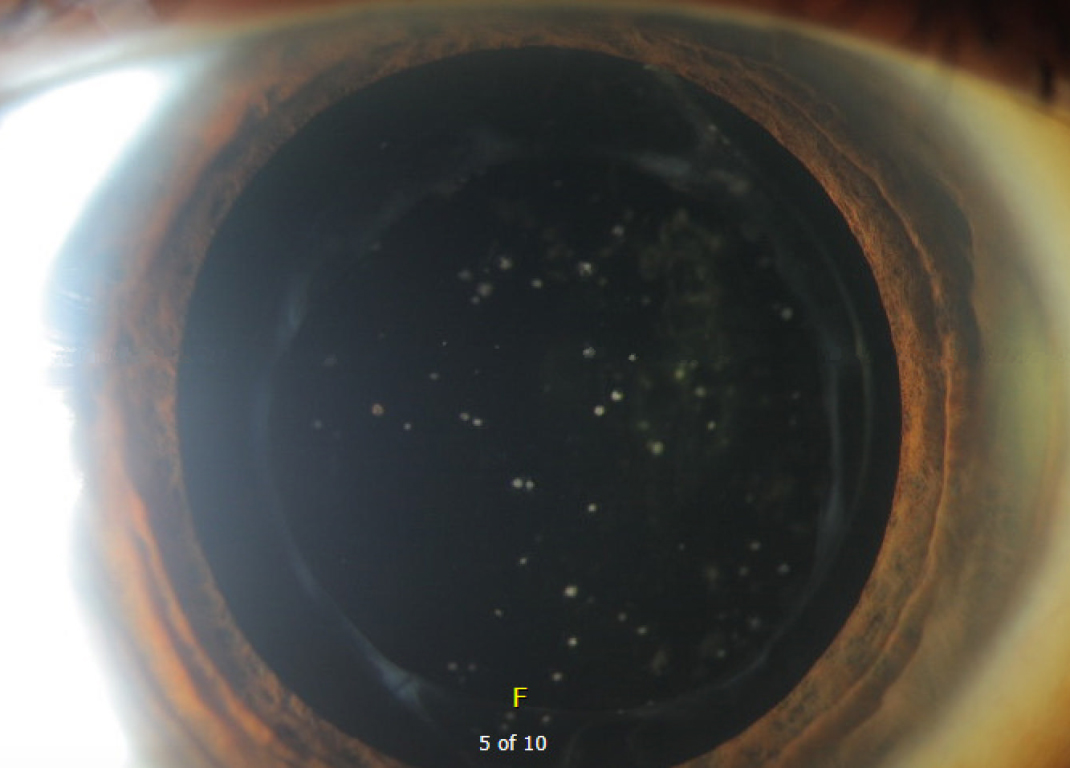 Figure 2: This image demonstrates the appearance of the IOL deposits 14 days after starting topical steroids. Compared to Figure 1, there is a considerable improvement in the density and number of deposits present on the surface of the IOL.
View Figure 2
Figure 2: This image demonstrates the appearance of the IOL deposits 14 days after starting topical steroids. Compared to Figure 1, there is a considerable improvement in the density and number of deposits present on the surface of the IOL.
View Figure 2
 Figure 3: This figure demonstrates the appearance of the IOL 4 weeks after initial presentation. There is a marked improvement in the appearance of the IOL compared to the initial presentation with near complete resolution of the deposits and vision restored to baseline.
View Figure 3
Figure 3: This figure demonstrates the appearance of the IOL 4 weeks after initial presentation. There is a marked improvement in the appearance of the IOL compared to the initial presentation with near complete resolution of the deposits and vision restored to baseline.
View Figure 3
Post-operative IOL cellular deposits are rarely clinically significant and can remain on the lens up to a year after surgery [8]. The foreign-body reaction directed towards the IOL occurs secondary to the breakdown of the blood-aqueous that occurs with cataract surgery and subsequent implantation of the IOL [9]. This blood-aqueous breakdown leads to an influx of inflammatory cells and proteins into the anterior chamber that eventually leads to a cascade of inflammatory activation and eventual cellular response against the IOL [3]. Numerous studies have evaluated the biocompatibility and inflammatory response of numerous IOL biomaterials with some studies suggesting that the magnitude and duration of inflammatory response can be influenced by the IOL material [3,5,6]. While the biomaterials of implanted IOLs have changed and evolved significantly over time and these values may not be indicative of current patterns, a study from the early 1990s described and quantified the cellular response of implanted PMMA lenses at 1, 3, 6 and 12 months postoperatively [8]. This report described the peak in the small, round cell response at 1 month and the giant cell response at 3 months with a steady decline thereafter with eventual resolution, reinforcing that it is a normal component of the postoperative course [8]. Additionally, we must distinguish the cellular deposition that can be visualized on the surface of IOLs as a distinct entity from that of IOL opacification. Although a coalescence of IOL surface deposits can mimic opacification [10], IOL opacification is an uncommon, irreversible complication of cataract surgery with numerous etiologies including discoloration, calcification, silicone oil deposition, or formation of glistenings [11,12]. IOL opacification is associated with numerous contributing factors including but not limited to comorbid ocular and systemic disease, surgical technique, IOL storage and composition [11,12].
The cellular precipitates reported in our case are unique from what has been previously reported in the literature. As previously mentioned, inflammatory IOL deposits typically occur in the early postoperative period and begins to dissipate after 3 months [8]. In the presently described case, the patient underwent uncomplicated cataract surgery and successful implantation of a hydrophilic, acrylic IOL with an uncomplicated postoperative course. He was able to be tapered off a standard regimen of post-operative steroid drops without a recurrence of inflammation and a notably clear IOL was reported at 1 month post-operatively. At 8 months post-operatively, he spontaneously presented with new, visually significant, white deposits on the anterior surface of the IOL. Although the patient had a history of uveitis, his exam was absent of anterior and posterior inflammation. Samuelson, et al. described the development of giant-cell deposits on IOLs after combined cataract and glaucoma surgery in the absence of other signs of intraocular inflammation, but only when adjacent to posterior synechiae, which was not present in our case [13]. This pattern of late-onset inflammatory giant cell deposits after a previously clear IOL is unique from the normal postoperative foreign-body response with a peak in the giant-cell response early in the postoperative period [4,5,8]. Given the absence of associated inflammation and posterior synechiae, we suspect that there are host-specific factors potentially related to the history of congenital toxoplasmosis chorioretinitis that contributed to the cellular deposition on the surface of the IOL. The decision to initiate topical corticosteroids was made based on the patient's history of uveitis and hesitation to pursue surgical intervention given his poor vision in the contralateral eye. The patient had an excellent response to topical corticosteroids with near complete resolution of the deposits, despite lacking other signs of intraocular inflammation. Although our case does not include microscopic examination of the deposits, given the patient's history of uveitis and response to steroid, we suspect that these deposits were inflammatory in nature.
In summary, we present a case of presumed inflammatory IOL deposits that demonstrated excellent improvement and response to topical steroid therapy alone. Our cases suggest that a trial of topical steroid therapy should be considered in patients presenting with late-onset IOL deposits, even in the absence of other signs of intraocular inflammation, particularly if the patient has a history of uveitis.
This study has been performed in accordance with the Declaration of Helsinki. As a single case, no formal human subjects research review was required by the institution.
Written and signed consent was obtained from the patient for permission of publication of this case report.
All authors have read and approved the final manuscript. All authors equally participated in the drafting of the manuscript and review of the literature.
Authors declare they have no competing interests.
This study had no funding.