Because of the worldwide increasing incidence of cancer new alternative therapies are needed. The use of nanotechnology for clinical applications is constantly growing. The objective of this work was to evaluate the effect of the administration of platinum inorganic nanoparticles over livers from Wistar rats. In order to reach this goal cellular Hepatocarcinoma was first induced with N-nitrosodiethylamine (NDEA) and Carbon tetrachloride (CCl4) injections. Subsequently, Pt(acac)2/TiO2 nanoparticles prepared by the sol-gel method, were injected in the portal vein to assess its effects by means of an animal model. After treatment, animals were sacrificed and histophatological studies of the extracted organs (liver mainly) were carried out to determine the morphology of the cells and therefore the presence or absence of cancer. Earliest results confirm the presence of nanoparticles located in the liver but also in small intestine epithelium. We have determined that the effect of a route of systemic administration of the nanoparticles should be tested in later studies.
Nanomedicine, Hepatocarcinoma, Titania nanoparticles, N-nitrosodiethylamine, Carbon tetrachloride, HCC
Cancer is one of the more common sicknesses and one of the main causes of death worldwide. It has caused 7.6 million of the deaths occurred in 2008. More common cancers are breast (1,384.155 cases in 2008), lung (1,608,055 cases in 2008), colon (1,235,108 cases in 2008) and cervix (530,232 cases in 2008). The types of cancer that cause the greater number of annual deaths are lung (1,376,579 deceases), stomach (737,419 deceases) and liver (695,726 deceases) and they are responsible of the 37.3% of the total worldwide deaths.
In 2008, 749,744 new cases of liver cancer were registered, with a death incidence of 92.7% (695,726 deceases). Because of these events, it is one of the cancer types with the smallest survival percentage (7.3%) [1].
One of the main problems of public health in Mexico is cancer; it occupied the third place of the principal death causes in 2008 with 127,604 new cases registered of which 6,178 belong to liver cancer with a death incidence of 99.6% (6,153 deaths in 2008). Liver cancer has a bigger incidence in women with the 52.4% compared to men with a register of 47.6% of the cases [2].
Hepatocellular carcinoma (HCC) is one of the more common malignant tumors in Asia, Africa, and South Europe, United States and other countries, causing one million deaths per year. There are different chronic liver diseases that can develop into hepatocarcinoma, caused by the exposure to toxic chemicals, drugs, alcohol and or hepatitis viruses [1,3], synthetic materials, as well as environmental pollutants [4,5].
On the other side, science advances every day and new ways of prevention and cures are investigated for the different types of cancer, leading a great variety of therapies and drugs. In the last years, the effect of nanoparticles of silicon and titanium oxides has been studied as a treatment to counteract or cure some kinds of cancer [6,7]. This new approach is known as nanomedicine and it has become a new alternative that consists in the synthesis of materials at nanometric level (manometer = 1×10-9) for medical applications [8-11]. This new technology has its origins in the nanotechnology which has had an important growth in the last years.
The objective of the present study was to investigate alterations in male Wistar rats liver by using N-nitrosodiethylamine (NDEA) and carbon tetrachloride (CCl4) in order to examine both the development of hepatocellular carcinoma and the effect of platinum nanoparticles, we mainly focused on the incidence of hepatocarcinoma to further address the important roles of platinum nanoparticles in contributing to the anti-cancer therapies.
The Pt(acac)2/TiO2 nanoparticles were synthesized by the sol-gel method as described by T. Lopez et al. obtaining particles from 1 to 2 nm, which present micropores and mesopores on its surface [12].
Wistar strain male rats with an average weight of 180-220 g were used to develop the HCC model. The rats were placed in aluminum cages, in an air conditioned room with periods of twelve hours light-darkness. They were allowed to freely consumption of food and water. Four groups of fifteen Wistar male rats were formed. Animals from each group were randomly numbered from 1 to 15. The rats from the one designated as control group, did not received any type of drug or reagent, leaving them with free evolution. Animals designated as group 1, were administered with an interperitoneal dosage of 200 mg/kg of NDEA (Sigma-Aldrich) on the first week, one week later, all fifteen animals received subcutaneous weekly injection of CCl4 3mg/kg (REASOL) during six weeks. Animals corresponding to group 2 were administered with three interperitoneal doses of 200 mg/kg of NDEA on weeks 1, 3, and 5, but none CCl4 dosage was administered. Group 3 animals received three interperitoneal injections of 200 mg/kg of NDEA on weeks 1, 3, and 5, the animals also received an interperitoneal daily injection of 3 mg/kg of CCl4 for five days on weeks 2, 4, and 6 intercalating this administration between the weeks of NDEA, administration followed the same methodology as group 1, but in this case, CCl4 was administered by an intraperitoneal route (Table 1) [13-16].
Table 1: Dosing scheme for NDEA and CCl4. View Table 1
Ten weeks after the beginning of the treatment for the generation of HCC, animals were sacrificed and livers were extracted for its later histopathologic processing and analysis.
Ten weeks after the experiment initiation the five rats from the control group that were not sacrificed were randomly chosen and were administrated with an injection of 200 mg of Pt(acac)2/TiO2 nanoparticles suspended in 200 microliters of isotonic sodium chloride solution by means of liver surgery.
All the equipment, instruments, surgery area as well as gauze, and gloves were sterilized before the procedure. During surgery, animals were anesthetized with an interperitoneal injection of a combined dose of 80 mg/kg of ketamine and 10 mg/kg of xylazine. Once anesthetized, the abdominal area of the animals was shaved and disinfected with povidone-iodine. Exploratory laparotomy was realized and Pt(acac)2/TiO2 nanoparticles were infiltered by the portal vein with an intravenous catheter Radiopaque 16G × 51mm Punzocat® catheter (Figure 1).
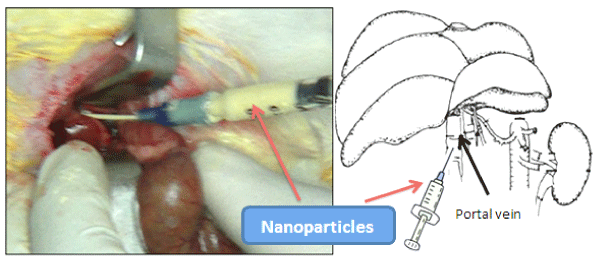 Figure 1: Administration of nanoparticles. View Figure 1
Figure 1: Administration of nanoparticles. View Figure 1
Once the whole material was infiltered, the catheter was removed and bleeding was controlled by applying pressure on the zone, after bleedings were controlled, the incision area was sutured layer by layer. The first layer (muscle) was sutured with anchor technique with 4-0 silk suture. The second layer (skin) with the stitch saturation technique with 4-0 silk suture, and they were supplied with ten units of gentamicin to avoid future infection. The temperature of the animals was monitored and controlled with warm blankets for a two-hour period after the surgery.
After two weeks of initiated the treatment, animals were sacrificed and the organs were extracted.
All the samples were processed and embedded in paraffin. Organs were sectioned with a steel knife mounted in a microtome to obtain 5-micrometer-thick tissue sections which were mounted on a microscope slide individually and then treated with Hematoxylin-Eosin (H&E) stain. This procedure was realized for all samples to prepare them for its later light microscopy analysis.
Significant differences in food and water consumption were observed, among the different groups during the whole study period. Corporal weights of the different study groups are presented in Table 2. A significant weight loss in the three study groups was observed in contrast to the control group.
Table 2: Summary of the mean weight. View Table 2
For group 1 subcutaneous CCl4 and intraperitoneal NDEA doses were suspended because the animals developed skin ulcers over the area where CCl4 was administered, therefore they had to be sacrificed before concluding the induction period. For group 3, NDEA was intraperitoneally administered as for group 1, but CCl4 administration route was also intraperitoneal in contrast to group 1, and any type of skin lesion was developed.
The death rate of group 3 was very high, 73.33% of the animals died before concluding the induction of HCC. The death rate of group 2 was lower, as it is shown in Table 3. One of the probable causes of this differences in the death rate is CCl4 supply, because it might have produced a toxic effect in the liver and kidneys [17,18].
Table 3: Summary of the death rate. View Table 3
Even though the animals from group 1 were sacrificed and several from group 3 died before concluding the induction period, livers were extracted and analyzed in order to compare the morphological changes observed in the three groups.
Histopathologic analysis showed that slides from rats in groups 2 and 3 did not present any evidence of neoplasia but they did presented some signs of inflammation in the hepatocytes and fusiform cells, in contrast with the rats in group 1 in which any evidence of neoplasia or hepatocytes inflammation was found (Figure 2).
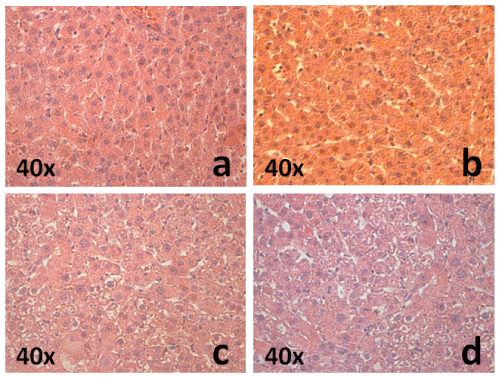 Figure 2: Histologies from the livers extracted from a) control group b) group 1, c) group 2, and d) group 3. View Figure 2
Figure 2: Histologies from the livers extracted from a) control group b) group 1, c) group 2, and d) group 3. View Figure 2
The diagnosis of hepatocellular carcinoma is based on the confirmation of the histopathologic analysis of the extracted livers. The analysis showed a higher incidence in group 3 with an 80%, meanwhile in group 2 the incidence of hepatic neoplasia was 50% and there was any HCC case in group 1 (Figure 3).
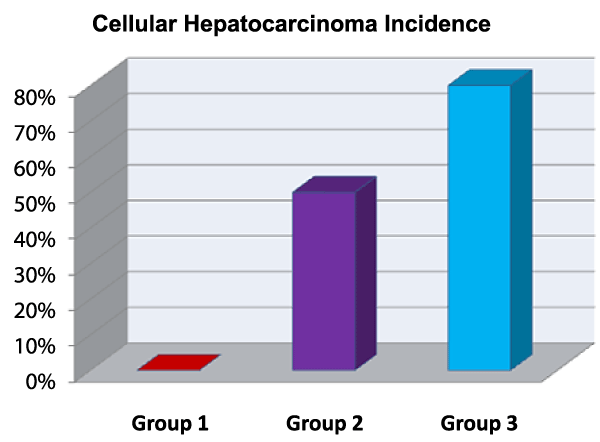 Figure 3: Percentage of the incidence of cellular hepatocarcinoma of group 1, group 2, and group 3. View Figure 3
Figure 3: Percentage of the incidence of cellular hepatocarcinoma of group 1, group 2, and group 3. View Figure 3
Histologies from groups 2 and 3 showed big tumoral polygonal cells with abundant cytoplasm, nuclei are arranged in a trabecular pattern. The tumors do not have any fibrosis capsule, even though the limit between the tumor-like nodule and the empathic parenchyma was normal. Most of the tumors were viable with only small dispersed coagulation islands. Histology slides from the control group were normal (Figure 4).
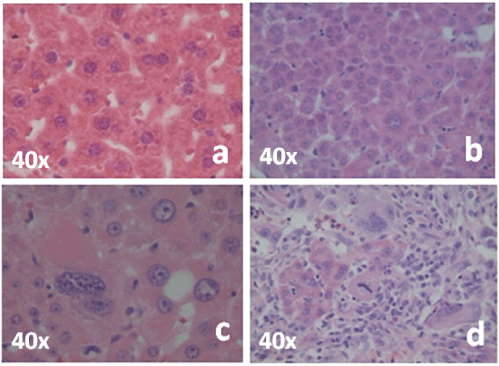 Figure 4: Percentage of the incidence of cellular hepatocarcinoma of group 1, group 2, and group 3. View Figure 4
Figure 4: Percentage of the incidence of cellular hepatocarcinoma of group 1, group 2, and group 3. View Figure 4
The organs from five rats of the control group supplied with Pt(acac)2/TiO2 were analyzed by observation with the naked eye to ensure that the infiltration of the nanoparticles was successful. Yellowish areas in the liver were observed, so that confirmed the presence of nanoparticles in the liver (Figure 5). Due to this observation they were sent to further histopathologic investigation to have a microscopic validation of the nanoparticles localization.
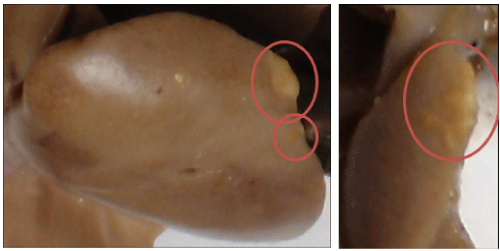 Figure 5: Percentage of the incidence of cellular hepatocarcinoma of group 1, group 2, and group 3. View Figure 5
Figure 5: Percentage of the incidence of cellular hepatocarcinoma of group 1, group 2, and group 3. View Figure 5
Animal models are essential tools for medical research, in this particular case hepatocarcinoma, because they allow us to assess new treatments. The use of Wistar rats gives us a clear idea of how hepatocellular carcinoma is developed. In this work three proposed models were evaluated, and we have determined that model corresponding to group 2 is the most adequate for cancer induction, due to the high incidence of HCC and the low mortality percentage, which allow us to further evaluate the therapeutic effect of nanoparticles. Group 1 was discarded because subcutaneous injection of CCL4 provoked skin lesions and animals had to be sacrificed, animals from group 3 did not present any kind of reaction but the mortality percentage was too high so it was also discarded.
Preliminary results of Pt(acac)2/TiO2 nanoparticles administration showed evidence of the material allocated in the liver, confirming that this rout of administration can be used in later studies to evaluate the anticancer effect using the model previously.