Resident physicians are often required to perform central venous catheter (CVC) or intraosseous catheter (IO) placement when supervision is not readily available. We assessed whether brief CVC and IO simulation-based training increases resident knowledge and comfort performing these procedures unsupervised. Residents were assigned to either a 60-minute CVC training or a control group that received no training; they were also assigned to either a 30-minute IO training or a control group. Both trainings improved resident comfort performing the respective procedures (47% CVC intervention group vs. 26% CVC control group, p = 0.051; 47% IO intervention group vs. 16% IO control group, p = 0.006). There was also a statistically-significant summative effect of receiving both trainings, as 75% of the residents who received both trainings reported comfort obtaining unsupervised emergent vascular access. This suggests that residencies should include not only CVC, but also IO, simulation-based training sessions to prepare residents for emergency situations.
Education, Resident, Procedural skills, Procedures, Simulation-based training, Vascular access
Vascular access is considered a core competency of hospital medicine, yet many residents report discomfort when performing these procedures unsupervised [1,2]. Despite this, senior residents are frequently called upon to obtain access in emergent situations when direct supervision is not available. Because central venous catheter (CVC) placement has a high failure rate and is time-consuming, intraosseous catheter (IO) insertion is an appealing alternative for emergent vascular access [3]. For this reason, the Advanced Cardiac Life Support Guidelines endorse IO placement with a Class IIa recommendation, whereas CVC placement only has a Class IIb recommendation [4].
Although past research has shown a benefit to simulation training for CVC and IO placement, none of these studies have used a true control group to measure the impact of IO training, and no studies have compared CVC and IO training head-to-head [5-8]. The aim of our study was to assess the impact of brief CVC and IO simulation-based training on internal medicine senior residents when compared both head-to-head and against a control group. We hypothesized that both interventions would improve residents' knowledge of the procedures and increase resident comfort with performing unsupervised emergent vascular access.
This was a prospective study of second-year internal medicine residents at a university-based tertiary care hospital. The study occurred during the first week of the academic year for two consecutive years (July 2015 and July 2016). Residents were grouped into blocks of six or seven residents. Each block was randomly assigned to either a 60-minute CVC training or a control group, as well as either a 30-minute IO training or a control group. The control groups received a workshop on leadership that was unrelated to procedural training.
Percutaneous, ultrasound-guided internal jugular CVC placement was taught via the Seldinger technique by critical care attending and fellow physicians. A 10-minute didactic was followed by a 15-minute skill demonstration. The learners then had 35 minutes of supervised deliberate practice with an ultrasound, CVC kit, and manikin. IO simulation-based training was similar in format but conducted over only 30 minutes and led by a chief resident. The curriculum covered humeral and tibial insertion techniques with the aid of artificial bones. Both IO and CVC training were based on previously-published conceptual frameworks [9,10].
After each training session, participants completed a previously-validated survey that asked whether they had received adequate procedural training and if they felt comfortable performing the procedure without supervision (survey available in the Appendix) [11]. The survey was designed to measure satisfaction, self-assessment, and behavior change, and thereby assesses levels one, two, and three, respectively, of the Kirkpatrick model of evaluating training programs [12]. Residents randomized to the control group completed the same questionnaire. During the 2016 training session, participants completed a previously-validated CVC knowledge assessment and an IO assessment that was created by the authors (knowledge assessments available in the Appendix) [13]. The knowledge assessments were reviewed by the UCLA Medical Education Research department for content and response process validation.
Of note, residents randomized to each control group later received both the CVC and IO simulation trainings (i.e., during the same training day). This was done to maintain equipoise given the established benefits of simulation training on trainee procedural competency. Because both of these trainings occurred after the control group residents had completed their surveys, however, the trainings didn't interfere with the study question.
All statistical analyses were performed with GraphPad software. Statistical significance was set at p < 0.05 for all calculations. Data was first analyzed for each simulation training using a two-group comparison between the control group and the intervention group. The Student t test was used for analysis of continuous variables and the Fisher's exact test (two-tailed) was used for categorical data. Subgroup analysis based upon the number of procedures previously performed was also calculated to assess for differences in response related to previous experience.
A 2 × 2 factorial design was then used to assess the summative effects of both trainings. Analysis via a 2 × 2 Fisher's exact test was used for Table 1; a 2 × 4 Fisher's exact test was used in Figure 1.
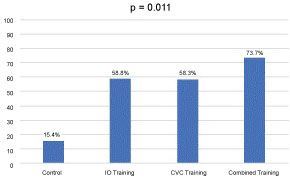 Figure 1: Percent of residents ready to perform unsupervised vascular access*.
Figure 1: Percent of residents ready to perform unsupervised vascular access*.
*Based on participants' self-assessments (from survey responses).
View Figure 1
Table 1: Baseline characteristics of participants (n = 89). View Table 1
Eighty-nine residents participated in the study, as is outlined in the flowchart in Figure 2. Baseline characteristics of the study participants are outlined in Table 1. The CVC control group had performed more IOs than the CVC intervention group (p = 0.0443), and the IO training group had performed more CVCs than the IO control group (p < 0.0001), but otherwise baseline demographics were similar between groups. Whereas 88% of residents had placed at least one CVC, only 7% had placed an IO prior to the study. The average number of CVCs placed was also significantly higher than the average number of IOs placed per resident (2.95 vs. 0.07, p < 0.0001).
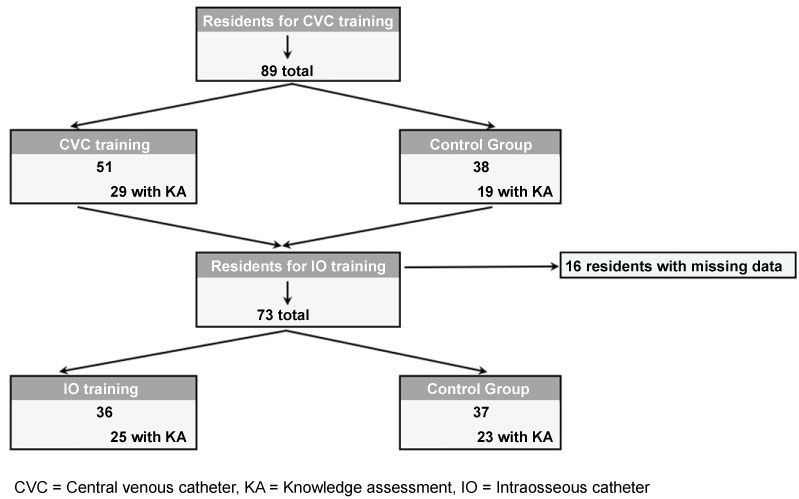 Figure 2: Flowchart for patient inclusion.
View Figure 2
Figure 2: Flowchart for patient inclusion.
View Figure 2
A two-group comparison between residents who received IO training and those who did not (Table 1) demonstrated that the training improved the proportion of residents who felt ready to perform IO placement without supervision (47% vs. 16%, p = 0.006) as well as the proportion who felt that they had received adequate training (69% vs. 11%, p < 0.0001). The CVC training similarly improved resident comfort placing CVCs unsupervised when compared to the control group (47% vs. 26%, p = 0.051) and satisfaction with training (71% vs. 42%, p = 0.009). Knowledge assessment of the 25 residents who received IO training showed that the training led to a higher mean score compared to the 23 who did not receive the training (77% vs. 40%, p < 0.0001). CVC training also led to an increase in the scores of the 29 who received the training compared to the 19 who did not, though this was not statistically significant (46% vs. 38%, p = 0.066).
Subgroup analysis of the residents who had placed fewer than 4 CVCs revealed that CVC training did not lead to a statistically significant increase in comfort performing CVCs unsupervised (31% in training group vs. 20% in the no training group, p = 0.53). The percentage of residents in this subgroup who felt that they had received adequate training also did not increase significantly with the simulation training (48% vs. 32%, p = 0.27). In contrast, even among residents who had never previously placed an IO, simulation training increased comfort and satisfaction when compared to those who did not receive the training (comfort 42% vs. 21%, p = 0.03; satisfaction 70% vs. 9%, p < 0.0001).
In the analysis of the 2 × 2 factorial design, 13 residents received no training, 17 received only IO training, 24 received only CVC training, and 19 received both IO and CVC training. Sixteen residents had to be excluded from the analysis because of missing data. Only 15% of residents in the control group felt comfortable performing emergent vascular access compared to 58% in the IO training group and 59% in the CVC group, while 75% of residents in the group that received both IO and CVC training reported comfort (p = 0.011) (Figure 1). Simulation training also increased the number of residents who felt that they had received adequate training in obtaining emergent vascular access (38% in control group, 75% in CVC group, 82% in IO group, and 84% in combined, p = 0.03).
In hemodynamically-unstable patients, the placement of vascular access devices to administer volume expanders, vasopressors, or antiarrhythmic medications can be lifesaving. At our institution, senior medicine residents are often the first responders to these emergencies. As with prior publications, this study confirmed our hypothesis that simulation-based training improved residents' confidence in IO and CVC placement [5-8]. In addition, since the most common reason for not using IO catheters is a lack of knowledge, the superior knowledge assessment scores in the IO group supports the benefit of IO simulation-based training [14].
Our study also suggests the novel implication that a 30-minute IO training session is as effective as a 60-minute CVC session in achieving this goal. Indeed, the simplicity of IO catheter placement results in faster and more successful emergent vascular access than CVC placement [3]. This may account for the effectiveness of such brief IO training even in residents with little past exposure to IO catheters. This contrasts with the subgroup of residents with minimal CVC exposure who did not derive benefit from the brief simulation-based CVC training. It is possible that novice CVC learners need longer than one hour, instead requiring a more comprehensive and prolonged curriculum, as has been supported by prior publications as well [5].
This study supports the summative effect of combining both IO and CVC training together to increase the number of residents ready for unsupervised emergent vascular access, as the combined training group had a higher percentage of comfortable residents than either of the individual training groups. Given that many residency programs incorporate formal CVC training but may not formally teach IO placement, our results suggest that programs should consider adding IO simulation-based training to their formal curricula. Importantly, IO training does not negate the need for CVC training: IOs cannot be used for the infusion of multiple medications, hemodialysis, or the placement of pulmonary catheters or temporary pacemaker wires. In addition, their longevity is limited to 24 hours and their utility is limited in the ambulatory patient. Programs with neither CVC nor IO simulation-based training, however, could consider starting with IO training, as this is simpler, faster, and appears just as effective as CVC training in achieving emergent vascular access.
There are several limitations to this study. First, this was performed at a single institution, so the results may not be generalizable across all institutions. Second, the survey and knowledge assessment were conducted on the same day as training, so future studies should ensure that the knowledge and confidence gains are retained over time. Finally, though there was objective improvement in knowledge of the procedures, our study also relied upon the learners' self-assessment of competency. Residents' self-reported confidence has been shown to be a valid predictor of their skill and complications during CVC placement, but further research is needed to prospectively compare objective skill and patient outcomes [15]. Indeed, to further assess level four of the Kirkpatrick model (which evaluates whether a training had a tangible impact on results), future studies should evaluate whether there is an improvement in the number of CVCs and IOs successfully inserted on patients after a simulation training such as ours [12].
In summary, IO and CVC simulation-based training improves residents' knowledge of and readiness to perform vascular access procedures independently. Both training sessions individually increase resident comfort obtaining emergent vascular access without supervision and combining the two further augments this effect.
None.
Conception and Design: KS, JF, ML, JW; Analysis and Interpretation: KS, JW; Drafting the manuscript for intellectual content: KS, JW; Final approval of the version to be published: KS, JF, ML, JW.