Takotsubo cardiomyopathy is commonly associated with a sudden psychological, emotional or physical stress such as a death of a family member. Here we present a case of Takotsubo cardiomyopathy occurring after revascularization of an acute coronary syndrome providing evidence that these two discrete, separate, but inherently inter-related events can coexist.
Takotsubo cardiomyopathy, Acute coronary syndrome, Stress cardiomyopathy
Takotsubo cardiomyopathy or stress cardiomyopathy was first described in 1990 by Sato, et al. and is characterized as a transient cardiac syndrome with reversible left ventricular systolic dysfunction precipitated by a sudden psychological, emotional or physical stress [1]. Examples of such stressors are the death of a family member, a heated argument or winning a lottery. Takotsubo cardiomyopathy following medical procedures, especially after coronary angiography for an acute coronary syndrome has been rarely described in the literature [2-4]. We present a case of Takotsubo cardiomyopathy following percutaneous coronary intervention for an inferior ST segment elevation myocardial infarction (STEMI) occurring during a dobutamine myocardial perfusion scan, providing additional evidence that the somatic stress of an acute coronary syndrome can also precipitate Takotsubo cardiomyopathy.
An 89-year-old female with multiple cardiac risk factors was experiencing atypical chest pain and was referred for a diagnostic elective dobutamine myocardial perfusion scan. Past history included asthma, and the test was performed off her usual medications. Baseline electrocardiogram (ECG) showed sinus rhythm with a partial right bundle branch block pattern (Figure 1A). Rest images of myocardial perfusion were performed after the rest injection of 214 MBq of Tc-99m sestamibi (Figure 2). This showed normal perfusion and normal LV function with LVEF > 80% at rest. No regional wall motion abnormality was seen. Twelve minutes into a standard dobutamine infusion, at 40 ug/kg/min, the ECG evolved into ventricular bigeminy with inferior and lateral ST elevation (Figure 1B). She described no chest pain, but looked "unwell". The dobutamine infusion was immediately ceased; the stress dose of Tc-99m sestamibi was not injected. The patient was taken for urgent coronary angiography, developing chest pain in transit that revealed a subtotal occlusion of the right coronary artery with minor disease in the left coronary arteries (Figure 3A). A single drug eluting stent was inserted into the right coronary artery with no complications (Figure 3B). Following percutaneous coronary intervention, she was transferred to the recovery bay feeling well with normalisation of her ECG. Thirty minutes after the end of the procedure upon learning she had suffered a myocardial infarction the patient developed severe central chest pain and dyspnoea. ECG now showed sinus rhythm with anterolateral ST segment elevation (Figure 1C). A repeat urgent coronary angiogram was performed which showed unchanged minor left coronary artery disease and a patent right coronary artery stent (Figure 3C). Left ventriculography demonstrated the classical apical ballooning of Takotsubo cardiomyopathy (Figure 3D). This was confirmed on subsequent transthoracic echocardiography the next day, which showed apical akinesis with hyperkinetic basal segments (Figure 4). High sensitivity troponin after her second angiogram was elevated at 399 ng/L (normal < 15 ng/L). She was managed with dual antiplatelet therapy for stent protection, a beta blocker and ace-inhibitor for her left ventricular dysfunction and was discharged 22 days later after inpatient rehabilitation. At 6 week follow-up, she was asymptomatic with normalization of left ventricular function on echocardiography.
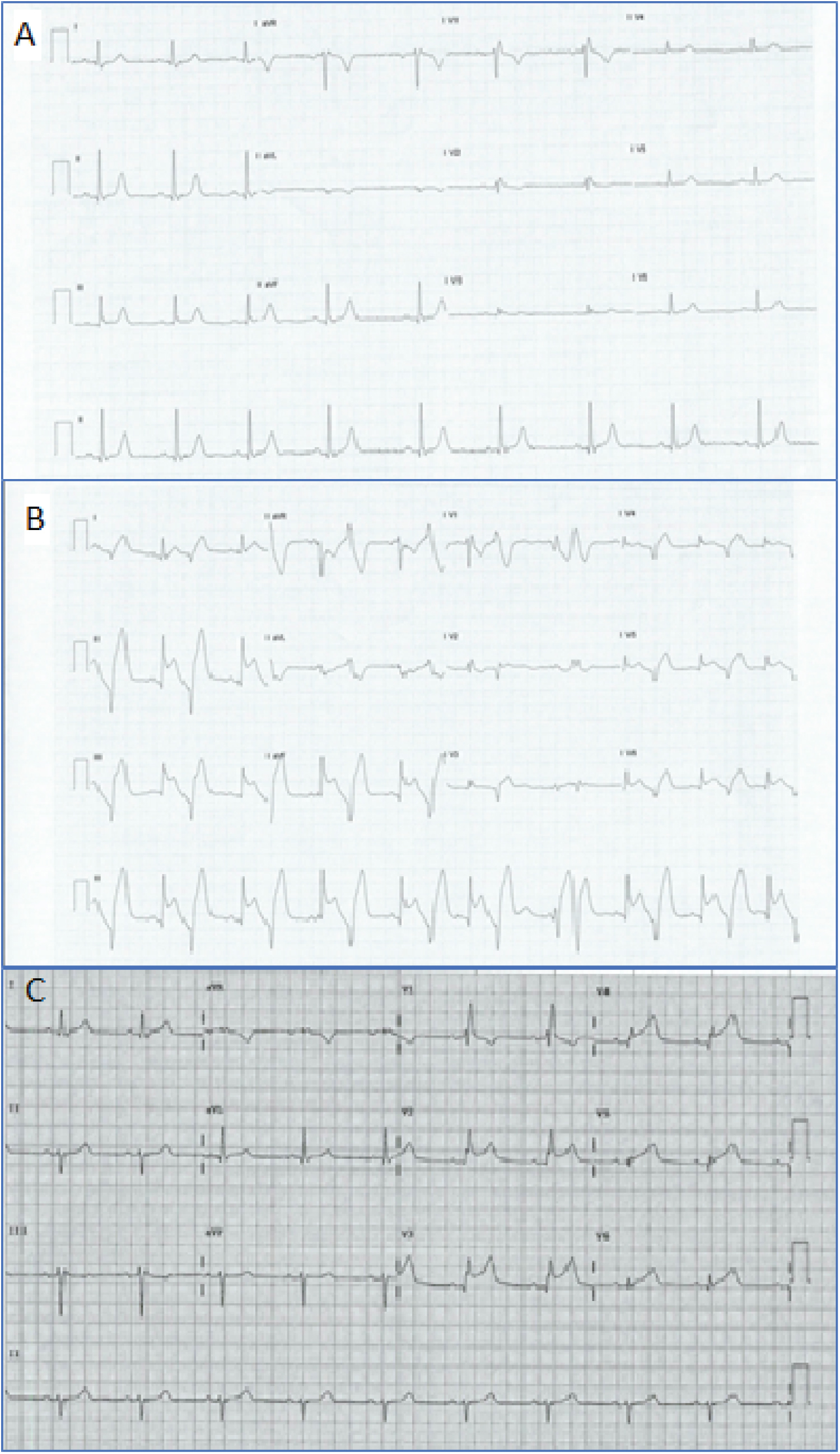 Figure 1: Electrocardiograms.
Figure 1: Electrocardiograms.
Panel A: Rest ECG prior to commencement showing a partial RBBB pattern; Panel B: ECG during dobutamine infusion showing frequent ventricular ectopy and the pattern of an inferior STEMI; Panel C: Repeat ECG, with chest pain 30 minutes post PCI to RCA suggesting an anterior STEMI.
View Figure 1
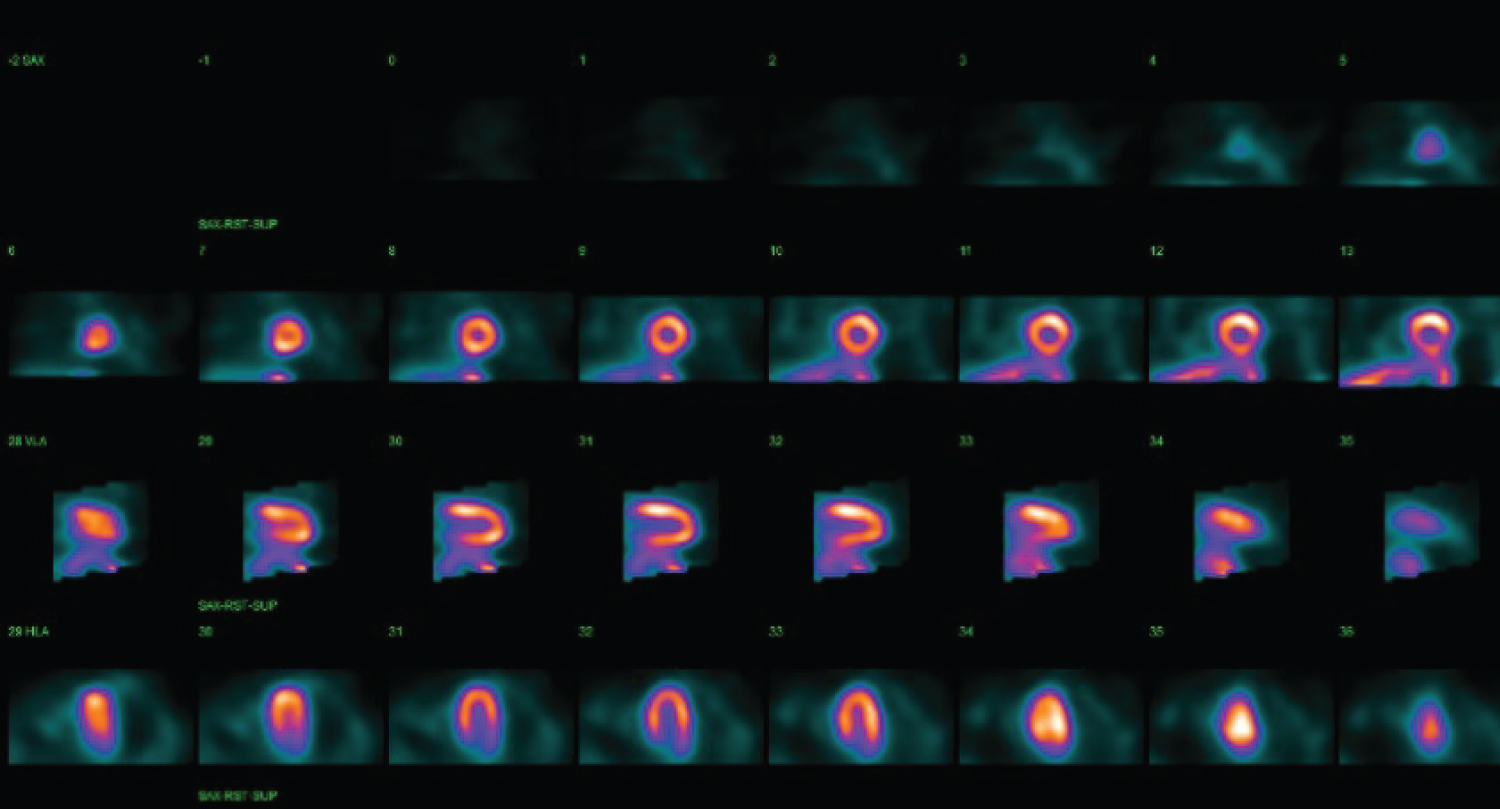 Figure 2: Rest Tc-99m sestamibi study is seen to be normal. SPECT images are shown with short axis (top row), vertical long axis (middle row) and horizontal long axis (bottom row).
View Figure 2
Figure 2: Rest Tc-99m sestamibi study is seen to be normal. SPECT images are shown with short axis (top row), vertical long axis (middle row) and horizontal long axis (bottom row).
View Figure 2
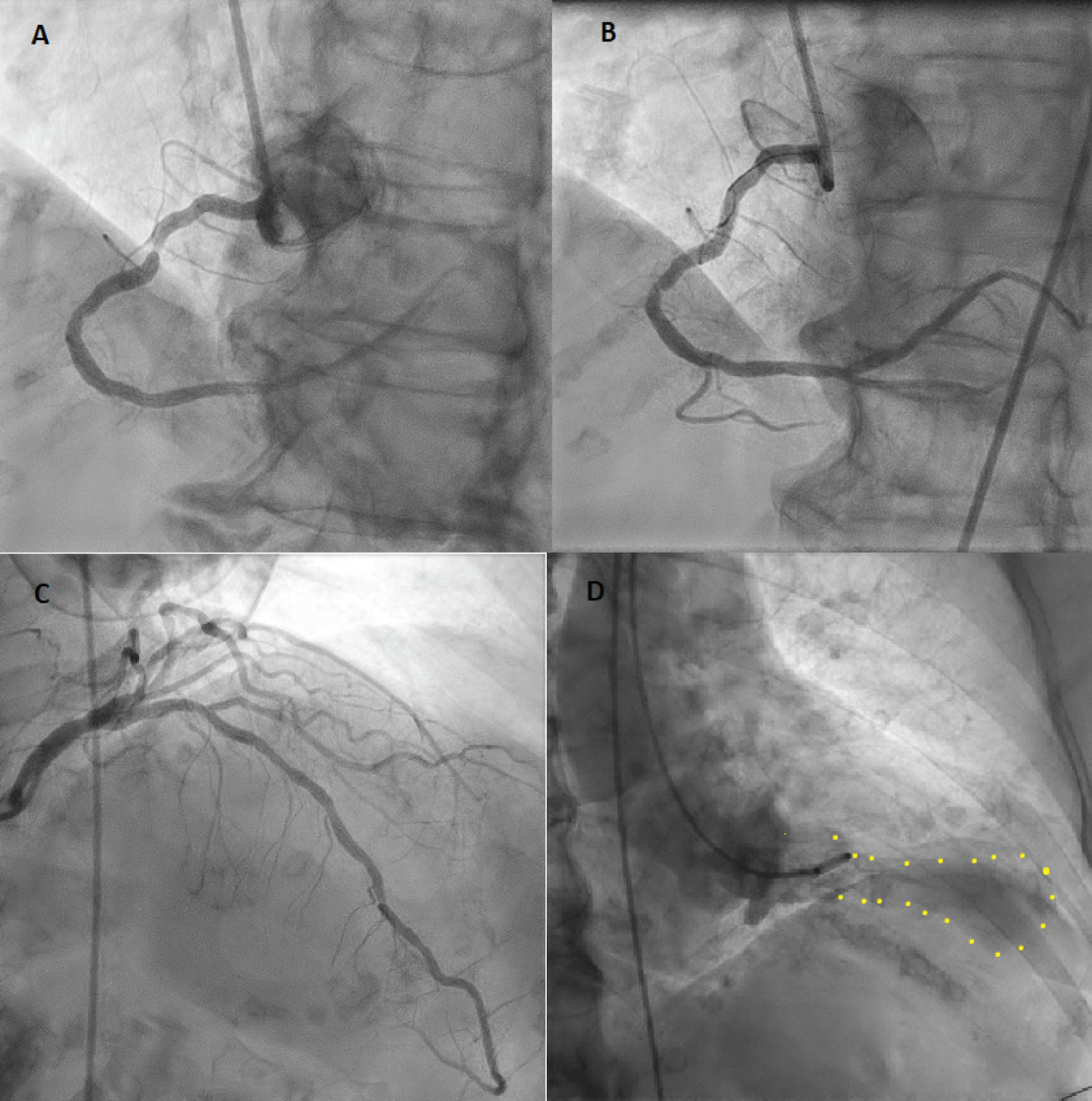 Figure 3: Coronary angiography images.
Figure 3: Coronary angiography images.
Panel A: First angiogram demonstrating high grade right coronary artery stenosis; Panel B: Post insertion of a drug-eluting stent to right coronary artery; Panel C: Second angiogram demonstrating minor left coronary disease, unchanged from the first angiogram. No significant stenosis is seen, suggesting physiology of a Takotsubo syndrome; Panel D: Left ventriculogram in end systole showing the classical pattern of Takotsubo cardiomyopathy with apical ballooning.
View Figure 3
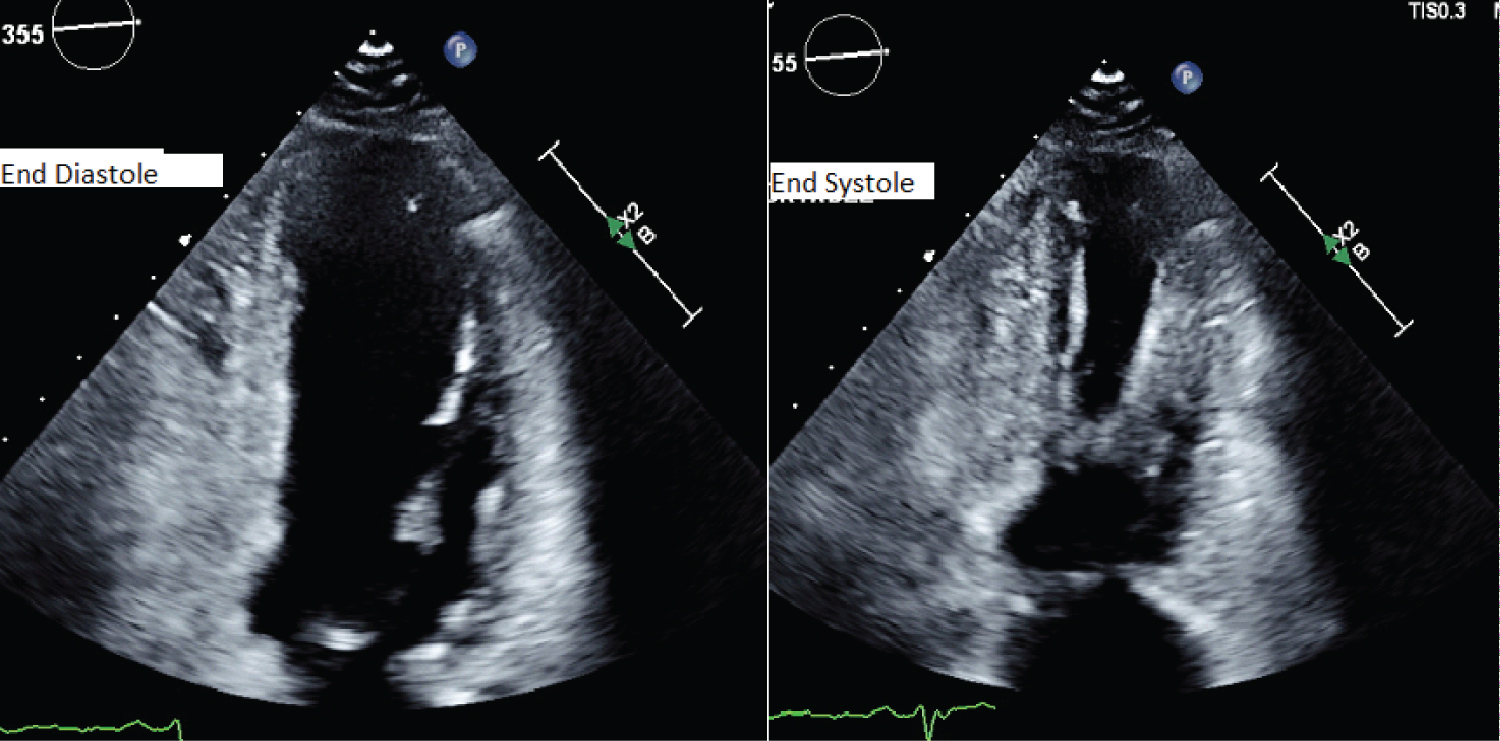 Figure 4: Transthoracic echocardiogram images.
Figure 4: Transthoracic echocardiogram images.
End diastolic and end systolic images one day post event showing a classical pattern of Takotsubo cardiomyopathy with akinetic apical segments and hypercontractile basal segments.
View Figure 4
Takotsubo cardiomyopathy in most cases has an identifiable stressor which leads to a hypersympathetic response with a sudden surge of catecholamines such as noradrenaline and adrenaline [4]. At supraphysiologic concentrations these catecholamines result in a negative ionotropic effect, with a predominance in the apical myocardial regions that contain the highest concentration of β2-adrenoceptors, giving rise to the classical wall motion abnormalities seen in this condition [5]. Takotsubo cardiomyopathy can mimic clinical features of an acute coronary syndrome with chest pain, dyspnoea and ST elevation as seen in our case. A confirmatory diagnosis requires exclusion of coronary artery disease, particularly the left anterior descending artery. Takotsubo cardiomyopathy has rarely been described in association with coronary syndromes [2,3,6]. In all of these cases, left ventriculography demonstrated the segmental wall motion abnormality of Takotsubo cardiomyopathy that could not have been caused by the non-LAD occluded vessel causing the STEMI. In our case, the development of Takotsubo occurred within 30 minutes of revascularization of an acute STEMI from one vessel and presented with an infarct pattern of potential STEMI in another vessel that showed no significant stenosis both immediately before and immediately after the chest pain event. This case adds to the evidence that Takotsubo cardiomyopathy and acute coronary syndromes can co-exist with two discrete, separate, but inherently inter-related events occurring within a very short period of time. It may not be over until it is over!.
The authors have no conflicts of interest to disclose.
All authors contributed equally for the preparation of the manuscript.
No funding was received for the preparation of the manuscript.