A coronary arterial fistula (CAF) is a connection between a coronary artery and a cardiac chamber or another blood vessel. Coronary artery fistulae are rare, with a reported incidence of 0.2%, and primarily congenital [1]. However, they have also been reported after cardiac surgery [2,3].
It has been reported that the incidence of CAF in post-surgical individuals seems to rise with the increasing complexity of the surgery. Heart transplant sees the highest incidence at 20.4%, followed by coronary artery bypass (CABG) with valve surgery, then CABG alone [4]. While the exact mechanism is unknown, a possible cause could be endothelial and vascular growth factor activation from injury.
The follow up of heart transplant patients is also implicated in the formation of CAF. Most of the post-transplant CAF are cameral-coronary fistulae, and the right ventricle is the most commonly-involved chamber. Heart transplant patients get regular cardiac biopsies to look for rejection, especially in the period just after transplant. It is believed that repeated right ventricular biopsy is associated with the formation of CAF. The incidence of cameral-CAF is noted to have declined over the years in cardiac transplant patients with the development of better endomyocardial biopsy techniques, such as using high-resolution fluoroscopy, dedicated labs staffed by cardiologists for biopsy, and obtaining specimens from the interventricular septum [5,6].
However, in a pediatric group of cardiac transplant patients, non-cameral CAF was more common and reported to be predominantly into the ipsilateral recipient pulmonary vasculature. This incidence seemed to correlate with longer donor ischaemic time [7].
Most post-transplant CAF are asymptomatic. However, CAF are known to be complicated by a myriad of conditions, such as myocardial ischaemia, myocardial infarction, congestive cardiac failure, or sudden death [8].
We describe a case of coronary artery-pulmonary artery fistula in a patient who had an orthotopic heart transplant.
A 40-year-old Indian gentleman has a history of dilated cardiomyopathy secondary to viral myocarditis. He was treated with heart failure medications, received an automated implantable cardioverter-defibrillator in 2005, and left ventricular assist device (LVAD) in 2009. He subsequently underwent an orthotopic heart transplant in 2011 at the age of 34. The donor was a 38-year old male patient with a brain tumour, which was complicated by seizures that resulted in hypoxic-ischaemic encephalopathy. The transplanted heart was noted to have a LV ejection fraction of 50% with anterior wall hypokinesia and normal coronary arteries. The immediate post-operative period was complicated by arrhythmias (atrial fibrillation, atrial flutter, and supraventricular tachycardia), ischaemic hepatitis, acute kidney injury, sepsis from an unknown source, and metabolic encephalopathy, all of which gradually resolved. The patient was discharged well.
He was followed up closely with endomyocardial biopsies at 1, 3, 4, 6, 8, 10, 16, 21, 26, 31, and 41 weeks post-transplant, and then at 1, 2, 3, and 5 years post- transplant. These showed either Grade 1R rejection or no rejection. Myocardial perfusion imaging done at 2, 3 and 4 years post-transplant was negative for myocardial ischaemia. He also underwent coronary angiograms at 1, 3 and 5 years post-transplant, and they showed normal coronary arteries and no fistulae were noted. Serial echocardiograms all showed the preservation of a normal ejection fraction and normal pulmonary artery pressures. A dobutamine-stress echocardiogram performed 5 years after transplantation was negative for myocardial ischaemia.
A dobutamine-stress echocardiogram done 6 years post-transplant, however, showed inducible wall motion abnormality suggestive of myocardial ischaemia in the anterior septal wall. Left and right ventricular size and function were otherwise normal. The left atrium was moderately dilated and the right atrium mildly dilated. Pulmonary artery systolic pressure was estimated to be normal.
A CT coronary angiogram (Figure 1 and Figure 2) showed no significant luminal disease in all three coronary arteries; however, the presence of small vessels originating from the left anterior descending (LAD) artery which extended to the pulmonary trunk, suggestive of small fistulae extending from the coronary artery to the pulmonary artery.
 Figure 1: The coronary artery-pulmonary artery fistulae (arrowed) seen on CT coronary angiogram.
View Figure 1
Figure 1: The coronary artery-pulmonary artery fistulae (arrowed) seen on CT coronary angiogram.
View Figure 1
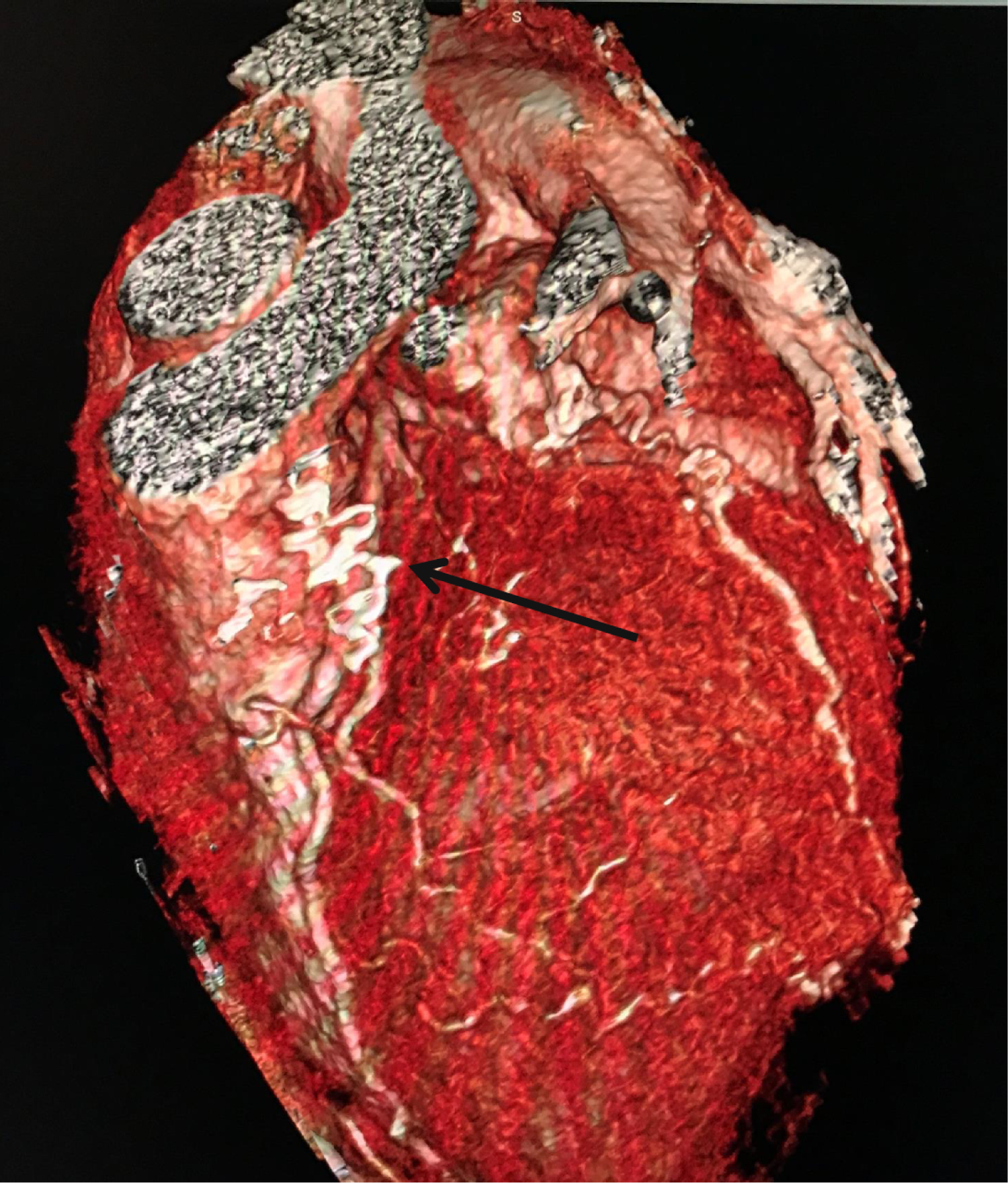 Figure 2: A 3D-reconstruction of the CT coronary angiogram showing the coronary artery-pulmonary artery fistulae (arrowed).
View Figure 2
Figure 2: A 3D-reconstruction of the CT coronary angiogram showing the coronary artery-pulmonary artery fistulae (arrowed).
View Figure 2
The patient was otherwise asymptomatic and there were no murmurs heard on auscultation. The patient opted not for invasive coronary evaluation at this stage due to the lack of symptoms and the CAF being small on the CT coronary angiogram. He remains on regular follow-up and surveillance.
CAF are primarily congenital and rare. However, an unusually high incidence of CAF has been reported in heart transplant patients.
These are primarily cameral-CAF (to the right ventricle), possibly as a complication of repeat routine endomyocardial biopsies. Potential causes could involve septal branches into the right ventricle which are 'unroofed' during the performance of right ventricular biopsies, and perhaps also further induced by cytokines (such as endothelial and vascular growth factor) and inflammation from the extensive surgery of cardiac transplantation. It is also possible that in the donor heart these CAF were congenital, or that the donor heart had sustained an injury from an accident prior to harvest and this could further explain non-cameral CAF [9].
Our patient has had a relatively smooth post-transplantation course with minimal rejection, normal coronary arteries, no symptoms, and no related admissions. While he was asymptomatic, his dobutamine-stress echocardiogram was found to be abnormal in the LAD territory, corresponding to the area that might have been affected by the coronary artery-pulmonary artery fistulae [10]. A 'steal' syndrome with substantial blood flowing into the fistula could potentially cause myocardial ischaemia. However, the vessels in question, were noted to be small, and the sizes of the coronary artery and pulmonary artery were normal.
Other known complications of coronary artery fistulae include infective endocarditis and embolic phenomena.
There is limited long-term data on coronary artery fistulae after cardiac transplantation, but outcomes are generally good with conservative management for asymptomatic patients [11]. There are even fewer case reports on fistulae that subsequently underwent intervention. Zoghi, et al. reported an asymptomatic case of coronary-pulmonary arterial fistula that had regional wall motion abnormalities on echocardiography with ischaemia diagnosed on a myocardial perfusion study. The abnormalities resolved with coil embolization of the fistula [12].
As our patient was asymptomatic, the decision was made to continue surveillance for further potential complications.
Acquired CAF are rare, but have been noted to occur more frequently after cardiac transplant. These patients need to be monitored for potential complications, and treated when necessary. The underlying mechanism of occurrence and incidence should be further explored.