A 65-year-old caucasian man with a history of aortic valve replacement with a St. Jude bi-leaflet mechanical prosthesis in 1984, mitral valve replacement in 2004 with Edwards Starr Ball and Cage prosthesis, atrial fibrillation and ulcerative colitis presented to our emergency department with central chest pain radiating to his jaw with associated diaphoresis and nausea. Symptom onset was an hour pre-admission.
A previous angiogram demonstrated non-occlusive coronary arteries 12 years prior. Additional medical history was notable for ulcerative colitis with distant history of colectomy and stoma formation in 2015, rheumatic heart disease, dyslipidemia and splenectomy secondary to trauma. Of interest he had recently been assessed by hematology with a working diagnosis of hypereosinophilic syndrome. He was known to cardiology and underwent annual echocardiogram. Medications included warfarin, bisoprolol, digoxin, atorvastatin, penicillin V. He had no known drug allergies. As regards time in therapeutic range with warfarin, his international normalized ratio (INR) had been fluctuant.
On arrival, his vital signs included a heart rate of 97, blood pressure of 111/32, which dropped further to 86/56 within 30 minutes, respiratory rate of 22, oxygen saturation of 94% on room air and a temperature of 36.2 degrees. He was in significant discomfort clutching his chest. Physical exam revealed an irregular, tachycardia rhythm with a metallic S1 and S2, with flow murmur audible for both. There was no gallop rhythm and he had a slightly displaced apex beat. There were mild bi-basal fine crepitations consistent with pulmonary oedema. Jugular veins appeared flat. His abdomen was soft and non-tender. Peripherally he had mild oedema.
ECG showed left bundle branch block with significant elevation in leads aVR, V1 with reciprocal depression in infero-lateral leads (Figure 1). An old ECG from previous admission demonstrated non-specific intraventricular conduction delay. There were some minor dynamic changes with 1 mm aVR lead ST elevation with reciprocal changes in other leads. A right sided ECG did not demonstrate electrical evidence of right ventricular infarction. An urgent portable chest X-ray demonstrated increased pulmonary markings, moderate right-sided effusion, and two mechanical valves. On the basis of the patient's clinical presentation and concern over dynamic ST elevation with reciprocal change the decision was made to urgently perform cardiac catheterization. Laboratory studies returned while the patient was on the table, international normalized ratio returned at 1.8, hemoglobin was 9.4 g/dl, white cell count was 15.2 by 10^9/L, platelet count was mildly elevated at 491 by 10^9/L and his estimated glomerular filtration rate was calculated as 82 ml/min/1.73 m2 (similar to previous 86). There was eosinophilia of 3 by 10^9 cells (0.04-0.4) compromising 24% white cells and had persistent for the preceding 2 years. High sensitivity Troponin-T level returned at 112 ng/L (< 14), (Figure 1).
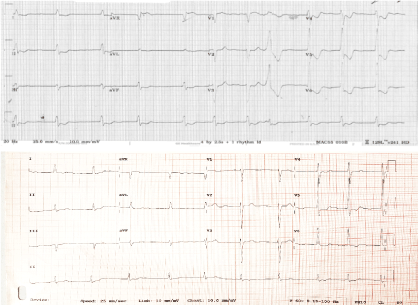 Figure 1: Top: ECG 1; on presentation. Bottom: ECG 2 from a year previously. View Figure 1
Figure 1: Top: ECG 1; on presentation. Bottom: ECG 2 from a year previously. View Figure 1
The patient was loaded with dual antiplatelets. The patient was in cardiogenic shock at this stage and dobutamine infusion at 7.5 ug/kg/min and noradrenaline was commenced at 7. Coronary angiogram demonstrated 50% occlusion in the mid left circumflex artery, with TIMI 3 flow distally. His remaining coronary vasculature was unobstructed (Figure 2). Fluoroscopy demonstrated a bi-leaflet tilting disk with absent/severely restricted motion of the left valvular prosthetic leaflet (Figure 3). An aortogram demonstrated severe aortic regurgitation. A high suspicion of valve thrombosis led to a decision for thrombolysis and transfer to the regional cardiothoracic centre for valve replacement. Alteplase 10 mg Slow IV followed by 90 mg over 90 minutes with NaCl, was given, and this was followed by unfractionated heparin 2000 IU loading and 1000 U/hour to achieve an APTTR 2.5. Bedside echo in ICU demonstrated severe Aortic regurgitation, moderate aortic stenosis, and good systolic function (Figure 2 and Figure 3).
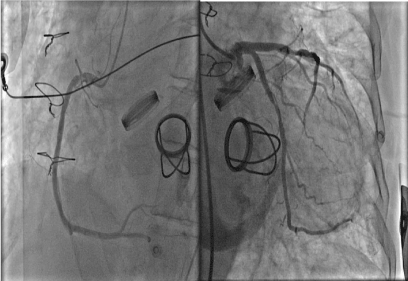 Figure 2: Coronary Angiogram and Aortogram (Left; Right coronary artery, Right: Left coronary artery. There is about 50% stenosis of the cirumflex artery which had distal TIMI 3 flow). View Figure 2
Figure 2: Coronary Angiogram and Aortogram (Left; Right coronary artery, Right: Left coronary artery. There is about 50% stenosis of the cirumflex artery which had distal TIMI 3 flow). View Figure 2
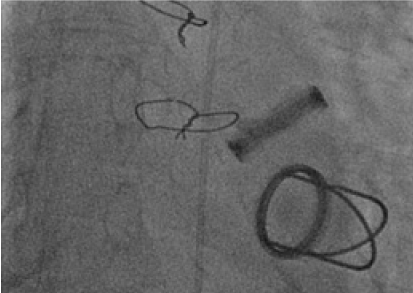 Figure 3: Systolic frame on fluoroscopy. Restricted motion of both tilting leaflets was observed. View Figure 3
Figure 3: Systolic frame on fluoroscopy. Restricted motion of both tilting leaflets was observed. View Figure 3
Transfer to the cardiothoracic centre was arranged overnight. (Figure 4) is an image of the valve itself. Histology of the valve demonstrated smeared basophilic nuclear debris but with only scattered viable inflammatory cells comprising scant neutrophils. Following the procedure this gentleman was weaned from supports. He had a CT brain, which did no demonstrate any embolic event. Following return back onto anticoagulation post-surgery aspirin was commenced. He received a steroid course for a flare up of his inflammatory bowel disease. He was also seen by hematology services who diagnosed myeloproliferative disorder, and hydroxyurea was commenced. He returned home 3 weeks after his surgery (Figure 4).
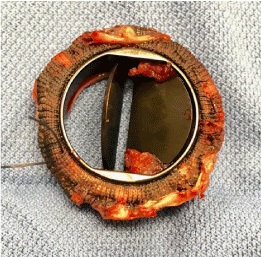 Figure 4: Bi-leaflet tilting Copper Valve. Thrombus had lead to limited mobility in one leaflet and absence of mobility in the other. View Figure 4
Figure 4: Bi-leaflet tilting Copper Valve. Thrombus had lead to limited mobility in one leaflet and absence of mobility in the other. View Figure 4
In this gentleman with a complex background medical history and acute valve thrombosis masquerading as an acute myocardial infarction, urgent coronary catheterization and fluoroscopy identified cause of presentation quite promptly. Due to lack of cardiothoracic care on-site and critical status urgent fibrinolytic therapy followed by transfer to the surgical centre led to successful outcome. Post-op care with optimization of therapy should improve long-term outcome. Randomized control trials form the evidence for concurrent use of aspirin along with anticoagulation with mechanical valves, with reduced mortality at the cost of a higher risk of intracranial hemorrhage [1].
Prosthetic valve thrombosis should be considered in the acutely unstable patient with chest pain. The use of transthoracic echocardiogram, trans-esophageal echocardiogram, computerized tomography plays a large role. In this case our patient was acutely unwell and deteriorating with a coronary occlusion within the differential diagnosis, the decision to bring to the cath lab urgently with a view to coronary angiogram and fluoroscopy super-ceding the use of transthoracic or trans-esophageal echocardiogram, which would have been performed had he been more stable. Fluoroscopy is usually diagnostic. ACC guidelines recommend emergency surgery where NYHA symptoms of 3-4 exist. This gentleman's raised troponin-T may have been secondary to transient obstruction of the coronary ostia by thrombosis of prosthesis valve thrombosis.
The pathogenesis of thrombosis in this case was complicated and likely due to a multitude of factors. This gentleman had a history of sub-therapeutic INR, multiple valve prosthesis, chronic inflammation in the form of inflammatory bowel disease, myeloproliferative disorder with eosinophilia, and atrial fibrillation.
Guidelines from the ACC [2] and ESC [3] both recommend an INR of 3 where mechanical aortic valve replacement exists with additional risk factors for thromboembolic events (in this case AF, hypercoagulable state and older generation mechanical mitral valve replacement). In this case the ESC guidelines would recommend an INR target of 4 given the presence of a Starr-Edwards valve prosthesis and additional patient related risk factors for thromboembolic events [3].
Eosinophilia is associated with hypercoagulabiltiy with multiple case reports describing valve thrombosis in this setting [4-6]. The thrombosis appears to have an unclear mechanism. Eosinophils release thrombogenic products, in particular the major eosinophil basic protein, during degranulation. This substance inhibits the capacity of thrombomodulin to function as a cofactor in the thrombin-induced activation of protein C, resulting in enhanced activity of the thrombotic cascade. Eosinophil peroxidase acts a powerful pro-coagulant and can inhibit the anticoagulant actions of heparin.
Our case highlights the importance of recognizing additional factors for prosthetic valve thrombosis. Awareness of these risks is warranted. This case also highlights the role of fluoroscopy in evaluation of valve motion. Fluoroscopy is the gold standard in assessing mechanical valve motion, but it does not allow visualization of thrombus. In this case it led to acute intervention which significantly changed this patient's outcome.
• Always consider Prosthetic valve thrombosis where clinical context exists.
• INR control is paramount.
• Fluoroscopy is quite helpful, with transthoracic echocardiogram and trans-esophageal echocardiogram first line in stable patient. CT can play a role.
• Pathogenesis of thrombosis in this case was be complicated, awareness of different causative factors, including hypercoagulable states, such as in this case is needed.