Egg allergy is the most common cause of food allergy in children under the age of 5, with a prevalence of 0.5-2.5%. In general, its prognosis is good; since in most cases tolerance is achieved within the early life in young children after following a exclusion diet. Alternatively, oral immunotherapy (OIT) or tolerance induction may be contemplated.
To analyze the epidemiological, clinical and analytical data of the patients included in the egg tolerance induction program.
Retrospective study in children diagnosed with egg protein allergy, included in the OIT program from June 2010 to December 2016. The values of egg protein-specific IgE and IgG4 were compared before and after OIT. They were divided into: group A, boiled egg tolerance; group B, partial; and group C, no tolerance.
50 patients, 60.6% male. Mean age at diagnosis 14.15 months (± 8.89). Atopic dermatitis associated in 84.8% of the patients while cow milk protein allergy in 42.4%.
During the OIT, 81.8% and 75.85% of the children showed gastrointestinal and cutaneous symptoms, respectively. After the procedure, 65.6% belonged to group A; 21.9% B and 12.5% to C.
The mean values of ovomucoid (OVM)-specific IgE were higher for group B and C both before and after OIT, as compared with A, although with no statistical significance. A significant decrease of egg white, OVM and ovoalbumin (OVA)- specific IgE values was found in the case of group A and OVM values in group B after OIT. No correlation was found between OVM and OVA-specific IgE and Ig G4 values.
The levels of egg protein-specific IgE are not predictors of tolerance and severity of the reaction during OIT.
Allergy, Egg, Desensitization, Immunoglobulin
In recent decades we have witnessed an increase in allergic diseases in general, and food allergy in particular, which is the most common cause of anaphylaxis in children [1]. In our midst, in the small child, egg allergy is the most common food allergy followed by cow's milk and fish.
Food allergy develops in the majority of cases in early childhood, producing an immediate hypersensitivity reaction mediated by IgE, which in most cases presents with cutaneous manifestations (urticarial and/or angioedema) in the two hours after taking the food. In some cases, an anaphylactic reaction may occur, with immediate digestive symptoms (vomiting), respiratory, cardiovascular and/or neurological symptoms.
These reactions should be distinguished from immunologically mediated digestive intolerance, in which late vomiting and/or diarrhea occur (more than 2 hours after ingestion), and other hypersensitivity processes such as esophagitis or Eosinophilic enterocolitis, whose pathophysiology is not fully known.
Egg allergy usually appears before two years of life and generally has a good prognosis, because in most cases it disappears in the first years of life [2,3], with 50% resolved at the age of 3 Years and 66% at 5 years, although there are recent studies suggesting a longer duration of allergy [4], being related to egg-specific IgE levels, with persistent allergy when there are higher levels of OVM. It has a prevalence of 0.5-2.5% in young children [5-8].
Egg white contains more than 20 different glycoproteins. The major allergens are: Ovomucoid (Gal d 1, OVM), ovalbumin (Gal d 2, OVA), ovotransferrin/conalbumin (Gal d 3) and lysozyme (Gal d 4).
The clinical history is fundamental for the diagnosis of egg allergy. In the presence of diagnostic suspicion, it is necessary to determine whether there is sensitization by in-vivo (prick test) and in vitro tests (specific IgE and serum). Subsequently, to confirm a clinical allergy to food, the controlled oral provocation test is performed.
Skin tests in children with Atopic Dermatitis (AD) and egg allergy show good sensitivity and high negative predictive value, but poor specificity. As a consequence, a negative test excludes a mediated IgE egg allergy, whereas a positive result does not accurately predict clinical reactivity [9].
Attempts have been made to establish specific IgE cutoff values versus clear to predict the outcome of the oral challenge.
IgG measurements and IgE/IgG4 ratios have been shown to be useful in monitoring the development of tolerance [10,11].
The controlled oral challenge test should be performed in a hospital setting by nurses and pediatricians experienced in detecting and treating potentially serious allergic reactions. This test is contraindicated in patients who have previously presented with anaphylaxis.
Until recently, the only possible treatment in egg allergy was to carry out an egg-elimination diet and derivatives, this is complicated because it is used in many processed and difficult-to-avoid foods, which carries a high risk of reaction by Inadvertent ingestion, which creates distress situations that affect the quality of life of allergic children and their families [12]. Treatment has been extended for several years by Oral Immunotherapy (OIT), which induces clinical egg tolerance in a large majority of allergic children [13-15].
The objective of the study is to provide the experience of the egg tolerance induction program of the Puerto Real University Hospital, analyzing and comparing the epidemiological, clinical and analytical data of egg-allergic patients, before starting OIT and after finish it.
A descriptive retrospective study of 50 children with allergy to egg (with specific and/or specific IgE and/or controlled oral challenge) in which OIT was performed between June 2010 and December 2016 in the Unit of Pediatric Allergy at Puerto Real University Hospital in Spain.
Patients who had not completed the ITO program at the time of data collection were excluded.
The procedure consists of administering dried and pasteurized powdered albumin, aliquoted and packed by the Pharmacy service in different doses, and increasing progressively until the equivalent dose of the usual daily oral intake is reached. The standard scheme would be 5, 10, 25, 50, 100, 225, 450, 900, 1800, 3600 mg and then provocation with boiled egg, in the form of clear cooked or overcooked omelet. In some patients, intermediate doses were used because of reactions that suggested more gradual increases (675, 2250 mg). The expected duration is about 12-14 weeks. Subsequently at home, indicate that they introduce biscuits-muffins, omelet undercooked, scrambled and finally mayonnaise and meringue.
After an explanation of the procedure and signed informed consent, a procedure was started, with the administration of antihistamine indefinitely until evolution was observed. Data from the clinical history and the complementary tests were collected: Determination of total IgE and specific IgE to egg white, OVM and OVA, as well as G4 Ig to ovomucoid and ovalbumin, before and after the end of OIT of patients with or without tolerance.
The results were analyzed according to three subgroups after ITO: Group A, children with good tolerance to boiled egg; Group B, with partial tolerance (patients who tolerate foods bearing eggs such as battering, pastries, etc.), and Group C, not tolerance.
Initially, a descriptive statistical study was performed for each variable considered. The results of the quantitative variables are presented in mean and standard deviations (mean ± SD).
The Kruskal-Wallis non-parametric test was used to compare the means of the three groups, because after the Shakira Wilk normality test, there was no value following a normal distribution.
In the next phase of the study (before-after the procedure), the comparison was performed in the 3 groups:
-Group A and B: Values that did not have a normal distribution, Wilcoxon T test was applied.
-Group C: Total IgE and specific IgE OVM, maintained a normal distribution, so that the Student's T test was applied for paired data and in the specific OVA E-E and clear when not following normal distribution, the test T of Wilcoxon.
A p < 0.05 was considered a statistically significant difference, with a confidence level of 95%.
Finally, the correlation between quantitative variables after the completion of OIT was studied. The variables used are OVA and OVM-specific immunoglobulins, as well as Ig G4 to OVA and OVM. With less than 30 missing data, the normality of the variables was demonstrated. As we did not follow a normal distribution, we calculated the Spearman rank coefficient. In addition, the relationship between the duration of OIT and specific Ig prior to desensitization was checked by performing the Pearson correlation.
The statistical analysis was done with the SPSS version 17 program.
50 patients, 30 men (60) and 20 women (40%), with age at the onset of ITO of 88.97 ± 33.3 months. The mean age at diagnosis was 14.15 months (± 8.89).
27.3% had Atopic Dermatitis (AD) and allergy to other foods, and 39.4% had rhinitis or asthma, and food allergy was most frequently associated with Cow's Milk Protein Allergy (CMPA), with a 42.4% and of all patients, 84.8% had AD in isolation.
57.6% of the children had at least one parent with a history of atopy and 18% were exposed to tobacco smoke.
During induction of tolerance, 81.8% and 75.85% of children presented digestive and cutaneous symptoms, respectively. The most frequent cutaneous symptom is generalized urticarial together with cutaneous lesions in the perioral area (54.6%) and the digestive tract, vomiting with 42.4%.
After the procedure, the patients were divided into three subgroups according to the tolerance acquired: A) With tolerance to the cooked egg (65.6%); B) With partial tolerance (21.9%) and C) Non-tolerance (12.5%), with a duration of 13.64 (± 6.44), 18.14 (± 9.96) and 12.25 (± 7.8) weeks, respectively.
No differences were found in serum levels of total IgE and IgE specific to egg proteins among the 3 groups.
Group C (non-tolerant) and group B (partial tolerance) showed mean levels of IgE versus OVM higher before and after OIT than the group of patients tolerating (group A), although not statistically significant (Figure 1).
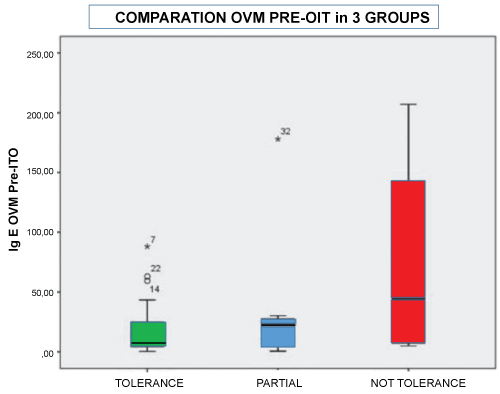 Figure 1: Comparison of ovomucoid IgE values (OVM) oral pre-immunotherapy (pre-ITO) in all 3 groups.
View Figure 1
Figure 1: Comparison of ovomucoid IgE values (OVM) oral pre-immunotherapy (pre-ITO) in all 3 groups.
View Figure 1
There was a decrease in the specific IgE to OVM values at the end of the program in the 3 groups. Those who tolerated had a lower mean age at onset of OIT (80.86 months ± 28.57) than the other two groups: group B (102.14 ± 44.31) and group C (106.75 ± 34.07) (Figure 2).
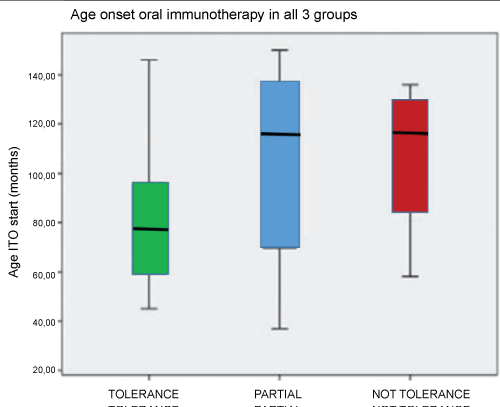 Figure 2: Age onset of oral immunotherapy (OIT) in all 3 groups.
View Figure 2
Figure 2: Age onset of oral immunotherapy (OIT) in all 3 groups.
View Figure 2
A significant decrease in IgE specific values was found against egg white, OVM and OVA after OIT in the group of patients they tolerate. However, there were no differences in total IgE.
In the other two groups, no differences were found, except in the partial tolerance group, in which the OVM levels if significant differences were found (Table 1 and Figure 3).
Table 1: Comparison of total and specific Ig E levels to egg proteins in allergic patients before and after desensitization (mean ± SD) EGG TOLERANCE. View Table 1
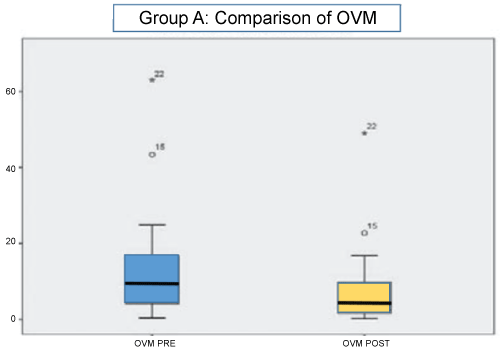 Figure 3: Comparison of ovomucoid IgE values (OVM) before and after desensitization in group A.
View Figure 3
Figure 3: Comparison of ovomucoid IgE values (OVM) before and after desensitization in group A.
View Figure 3
A linear relationship between Ig and OVM post/IgE and post OVA was observed, as well as post OVA IgG4 post/IgG OVA post, with r = 0.58 and p = 0.001, and r = 0.887 and p < 0.001, respectively. Linear correlation was also observed between the specific IgE values prior to the introduction to the program and the duration in ITO weeks. The linear relationship between pre-OVA, pre-OVA, pre-egg-clear and duration in weeks is statistically significant, with r = 0.438, r = 0.509 and r = 0.625, respectively (Table 2).
Table 2: Correlation between specific Ig Es prior to desensitization and duration in weeks of oral immunotherapy. View Table 2
In this study, it was found that the mean age at diagnosis corresponded approximately to the introduction of the egg, although in some families the recommendations made by Pediatricians were not followed, such as introducing the cooked egg, first the yolk and then the clear, progressively, giving by own initiative the clear little cooked (meringues, eggs passed by water, ...), which increases the risk of allergic reactions.
The fact that allergic reactions occur with the introduction of clear, is due to the fact that the egg-mucoid is the dominant allergen, having characteristics that make it especially allergenic, such as heat resistance and digestion by proteases, due to the presence of potent disulfide bonds. However, the yolk is usually well tolerated because of its low protein content, the main protein being alpha-livethine (Gal d 5).
There is great heterogeneity in egg allergy phenotypes, depending on the tolerance of whether the patient's specific IgE recognizes conformal epitopes or whether they are directed against sequential epitopes, in which case persistence is more likely and there is increased risk of serious reactions. The study of the specific IgE response to these epitopes is still under investigation, although it would be a great advance to apply it in the clinic.
Patients diagnosed with atopic dermatitis have a more frequent allergy to the egg, which suggests that they produce AD, but these children are usually diagnosed when they are breastfeeding, with no relation to the egg intake, although there are some which worsen eczema in a delayed manner, making it difficult to establish causality [16,17]. There are children with AD who tolerate the egg and present positive tests, this may be an epiphenomenon associated with the increase of total IgE. A high percentage of patients in our sample had atopic dermatitis, with no differences between sexes or in the subgroups in patients with egg allergy.
The presence of allergy to other foods is frequent, especially with a higher risk of developing peanut sensitization [18]. In this study, the most frequent occurrence was CMPA (42.4%) and then allergy to nuts in 9%, which is a frequent use in our environment, but not so much as in the Anglo-Saxon countries.
Among environmental factors, there is a low percentage of parents who smoke, it could be due to the fact that having a child diagnosed with allergy makes them more aware and avoid exposure to allergens and irritants such as tobacco smoke. This may constitute a bias of inverse causality, or a bias of answer.
The total IgE values are not very profitable, since they have little specificity. Its elevation may be due to several pathologies, among which is the atopic dermatitis; Thus elevated levels are not related to non-tolerance, finding no differences in this study.
Specific IgE allergens are a marker of allergic sensitization and not of allergic disease. That is, the existence of positive allergic tests, alone, does not justify the diagnosis of food allergy if there is no associated allergic reaction.
The cut-off values of IgE differ according to the studies [3,19,20]. In children younger than 2 years of age, it has been suggested that a specific IgE to clear value greater than 2 KU/L, has a Positive Predictive Value (PPV) of 95% reaction [21]. However, in the study by Ando, et al. values of ovomucoid-specific IgE greater than 11 KU/L have a higher risk of reacting to boiled egg, as well as specific IgE to egg white Greater than 7.4 KU/L, with a similar PPV. Therefore, specific IgE values are useful for assessing the potential risk of reaction in the controlled exposure test, being higher in patients with severe reactions.
Group A showed lower levels of specific IgE compared to the other two groups, finding differences before and after sensitization, which is interpreted by the acquisition of tolerance to the cooked egg. The decrease is not due to the elimination of specific IgE production but to a blockage of the allergic response. This justifies that allergic tests (prick test and specific IgE) continue to be positive in patients who tolerate food, either because they have acquired the tolerance naturally (subclinical sensitization) or acquired through OIT [4,13-15].
Prior IgE levels specific to egg white proteins are IgE values that should not be interpreted in isolation to predict the tolerance and severity of the reaction but in combination with the clinical history. In addition, attempts have been made to find biomarkers predictive of food tolerance before initiating OIT, such as MCP-1α and MIP-1 [22], which predicts tolerance in blood.
There is a significant decrease in pre and post-OVM IgE values, both in group A and in B, but not in C. This finding, together with the rise of specific IgG4, is similar to that found produces during the administration of specific immunotherapy against neumoallergens [23].
The increase in specific Ig G4 values has been related to the development of tolerance [10,11,24], so it is expected to find correlation coefficients between specific IgE and G4 Ig with higher values in the tolerant group. In the present study, a correlation was found between IgE versus OVA and OVM and IgG4 OVA and OVM in the values collected after the end of the sensitization. There seems to be no clear relationship between Ig G4 values and tolerance, because there are patients who have low levels of specific G4 Ig and do not tolerate it, perhaps there are other factors that influence these levels, such as elevation of IL-10 and TGF-β and other molecules that we do not know today and therefore we cannot measure.
A positive linear correlation was observed between the time it took to finish the program with the previous specific IgE levels, so that the lower value of these at the time of starting the OIT would be the time that it needed for its final and this correlation was significant, not differentiating between the groups. This is due to a greater frequency of reactions during the procedure, which causes intermediate doses to be introduced and as a consequence of a slower OIT procedure in patients with higher specific Ig values because they are at greater risk of allergic reaction, and it would be a bias.
The limitations of the work include a small sample size, as well as not knowing the status of the patients when the analysis was performed before and after the end of the program. In addition to the difficulty in pigeon-holding patients in group B, as there are some children who have tolerated, but in the months have stopped taking the cooked egg, due to a variety of causes including the presence of symptoms or simply refusing food for fear of a possible reaction.
It could be concluded that we found differences (although without statistical significance) in the specific IgE levels against egg white proteins as a function of the tolerance and the presence of reactions during the OIT process and that, similarly, we found decreases of IgE and increase of IgG4.