Arrhythmogenic right ventricular dysplasia (ARVD) is an inherited cardiomyopathy characterized by right ventricular structural abnormalities, dysfunction and ventricular tachycardia (VT) secondary to fibrofatty replacement of the myocardium [1]. We report a male teenager with negative family history, presenting with sustained VT after sports practice. He had waxing and waning troponin levels without myocardial ischemia and satisfied the revised task force criteria for diagnosis of ARVD. ARVD needs to be strongly considered in the differential diagnosis of phasic troponin elevation in the absence of myocardial ischemia.
ARVD, Troponin, VT
A 15-year-old previously healthy male with a negative family history, presented with sudden onset dizziness and shortness of breath immediately after baseball practice. He denied palpitations or chest pain. His symptoms persisted for 10 minutes before his parents decided to call an ambulance. His initial rhythm strip revealed wide complex tachycardia at a rate of 230 beats per minute with AV dissociation. Paramedics attempted chemical cardioversion with adenosine unsuccessfully but converted to sinus rhythm with amiodarone bolus. On admission, his baseline electrocardiogram (ECG) showed normal sinus rhythm with left axis deviation, incomplete right bundle branch block, suspicious epsilon wave and normal QTc interval. Baseline echocardiogram revealed normal structure, anatomy and function. He had a normal hemogram, serum chemistry, drug screen and elevated troponin level (3.53 ng/ml; normal < 0.05 ng/ml). Telemetry monitoring showed intermittent runs of VT as well as frequent premature ventricular complexes with bi-directional axis (Figure 1A). Differential diagnosis of possible myocarditis, arrhythmogenic right ventricular dysplasia (ARVD), and catecholaminergic polymorphic ventricular tachycardia (CPVT) were entertained as the QT measured normal. Comprehensive viral titers and cultures were negative. Cardiac MRI showed evidence of right ventricular (RV) wall motion abnormalities as well as increased indexed RV end diastolic volume of 122.87 ml/m2 and decreased RV ejection fraction (RVEF) of 41% without late gadolinium enhancement (LGE). However, it was planned to repeat cardiac MRI in 4 weeks to evaluate for any progressive changes. He continued to have intermittent non-sustained VT. Repeat troponin levels revealed a waxing and waning pattern (Figure 2A). Cardiac catheterization with coronary angiograms and endomyocardial biopsy was performed. He had unremarkable hemodynamics with normal LV and RV end diastolic pressures, normal right coronary dominant system without congenital coronary abnormalities or bridging. Endomyocardial biopsy from the right ventricular septum revealed diffuse interstitial fibrosis with residual myocytes < 50% without inflammatory cells (Figure 2B and Figure 2C). Serial ECGs showed T-wave inversion in the precordial leads V1-V4 in the absence of right bundle branch block with terminal activation delay in V1 (Figure 1B). Signal averaged ECG showed filtered QRS duration ≥ 114 msec, with normal duration of terminal QRS < 40 µV (≥ 38 msec) and normal root-mean-square voltage of terminal 40 msec (≤ 20 µV). Repeat cardiac MRI 4 weeks later failed to demonstrate LGE but revealed decreased RV function with mild dyskinesia of the RV outflow tract, indexed RV end diastolic volume 128 ml/m2 and RVEF 45%. He satisfied at least 3 major and 1 minor revised Task Force criteria for ARVD. He was initiated on oral Sotalol and a cardioverter-defibrillator (ICD) was implanted. He was restricted from competitive and strenuous physical activities. Two months later, he received an appropriate ICD shock due to fast VT, while jumping on the trampoline. The genetic testing from EDTA blood revealed pathogenic heterozygous mutation in the Plakophillin gene (PKP-2, c.368G >A(p.W123X)), previously reported in a family with ARVD. Family screening was offered to the first degree relatives.
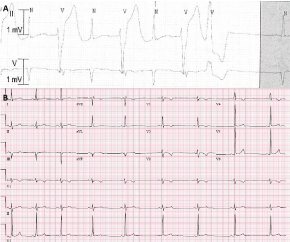 Figure 1: A) Bidirectional PVCs on telemetry monitoring; B) 12 lead electrocardiogram shows sinus bradycardia with a ventricular rate of 52 beats per minute, left axis deviation, T wave inversion in anterior precordial leads (V1-V4) without evidence of right bundle branch block. QTc is normal at 413 msec. There are other ECG features of ARVD: S nadir to QRS end in V1 of 60 msec (> 55 msec), and QT dispersion (> 65 msec). View Figure 1
Figure 1: A) Bidirectional PVCs on telemetry monitoring; B) 12 lead electrocardiogram shows sinus bradycardia with a ventricular rate of 52 beats per minute, left axis deviation, T wave inversion in anterior precordial leads (V1-V4) without evidence of right bundle branch block. QTc is normal at 413 msec. There are other ECG features of ARVD: S nadir to QRS end in V1 of 60 msec (> 55 msec), and QT dispersion (> 65 msec). View Figure 1
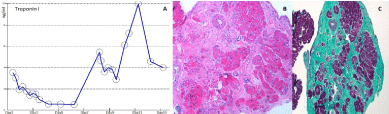 Figure 2: A) Serum Cardiac Troponin I Level shows persistently elevated level with intermittent rise. Initial level: 3.5 ng/ml, peak level: 10 ng/ml; B) Right ventricular endomyocardial biopsy demonstrates diffuse interstitial fibrosis > 50% by visual estimation, without evidence of active vasculitis, myocarditis or coronary artery intimal sclerosis in Hematoxylin and eosin stain (H&E stain); C) in Gomoro Trichrome stain (fibrosis- area in cyan, normal myocardium- area in magenta). View Figure 2
Figure 2: A) Serum Cardiac Troponin I Level shows persistently elevated level with intermittent rise. Initial level: 3.5 ng/ml, peak level: 10 ng/ml; B) Right ventricular endomyocardial biopsy demonstrates diffuse interstitial fibrosis > 50% by visual estimation, without evidence of active vasculitis, myocarditis or coronary artery intimal sclerosis in Hematoxylin and eosin stain (H&E stain); C) in Gomoro Trichrome stain (fibrosis- area in cyan, normal myocardium- area in magenta). View Figure 2
Arrhythmogenic right ventricular dysplasia (ARVD) is an inherited cardiomyopathy characterized by right ventricular structural abnormalities, dysfunction and ventricular tachycardia (VT) secondary to fibrofatty replacement of the myocardium [1-4]. It is an important cause of sudden death in young apparently healthy individuals. Advances in genetics have led to the discovery of desmosomal mutations in majority of cases with typical autosomal dominance with variable penetrance [5]. Desmosome functions as a cell-to-cell adhesion, therefore the disruption is associated with myocyte apoptosis and subsequent replacement of myocardium by fibrofatty tissue which is a pathologic hallmark of the disease. Due to its variable penetrance, the diagnosis of ARVD is often challenging. International Task Force criteria that facilitated the diagnosis of ARVD were proposed in 1994. The criteria were revised in 2010 with incorporation of novel diagnostic modalities and genetic fundamentals to improve diagnostic sensitivity while maintaining high specificity [3].
Our patient presented with sustained VT during the recovery phase of the exercise and had premature ventricular complexes with bi-directional axis on the telemetry. The initial differential diagnoses included viral or autoimmune myocarditis, Long QT syndrome, catecholaminergic polymorphic ventricular tachycardia and ARVD. Myocarditis and autoimmune conditions, such as scleroderma, lupus, sarcoidosis, were ruled out [6-8]. Waxing and waning troponin levels were indicative of alternative diagnosis than myocarditis. Myocardial ischemia was excluded with normal coronary angiograms. Our patient fulfilled 3 major and 1 minor criterion for definite diagnosis of ARVD based on the Revised Task Force Criteria. The regional RV dyskinesia and increased RV end diastolic volume on cardiac MRI accounted for 1 major criterion from the category of regional dysfunction and structural alterations. The RV endomyocardial biopsy illustrated fibrous replacement of > 50% myocytes accounting for another major criteria based on tissue characterization. The major criteria of repolarization abnormality was satisfied with evidence of T wave inversion in lead V1-V4 and terminal activation delay in the absence of right bundle branch block as well as minor criteria in the form of epsilon wave and 1 abnormal criteria on signal average ECG from the depolarization abnormalities category. The initial VT was documented on a rhythm strip and therefore characterization was difficult. The evidence of sustained VT has supported the diagnosis as our patient had already satisfied the definite diagnosis for ARVD. The genetic testing revealed a heterozygous mutation of Plakophillin-2 gene, previously reported in a patient with ARVD and also the most common gene mutation associated with ARVD [9,10].
The possible cause of waxing and waning troponin levels as seen in our patient can be related to various factors inclusive of frequent PVCs and runs of non-sustained VT, myocyte apoptosis, increased myocardial fiber injury through cytokines release and tendency for myocardial enzyme leakage due to constant remodeling of intercalated disc [11-13]. The troponin elevation in myocarditis is not phasic. Normal coronary angiograms ruled out ischemia as a potential cause. Elevation of serum cardiac troponin-I has been described in dogs with ARVD without myocardial ischaemia [14]. Our patient had no apparent reasons for elevated troponin levels in the form of increased ectopies, correlation with non-sustained VT or any external cardioversion/defibrillation. ARVD must be considered in the differential diagnosis of phasic troponin elevation in the absence of myocardial ischemia.
Dr. Chang Chung-Ho and Dr. Manjula Garapati for pathology slide and interpretation.
This research received no specific grant from any funding agency, commercial or not-for-profit sectors.
None.