Isolated central nervous system and dural metastasis from urothelial cancer are exceedingly rare. We describe 2 unique cases of intracranial lesions in patients with urothelial carcinoma-one with isolated brain and dural brain metastasis, and another with Nocardia brain abscess.
Case 1 presented with a solitary right frontal lobe metastasis, 8 years after the initial diagnosis of non-muscle invasive high grade urothelial carcinoma. Subsequently, rapid progression with dural carcinomatosis was noted, in the absence of additional extracranial systemic metastasis. Case 2 describes a patient presenting with seizure with identification of a single frontal lobe lesion about 2 years after initial diagnosis of metastatic urothelial carcinoma. Upon resection, it was diagnosed to be a bacterial abscess caused by Nocardia farcinica. He was treated with parenteral antibiotics that resulted in resolution of the brain lesions.
Our cases highlight two important messages. Intracranial metastases in urothelial cancer are uncommon but should be considered even if long after initial diagnosis or in the absence of systemic metastasis. Also, tissue diagnosis is essential to confirm the diagnosis, to rule out other treatable etiologies such as abscess.
Urothelial cancer, Intracranial metastases, Brain abscess
BCG: Bacillus Calmette-Guerin; CNS: Central Nervous System; MVAC: Methotrexate, Vinblastine, Doxorubicin and Cisplatin; SEER: Surveillance, Epidemiology, and End Results; WBRT: Whole Brain Radiation Therapy
Urothelial carcinoma is the sixth most common malignancy and the second most common genitourinary malignancy in the United States [1]. Based on the Surveillance, Epidemiology, and End Results (SEER) statistics fact sheet, around 79,000 new cases and around 16,000 deaths attributed to urothelial carcinoma were estimated in 2017 [2]. Brain metastasis are seen uncommonly, usually reported late in the course of the disease, and prognosis in these cases is dismal [3]. As systemic therapy improves, it is possible that the incidence of presentation with metastases to the brain as a sanctuary site can increase. The standard systemic chemotherapy used in urothelial cancer has minimal or no penetrance within the brain parenchyma, hence potentially enhancing the susceptibility for spread to the central nervous system (CNS). Radiotherapy including whole-brain radiotherapy (WBRT) and/or stereotactic radiation, surgical resection and systemic therapies, are the various treatment options available to these patients [4]. More effective therapies are needed both, to prevent against spread to the nervous system and to treat symptomatic disease. This manuscript describes 2 rare presentations of intracranial disease in patients with urothelial cancer. The first case discussed is of urothelial carcinoma with isolated brain metastases and subsequent dural carcinomatosis. The second description is of a patient with metastatic urothelial carcinoma whose brain lesion was eventually diagnosed to be a brain abscess on biopsy, growing Nocardia species (Table 1).
Table 1: Summary of cases. View Table 1
A 70-year-old woman with a remote history of breast cancer presented with hematuria and was diagnosed with non-muscle invasive high grade urothelial carcinoma after cystoscopy and biopsy. She was then treated with intravesical Bacillus Calmette-Guerin (BCG) and cystoscopy surveillance every 3 months. Seven years after this initial diagnosis, abdominal CT scan showed a recurrence with a mass in anterior bladder wall invading the abdominal wall. Subsequently, decision was made to start her on neoadjuvant chemotherapy with three cycles of cisplatin and gemcitabine, followed by radical cystectomy with salpingo-oophorectomy, bilateral pelvic node dissection and ileal loop urinary diversion, leaving negative margins. Pathology revealed thick trabeculae of urothelial carcinoma with extensive necrosis (Figure 1, Case 1). She was in complete remission for one year when she presented with left upper extremity monoparesis along with suspected seizure. MRI of the brain revealed a heterogeneous enhancing mass at the gray-white matter interface in the posterior right frontal lobe measuring, approximately 22 mm in diameter, with some surrounding edema and mass effect on the adjacent motor cortex (Figure 2, Initial). CT of thorax, abdomen and pelvis were performed at this time and did not any evidence of metastasis or disease recurrence. Soon thereafter, she underwent stereotactic right frontoparietal craniotomy with MRI-guided computer-assisted volumetric resection of right motor cortex tumor. Pathological examination showed high grade papillary urothelial carcinoma with extensive necrosis. The tumor cells were strongly immunoreactive for CK7 and CK20 and many had nuclear immunoreactivity for p63. No definite immunostaining was noted for TTF1, PAX2, BRST2, mammoglobin or RCC antibodies. The combined morphologic features and immunoprofiles were most consistent with metastatic urothelial carcinoma, rather than breast, as primary site. Nine months later, she had a witnessed seizure with subsequent MRI brain revealing diffuse pachymeningeal/dural enhancement throughout the right cerebral hemisphere with focal areas of dural enhancement overlying the right frontal operculum and right anterior temporal lobe (Figure 2, Restaging I). Overall, MR brain findings were highly suggestive of dural carcinomatosis. Following this, she underwent stereotactic right frontotemporal craniotomy for resection of focal dural-based metastases. Histopathological examination was again consistent with metastatic urothelial carcinoma. The patient underwent whole brain radiation therapy. Despite this, a follow up MRI brain done 4 and 6 months later revealed recurrent nodular dural thickening and enhancement overlying the right frontal and temporal lobes consistent with recurrent/persistent dural carcinomatosis (Figure 2, Restaging II and Figure 2, Restaging III). Given the paucity of therapeutic options and decline in performance status, hospice care was pursued.
 Figure 1: Case 1): Thick trabeculae of urothelial carcinoma with extensive necrosis; Case 2a): Abscess formation in brain parenchyma; Case 2b): Brown and Brenn stain branching weakly Gram positive bacilli, suggestive of Nocardia or Actinomyces species. View Figure 1
Figure 1: Case 1): Thick trabeculae of urothelial carcinoma with extensive necrosis; Case 2a): Abscess formation in brain parenchyma; Case 2b): Brown and Brenn stain branching weakly Gram positive bacilli, suggestive of Nocardia or Actinomyces species. View Figure 1
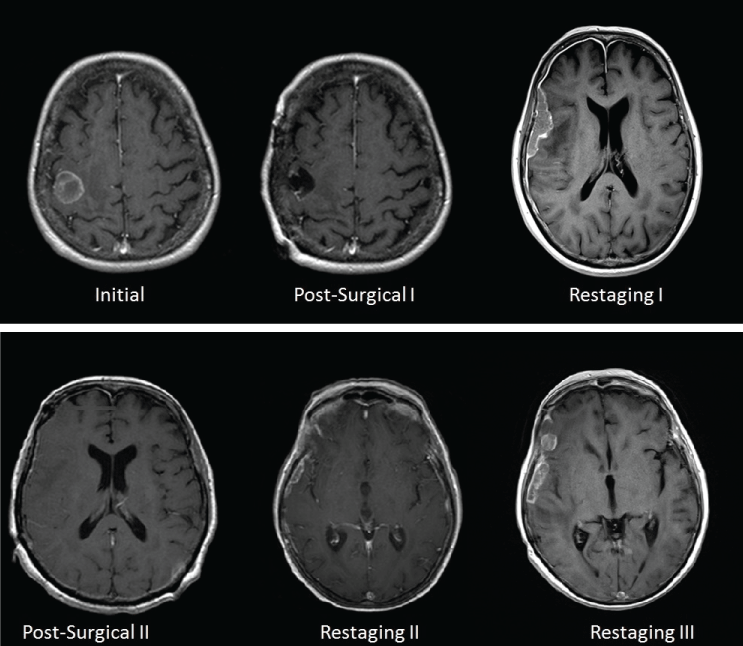 Figure 2: Case 1): Initial: MRI brain demonstrating a heterogeneously enhancing solitary right posterior frontal lobe brain metastasis; Post-Surgical I): MRI brain reveals adequate resection of brain metastasis; Restaging I): MRI brain demonstrates diffuse pachymeningeal enhancement with large focal plaque-like dural mass overlying the right lateral frontal and temporal lobes; Post-Surgical II): MRI brain revealing adequate resection of plaque-like right frontotemporal dural based metastasis; Restaging II): MRI 4 months after resection of dural metastasis demonstrates new focal nodular and plaque-like enhancing dural metastases overlying the right cerebral hemisphere; Restaging III): MRI 6 months post-resection of dural metastasis demonstrates continued progression of dural carcinomatosis. View Figure 2
Figure 2: Case 1): Initial: MRI brain demonstrating a heterogeneously enhancing solitary right posterior frontal lobe brain metastasis; Post-Surgical I): MRI brain reveals adequate resection of brain metastasis; Restaging I): MRI brain demonstrates diffuse pachymeningeal enhancement with large focal plaque-like dural mass overlying the right lateral frontal and temporal lobes; Post-Surgical II): MRI brain revealing adequate resection of plaque-like right frontotemporal dural based metastasis; Restaging II): MRI 4 months after resection of dural metastasis demonstrates new focal nodular and plaque-like enhancing dural metastases overlying the right cerebral hemisphere; Restaging III): MRI 6 months post-resection of dural metastasis demonstrates continued progression of dural carcinomatosis. View Figure 2
A 53-year-old man was diagnosed with high grade muscle-invasive urothelial carcinoma with a cystoscopy. Neoadjuvant chemotherapy with cisplatin and gemcitabine was followed by radical cystectomy. Pathology revealed urothelial carcinoma invading through muscularis propria into perivesical adipose tissue. Three of 11 pelvic lymph nodes were positive for metastatic carcinoma with extranodal extension. Three cycles of combination adjuvant chemotherapy with methotrexate, vinblastine, doxorubicin and cisplatin (MVAC) followed surgery. CT scan of abdomen and pelvic regions performed a year later revealed diffuse lytic osseous metastases in lumbar spine and pelvic bones. New retroperitoneal/abdominal lymphadenopathy including retrocaval lymph and left para-aortic nodes was also noted. While pursuing radiation therapy to the spine, progression with metastasis to the liver was noted. Prior to initiation of systemic therapy, he developed a seizure. MRI brain revealed a 2.3 cm bilobed enhancing right frontal lobe lesion at the gray-white matter interface with associated restriction diffusion and mild surrounding vasogenic edema, concerning for metastatic disease (Figure 3, Initial). He underwent craniotomy and resection of the brain lesion, the pathologic examination of which revealed features suggestive of an abscess (Figure 1, Case 2a). Brown and Brenn staining demonstrated numerous branching weakly Gram positive bacilli suspicious of Nocardia (Figure 1, Case 2b). Tissue culture was confirmatory for Nocardia farcinica. Treatment with intravenous meropenem and ceftriaxone was initiated, which was later switched to oral linezolid and amoxicillin-clavulanate based on culture susceptibilities. Follow up MRI, 4 months later, showed a complete resolution of the abscess and did not show a recurrent infection or new abscess. However, he continued to have systemic disease progression despite multiple chemotherapy regimens including platinum agents and pemetrexed. Given rapidly progressive disease and declining performance status, hospice referral was suggested and the patient died 3.5 years after the initial diagnosis.
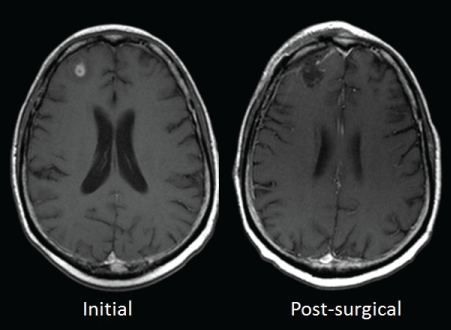 Figure 3: Case 2): Initial: MRI demonstrates bilobed enhancing right frontal lesion with restricted diffusion; Post-Surgical): MRI reveals adequate treatment of the brain abscess. View Figure 3
Figure 3: Case 2): Initial: MRI demonstrates bilobed enhancing right frontal lesion with restricted diffusion; Post-Surgical): MRI reveals adequate treatment of the brain abscess. View Figure 3
Neurological involvement with urothelial carcinoma can be variable and mandates consideration of an elaborate differential. We describe a patient with brain metastasis with subsequent dural involvement, with CNS being the only site of metastatic disease. Our second case describes a rare but potentially fatal complication of Nocardia farcinica brain abscess in a patient with metastatic urothelial cancer.
Intracranial metastasis and meningeal carcinomatosis with urothelial carcinoma is a rare phenomenon with previously reported rates varying between 0%-7% in various reports [5,6]. Isolated brain metastasis without extracerebral metastasis and meningeal involvement is further less common, reports only being in the form of case reports [7-11]. Our case represents a first case, to the best of our knowledge, of a patient presenting with both parenchymal intracranial as well meningeal metastasis in a patient with a history of urothelial carcinoma previously treated with surgery and adjuvant therapy, in the absence of any additional extracranial systemic metastasis. CNS remains to be a potential "sanctuary site" due to poor penetration of systemic chemotherapeutic agents attributed to the presence of blood brain barrier [12]. Treatment options for brain metastasis include neurosurgical or radiation therapy as stereotactic radiosurgery and whole brain radiation or a combination of these approaches [4]. Treatment options for meningeal involvement can be variable with no standard guidelines. Intrathecal therapy and radiation therapy have both been described [13]. With improved survival in urothelial cancers secondary to advent of effective chemotherapy and immunotherapy options, this incidence can be expected to increase. The increasing risk of CNS metastasis raises a question if surveillance neurological imaging should be considered in these patients. At this point, there is no evidence to pursue this approach. Nevertheless, CNS evaluation for metastases should be recognized in patients with neurological symptoms even in the absence of other systemic metastasis.
The second case describes another rare presentation of Nocardia farcinica brain abscess. Nocardia brain abscess are associated with high mortality rate of around 60% and are more commonly seen in immunosuppressed patients [14]. Hence, patients on chemotherapy treatment for metastatic cancers are at high risk and should be considered in the differential in patients with intracranial lesions. Our case underscores the importance of tissue diagnosis in patients presenting with brain lesions with an underlying metastatic malignancy. Mainstay of treatment of Nocardia brain abscess remains to be surgical aspiration or excision along with long term antibiotics [14]. Treatment is recommended with two intravenous antibiotics for 3-6 weeks followed by oral antibiotics for around 6-12 months [15,16]. Susceptibility testing should be used to guide antibiotic choice due to high frequency of anti-microbial resistance especially with Nocardia farcinica [17]. Linezolid has been shown good permeability in tissues and can be considered for the transition to oral therapy [18]. Clotrimazole is another commonly used oral antibiotic in these patients but frequent resistance should be acknowledged, as in our patient [19]. Early recognition and prompt treatment of the abscess resulted in complete response of the abscess in our patient; although he died subsequently due to progressive metastatic disease.
Isolated CNS metastasis should be acknowledged in patients with metastatic urothelial carcinoma. Tissue diagnosis of brain lesions must be pursued for accurate identification to rule out other, potentially treatable causes of brain lesions such as infection, in these immunosuppressed patients.
No conflict of interest or relevant financial disclosures.
No data repository exists for this case report.