Cocaine is a drug that is abused by 5 million Americans and most of the cocaine that comes to the United States has been tainted with Levamisole, a veterinary anti-helmintic, which recently has been linked to ANCA Associated Vasculitis (AAV) [1]. AAV is an unusual necrotizing vasculitis that attacks small vessels more often, and can have an ominous prognosis if left untreated [2].
We present a case of new onset P-ANCA associated vasculitis in a patient with a history of cocaine abuse who was lost to follow up and returned six years later with shortness of breath due to diffuse alveolar hemorrhage and heart failure with reduced ejection fraction, in addition to gross hematuria secondary to focal segmental necrotizing and sclerosing glomerulitis diagnosed with kidney biopsy in the setting of continued cocaine use.
The patient reappeared with purpuric lesions on her face, upper and lower extremities bilaterally with blebs and bullae that ulcerated. She was discharged after debridement but returned with worsened lesions when she discontinued wound care. Patient was readmitted with necrotic lesions complicated by myiasis; underwent re-debriment and was sent to a burn unit where she was treated with skin grafts.
To our knowledge there are only five other cases with greater Total Body Surface Area (TBSA) involvement, only one other case that reported membranous nephropathy and no other report with full-thickness skin necrosis and lupus-like nephritis combining membranous and focal proliferative/sclerosing glomerulitis.
Since most of the cocaine in the US is adultered with Levamisole, Levamisole induced AAV should be included as a differential diagnosis in patients presenting with characteristic purpuric lesions, hematuria, positive ANCA serology and history of cocaine abuse.
A 41-year-old Hispanic female with known medical history significant for bronchial asthma presents to the Emergency Department (ED) in February 2010, complaining of a 3-week long history of an erythematous tender rash with irregular margins on the tip of her nose, upper and lower extremities (Figure 1) accompanied by bilateral elbow, knee and ankle pain. The rest of her physical exam and laboratory data was essentially normal, except for a Urine Drug Screen (UDS) which was positive for cocaine. She was managed symptomatically with antihistamines and steroid therapy with moderate improvement. Given presenting complaint of joint pain, she was worked up for possible connective tissue disease; ANA was positive, ANCA titers were 1:160, with positive Myeloperoxidase (MPO-ANCA), negative Proteinase 3 (PR3-ANCA) and a noted perinuclear staining pattern (P-ANCA). Anti-dsDNA antibodies, rheumatoid factor, and anti-smooth muscle antibodies were negative. (Please see Table 1 for detailed laboratory results). A skin biopsy was done which later demonstrated a neutrophil-rich infiltrate around the vessels of the dermis with fibrin thrombi within the superficial vessels. She was discharged with an oral prednisone taper but lost to follow-up.
Table 1: Lab work from initial encounter 2010. View Table 1
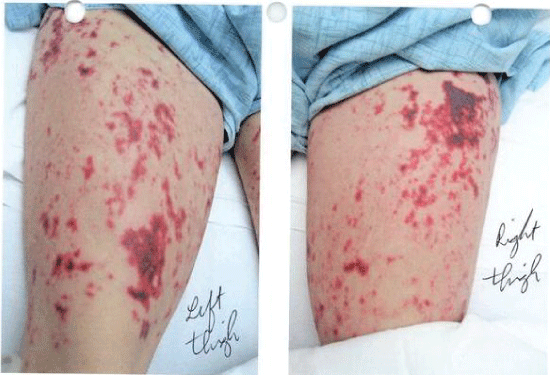 Figure 1: Skin lesions on initial presentation 2010. View Figure 1
Figure 1: Skin lesions on initial presentation 2010. View Figure 1
Patient reappeared approximately 6 years later; when she complained of gross hematuria, chest pain, hemoptysis, and generalized weakness. Physical exam showed her to be pale, moderately hypertensive with a Blood Pressure (BP) of 159/82 mmHg, with fine rales throughout the entirety of bilateral chest on auscultation. Review of initial laboratory data proved her to be leukopenic and anemic, with White Blood Cell (WBC) count of 3,200/uL, Hemoglobin (Hb) of 5.1 g/dL and Hematocrit (HTC) of 15.6%; she was now also in acute renal failure with creatinine of 6.3 mg/dL and BUN 53 mg/dL. Her Brain Natriuretic Peptide (BNP) was elevated at 1963 pg/ml and a urinalysis was positive for protein (100 mg/dL), WBC's (14/hpf) and Red Blood Cells (RBC) (> 180/hpf). UDS was again positive for cocaine. A Computerized Tomography (CT) of the chest showed prominent bilateral patchy infiltrates and symmetric moderate pleural and small pericardial effusions (Figure 2); whereas abdominal CT scan was unremarkable except for mild pelvic fluid and mild soft tissue edema. Patient was admitted, transfused Packed Red Blood Cells (PRBC’s), and started on high-dose steroid therapy. The work up done revealed diffuse alveolar hemorrhage diagnosed by bronchoscopy, acute kidney injury suspected to be due to rapidly progressive glomerulonephritis, anemia of chronic inflammation exacerbated by hematuria and new onset heart failure with grade III diastolic dysfunction and an ejection fraction of 45-50%. Immune serology continued to be P-ANCA positive, MPO-ANCA positive, and PR3-ANCA negative. (Please refer to Table 2 for details).
 Figure 2: CT scan thorax. View Figure 2
Figure 2: CT scan thorax. View Figure 2
Table 2: Lab work from subsequent encounter 2016. View Table 2
A kidney biopsy was done with preliminary report suggestive of type II/IV lupus nephritis/mixed connective tissue disease and final report showing focal segmental necrotizing and sclerosing glomerulitis (Figure 3).
 Figure 3: Renal biopsy electron microscopy, mid-magnification. View Figure 3
Figure 3: Renal biopsy electron microscopy, mid-magnification. View Figure 3
Patient was diagnosed with AAV and discharged two weeks later with oral steroids, scheduled rituximab infusions, and instructions to follow up with Rheumatology and Nephrology specialists. She was seen shortly after as an outpatient, and her rituximab infusions were discontinued and switched to cyclophosphamide due to worsening renal function.
A few months later, patient was readmitted to the hospital for violaceous rash with bullae and blebs that became ulcers onface, upper and lower extremities bilaterally (Figure 4). Initial impression was a drug reaction versus infectious disease. Methylprednisolone was started, along with daptomycin and piperacillin/tazobactam, and a skin biopsy was performed which showed minimal inflammation with no signs of leukocytic vasculitis. Vasculitis workup including CPK, aldolase, dsDNA, C3, C4, ANCA and Beta-2 glycoprotein came back negative and wound culture grew coagulase negative S. aureus. Antibiotic therapy was deescalated to daptomycin, an eschar was beginning to form and patient was discharged three weeks later, on prednisone, amoxicillin/clavulanate, doxycycline, and cyclophosphamide. Patient was seen as an outpatient one week after discharge where she stated she was doing her own wound care and having no complications on prednisone and cyclophosphamide, wounds were still healing.
 Figure 4: Skin lesions on subsequent presentation in 2016, demonstrating violaceous rash with bullae, blebs and ulceration. View Figure 4
Figure 4: Skin lesions on subsequent presentation in 2016, demonstrating violaceous rash with bullae, blebs and ulceration. View Figure 4
Three months later patient was readmitted because the ulcers in her upper and lower extremities became necrotic complicated with foul smell and wound myiasis after she discontinued wound care (Figure 5). Physical examination on admission showed full-thickness loss with granulating and necrotic tissue, accompanied by visible larvae in upper and lower extremities bilaterally. Antibiotic therapy was started with daptomycin and cefazolin; and surgical debridement was performed. Due to the extent of the debridement she was transferred to an institution with a burn unit for higher level of care and skin grafting was performed.
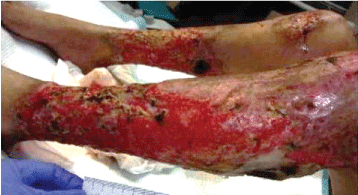 Figure 5: Infected skin lesions on readmission in 2016, with necrosis and myiasis. View Figure 5
Figure 5: Infected skin lesions on readmission in 2016, with necrosis and myiasis. View Figure 5
She was seen again three months later as an outpatient after skin grafts were done, wounds were healing well, no amputations were performed, renal function was stable and currently patient is only receiving prednisone with no evident complications.
Levamisole is an anti-helmintic, historically used in humans to treat cancer and rheumatologic diseases, given its immunomodulating properties. It has been banned by the Food and Drug Administration (FDA) for use in humans since 2000, due to its side effects of agranulocytosis and vasculitis. However, it continues to be available for veterinary use and is a known adulterant for cocaine, as it adds bulk and weight, potentially intensifying cocaine’s psychotropic effects [3].
Levamisole induced AAV is a relatively new entity with cases first being documented as early as 2009; its clinical presentation being a tender, purpuric rash in extremities, trunk, digits and nasal tip [4]. Considering that in 2016, 93% of the cocaine seized by the US Drug Enforcement Agency was cut with Levamisole and/or levamisole mixtures [5], and that the patient’s initial clinical presentation was similar to other cases described in literature; in retrospect, levamisole induced AAV is the likely culprit for this patient’s initial symptoms in 2010.
The presence of Levamisole cannot be recognized by a simple UDS. Exposure to the substance can be confirmed with Gas Chromatography-Mass Spectrometry (GC-MS) in urine [6] but this is not a readily available test in most hospitals. It may be requested in patients who present with similar rash, positive ANCA and history of cocaine abuse. However, according to some authors the identification of the substance may not be necessary to make the diagnosis [3].
The natural history of the cutaneous manifestations of levamisole induced AAV is spontaneous resolution after the substance is withdrawn; though immunologic markers may persist for up to 14 months [7]. In cocaine-induced systemic vasculitis, patients may be positive for PR3-ANCA, MPO-ANCA and/or the combination PR3-ANCA and MPO-ANCA may be found [8].
To our knowledge there are no defined criteria for the identification of levamisole induced AAV and its diagnosis remains based on exclusion. In recent years, some authors have proposed that once diagnosis is suspected, the staging of this condition be based on the clinical cutaneous findings, as such: The mild stage (I) consists of livedo with mild purpura and no areas of necrosis. The moderate stage (II) has fixed purpuric patches and or plaques, without significant skin ulceration and the severe stage (III) is characterized by extensi vereticular (IIIa) or confluent (IIIb) purpura accompanied bybullous involvement with or without cutaneous necrosis [9].
Although there are currently no definitive guidelines on the treatment of levamisole induced vasculitis, besides avoiding further exposure to the substance, the management is essentially supportive [1]. To our knowledge, four cases of severe levamisole-induced AAV have been described with extensive full-thickness skin necrosis, involving 15, 52, 49 and ~20% of TBSA [10,11]. On these patients, invasive therapy was performed including surgical debridement, skin grafting and amputations, with variable outcomes. A recently published article demonstrated that early excision and grafting of large surface levamisole-induced skin necrosisin dependent of autoantibodies results in successful outcomes and limits wound morbidity [10]. Our patient’s TBSA involvement was estimated at 13% and she eventually underwent extensive skin grafting and was managed in a burn unit, with good end results.
Renal biopsy findings were also remarkable since it reported a lupus-like nephritis combining membranous and focal proliferative/sclerosing glomerulitis which to our knowledge has not been reported in cases of Levamisole-induced AAV, even though there have been reports of two cases of membranous nephropathy with crescents [12,13].
Our patient’s course was complicated because she was lost to follow up for six years and remained untreated, presumably still using cocaine, as inferred from her subsequent positive UDS. She was seen again with a wide constellation of problems and multi-system involvement including diffuse alveolar hemorrhage and focal segmental necrotizing and sclerosing glomerulitis diagnosed by kidney biopsy. It is well known that patients with AAV have an increased mortality at one year if left untreated but the patient survived all these years without any therapeutic regimen [14].
The presence of a characteristic clinical picture and the presumed widespread use of levamisole as an adulterant for cocaine should raise suspicions for levamisole induced AAV whenever patients are seen with purpuric rashes, history of cocaine abuse and positive ANCA.
Remission of the disease occurs spontaneously after stopping the use of cocaine and patients should be strongly encouraged to abstain from it, since prolonged use may further cause systemic damage.