Auto antibodies, Auto immune myopathy, Myositis, HMGCR
Recent studies have reported an increased incidence of cancer in seronegative necrotizing autoimmune myopathy (NAM) [1] and anti-3-hydroxy-3-methylglutaryl-CoA reductase (HMGCR) NAM [1,2]. Herein, we report the case of a patient with a NAM associated with cancer in which myositis relapse led to the identification of cancer relapse.
In 2004, a 57-years-old female received rosuvastatin for secondary prevention of stroke. In 2010, an asymptomatic CK elevation around 900 IU/L is noticed on a routine workup and persisted despite statin discontinuation. A rise in CK level is progressively observed (up to 2,400 IU/L) and the patient referred in Neurology for consultation in March 2011. Proximal (deltoid: 4/5, psoas: 4/5) and axial (neck flexors: 4/5) muscle weakness was notedwith CK level around 2,000 IU/L. The patient had only low-titer antinuclear antibodies (ANAs: 1/80), both negative extractable nuclear antigen antibodies (anti-ENA) and dot myositis profile (including anti-Jo1, -PL7, -PL12, -Pm/Scl, -Scl70, -Ku, -SRP, -Mi-2 (immunodot, PMS7D, Blue DOT, D-tek)) and EMG was consistent with myositis [3]. A muscle biopsy performed in May 2011 showed only isolated and non-specific muscle fiber size variation. Neither muscle necrosis, muscle regeneration, MHC-I antigen, C5b9 complement deposition nor inflammatory infiltrate was reported. In September 2011, the patient is evaluatedin Internal Medicine and the possibility of a statin-induced autoimmune myopathy is suspected, though the anti-HMGCR autoantibodies (aAbs) testing was not available at the time. The aAbs were finally found positive in August 2012 (ALBIA-HMGCR). When reevaluated in October 2012, the manual strength evaluation deteriorated (psoas: 3/5, neck flexors: 3/5) and CK level was 3,300 IU/L. In December 2012, the patient received high-dose oral corticosteroids (1 mg/kg/day) and methotrexate (20 mg/week) for three months, but subsequently 6 plasma exchanges were performed followed by the introduction of intravenous immune globulin (IVIg), because of deteriorating muscle function. Improvement permitted slowly-progressive steroid tapering over the following months to low-dose prednisone (10 mg/day) with methotrexate and the patient recovered normal strength. In March 2014, new-onset dysphagia led to distal sub occlusive esophageal stenosis discovery and epidermoid carcinoma T3NxM0 diagnosis. During the same period of time a rise in serum CK (900 IU/L) accompanied by muscle strength worsening (deltoid: 4/5, psoas: 4/5) was observed. Chemotherapy was rapidly initiated followed by concomitant radiotherapy and, in this context, MTX replaced by IVIg. By the end of 2014, she was in complete remission of both cancer and myositis without treatment (deltoid: 5/5, psoas: 4/5, CK: 35 IU/L). Fifteen months later, in March 2016, the patient complained of exercise intolerance and shortening of her walking distance. Her manual strength examination declined (deltoid: 4/5, psoas: 4/5) and a rise in CK 1200 IU/L occurred suggesting myositis relapse. On cancer screening reevaluation, the progression of a pulmonary nodule (15 mm) was found with hypermetabolism on PET-scan (SUV max: 6.5) suggesting metastasis from esophageal cancer relapse. The Oncology Board decided to treat the tumor with stereotaxic radiosurgery. The patient received concomitant IVIg monotherapy [4] in June 2016 for treatment of myositis.
Clinical and epidemiological data have confirmed the temporal relationship between the onset of myositis and malignancy [5], and cancer-associated myositis has been defined as a cancer occurring within 3 years of diagnosing myositis [6] (other studies report an excess risk of cancer up to 5 years). A strong correlation between the clinical course of myositis and malignancy has been reported in other studies [7], as relapse of the cancer is often accompanied by reappearance of myositis, and the successful treatment of the malignancy helps improve myositis symptoms. Myositis-specific autoantigens are expressed in several cancers known to be associated with myositis and these autoantigens are expressed at higher levels in regenerating muscle cells [8]. It has been suggested that mutated forms of self-antigens may arise in cancer cells, triggering an autoimmune response against wild-type forms of the proteins [9] expressed in other tissues such as muscle. The reappearance of myositis at the time of cancer diagnosis, the treatment-free remission of myositis following cancer eradication and the detection of cancer relapse following myositis relapse in our patient (Figure 1) supports the proposition of a pathophysiological link between the two events, and highlights the importance of aggressive cancer workup when facing an anti-HMGCR NAM.
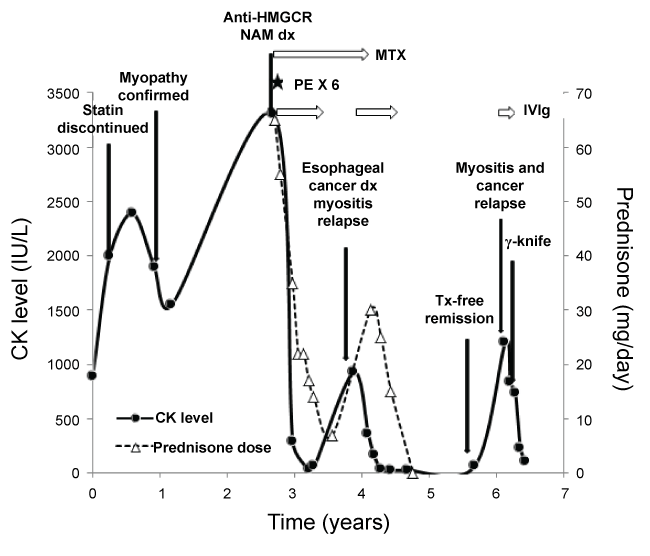 Figure 1: CK and prednisone evolution over in a patient with anti-HMGCR NAM associated with cancer.
CK: Creatine Kinase; HMGCR: 3-Hydroxy-3-Methylglutaryl-CoA Reductase; NAM: Necrotizing Autoimmune Myopathy; MTX: Methotrexate; PE: Plasma Exchange; IVIg: Intravenous Immune Globulin; dx: Diagnosis; Tx: Treatment; γ: Gamma. View Figure 1
Figure 1: CK and prednisone evolution over in a patient with anti-HMGCR NAM associated with cancer.
CK: Creatine Kinase; HMGCR: 3-Hydroxy-3-Methylglutaryl-CoA Reductase; NAM: Necrotizing Autoimmune Myopathy; MTX: Methotrexate; PE: Plasma Exchange; IVIg: Intravenous Immune Globulin; dx: Diagnosis; Tx: Treatment; γ: Gamma. View Figure 1
No specific operating grant was received to carry out the work described in this manuscript. Dr. Landon-Cardinal is the recipient of Clinical Fellowship awards from the Université de Montréal Rheumatology Program - AbbVie Educational Grant and the Association des médecins rhumatologues du Québec - Visithan-Khy Educational Grant.
The authors have declared no conflicts of interest.