Sex disparity in anterior cruciate ligament injury is multifactorial. Sex hormones, such as relaxin, may play a role in the increased risk of injury in female athletes. In order to fully investigate this relationship, optimal strategies for capturing serum relaxin concentration across the menstrual cycle must be determined. The aim of this study was to describe the variability in the timing and magnitude of relaxin concentration changes across the menstrual cycle.
Serum samples from 26 recreationally active women from one menstrual cycle (6 days of menses and 8-10 days of the luteal phase) were analyzed with enzyme linked immunosorbent assay. Line plots were used to analyze the relationship between the relaxin or progesterone peak concentration and day of the menstrual cycle, day of the luteal phase, and the start of menstruation. A repeated measures ANOVA at the 95% confidence interval was used for this data. Z-scores of the average daily means were calculated in order to compare the variability between progesterone and relaxin concentrations on the same scale [1].
The mean peak relaxin concentration was 49.6 ± 30.5 pg/mL. The peak values occurred later than expected with 65% of subjects demonstrating peaks on luteal days 9 or 10 and a mean of 3.03 ± 1.66 days after peak progesterone concentration.
Like other sex hormones, there is significant inter-subject variability in the magnitude and timing of peak relaxin concentration across the menstrual cycle. Future investigation of the role of relaxin in anterior cruciate ligament injury should employ serial sampling, particularly throughout days 8-12 of the luteal phase to increase the likelihood of accurately capturing the peak relaxin concentration.
Relaxin, Anterior cruciate ligament, Injury, Female athlete
ACL: Anterior Cruciate Ligament; MMPs: Matrix Metalloproteinases; BMI: Body Mass Index; M1: Menstrual Cycle; L1: Luteal Cycle
The increased risk of anterior cruciate ligament (ACL) injuries in female athletes is a multifaceted problem partly governed by sex differences in anatomy, neuromuscular control and sex hormones which have been linked to compromised knee stability [2]. As ACL injuries are more likely to occur in the peri-ovulatory phase (while estrogen is rising and progesterone is stable) than in the post-ovulatory phase (when progesterone reaches peak concentration) [3], estrogen has been the primary hormone of interest. However, the potential role of relaxin as a hormonal contributor to ACL injury has gained credibility since this "pregnancy hormone" was found to fluctuate in non-pregnant women across the menstrual cycle [4,5], relaxin receptors were identified on the human ACL [6], and relaxin was found to be associated with ACL injury risk [7].
Relaxin is a member of the insulin-like superfamily and is primarily recognized for facilitating vaginal delivery by increasing pelvic ligament laxity [8]. There are three known relaxin peptides in humans, each with varying functions [8]. Whereas relaxin-1 and relaxin-2 regulate collagen expression, metabolism in fibroblasts, and changes in proteolytic enzymes in the corpus luteum, decidua, and endometrium, relaxin-3 is specific to the brain [9]. Relaxin-2 is also present in non-pregnant women [10] and has been linked to changes in connective tissue resulting in altered mechanical properties of ligaments, tendons, muscle and cartilage [5,11]. How relaxin alters connective tissue properties is not fully understood, but proposed mechanisms, include:
1. Relaxin facilitates collagen degradation by initiating a ligand-gated cascade through G-protein coupled receptors leading to changes in the concentration of collagen remodeling mediators such as matrix metalloproteinases (MMPs), plasminogen activator, gelatinases, stromelysins, collagenases, alpha smooth muscle actin and tumor growth factor beta [5,11].
2. Relaxin reduces collagen production by decreasing gene expression for collagen I and III [8].
3. Relaxin decreases inter-collagen fibril interactions leading to increased collagen fibril sliding and ligament length, also known as the "Sliding Fibril Hypothesis" [12].
As the mechanism of relaxin continues to be explored, a collection of animal and human studies have demonstrated an important relationship between relaxin and ACL function. In a guinea pig model, relaxin and relaxin + estradiol exposure led to weaker ACLs with a reduced load-to-failure [13]. In a follow-up study of human ACL cells, relaxin exposure resulted in increased expression of matrix metalloproteinases 1 and 3 (mediators of collagen degradation) and decreased expression of collagen types I and III [14]. Of interest, this intracellular upregulation only occurred in female-derived ACL cells. Finally, Dragoo, et al. conducted a prospective study of female collegiate athletes and found that a serum relaxin concentration of 6.0 pg/mL had more than a four-time greater risk of ACL injury [7].
Dragoo's findings suggest that high serum relaxin concentration is an important, and possibly modifiable, risk factor for ACL injury in female athletes. However, further investigation is warranted as only 46% of the athletes in the Dragoo, et al. study had detectable levels of serum relaxin. It is possible that there are some athletes who do not produce detectable levels of relaxin, but it is also possible that the limited sampling frequency (only two samples on post-ovulatory days 6 and 8) during one menstrual cycle may have missed the window for detectable concentrations of serum relaxin in some women. In support of the latter, two prior studies showed that relaxin peaks later in the menstrual cycle in the majority of women (days 10-12). However, these studies were limited by small sample size and the physical activity status of these women is unknown [15,16]. Therefore, important next steps include identifying the optimal sampling strategy for accurate detection of peak relaxin levels in young, female athletes. Further, an understanding of the inter-individual variability of relaxin would inform study design to determine the impact of relaxin on musculoskeletal function and injury.
The objective of this study was to characterize the inter-individual variability in the timing and magnitude of relaxin concentration changes across the menstrual cycle, and determine the optimal time for sampling peak relaxin concentrations. We hypothesized that 1) the timing and magnitude of peak relaxin concentration fluctuations will vary widely between eumenorrheic, active females; 2) through serial measurements, we can determine the optimal window to measure peak serum relaxin concentration during the menstrual cycle so as to inform future research in this area.
The subjects included in this study came from a larger study examining sex hormone profiles in menstruating females [1,17]. All participants were informed of the study procedures and signed a consent form approved by the university's Institutional Review Board for the Protection of Human Subjects. As part of this larger prospective study, 60 women were tracked for two menstrual cycles, but serum from only one cycle was analyzed for relaxin concentrations. All subjects met the following inclusion criteria: 1) BMI < 30; 2) Non-smokers; 3) Recreationally active 2.5 to 10 hrs/week for the past 3 months; 4) History of normal menstrual cycles lasting 26-32 days in the past 6 months that vary in length no more than + 1 day; 5) No use of oral contraceptives or other hormone stimulating medications in the past 6 months, and 6) No history of pregnancy or plans to become pregnant during the course of the study. Thirty-four (57%) of the 60 women were excluded: 6 had anovulatory cycle (defined as luteal progesterone < 3 ng/mL) [17], 7 had no clear rise in either progesterone or relaxin, 7 had no clear peak for either relaxin or progesterone, 13 did not reach peak relaxin values during the sampling window and one subject was missing data (Figure 1). The remaining 26 subjects were the final study sample for the current investigation.
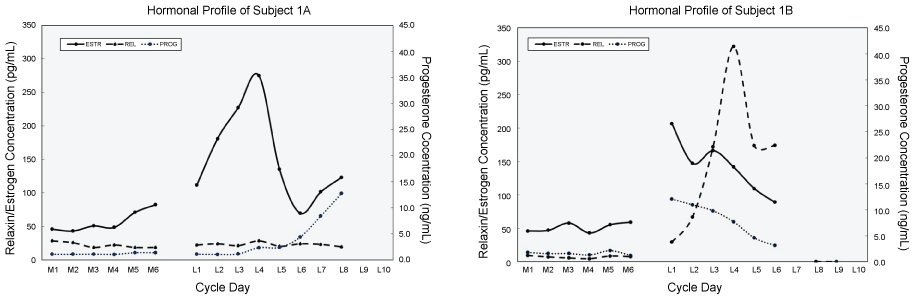 Figure 1: Examples of inaccurate detection of ovulation. a) The luteal phase testing was begun too early in this subject. It is most likely that true ovulation occurred around L4 or L5 given the large peak in estradiol and that the progesterone just begins to rise at L8 (L9 and L10 data was not collected); b) The luteal phase testing was begun too late in this subject. We missed the progesterone peak, but clearly observed the relaxin peak.
View Figure 1
Figure 1: Examples of inaccurate detection of ovulation. a) The luteal phase testing was begun too early in this subject. It is most likely that true ovulation occurred around L4 or L5 given the large peak in estradiol and that the progesterone just begins to rise at L8 (L9 and L10 data was not collected); b) The luteal phase testing was begun too late in this subject. We missed the progesterone peak, but clearly observed the relaxin peak.
View Figure 1
The first day of the menstrual cycle (M1) was defined as the first day of bleeding. Menstrual phase testing was conducted on the first day of the menstrual cycle through the subsequent five days (M1 through M6). The day of ovulation was determined with a commercially available ovulation kit (CVS One Step Ovulation Predictor [sensitivity 20 mIU/ml LH, accuracy 99%]; CVS Corporation, Woonsocket, RI). Luteal phase testing commenced with the first day following the positive test post-menses (L1) and continued for ten days (L2-10). The cycle length was determined by tracking the onset of the next menses. Testing consisted of daily venipuncture during M1-6 and L1-10. Blood (10 cc total) was obtained between 7-9 am and serum was stored at -80 ℃.
Serum samples were analyzed in duplicate for relaxin-2 using Quantikine Human relaxin-2 Immunoassay (DRL200 R&D Systems Inc, Minneapolis, MN). Using the relaxin standards supplied with the kit, two additional standards (3.9 pg/ml and 10.4 pg/ml) were added to the manufacturer recommended curve to maximize separation at low concentrations (3.9 pg/mL and 10.4 pg/mL).
All biochemical analyses were completed by the same trained technicians. The assay limit of blank (LOB = 0.739 pg/mL) and limit of detection (LOD = 2.591 pg/mL) was calculated according to previous recommendations for verification of LOD [18]. The intra- and inter-plate coefficients of variation for the assay were 8.21% ± 13.21 and 18.22% ± 3.17, respectively. The progesterone peak was defined as the highest concentration during M1-6 and L1-10, as confirmed by at least one lower value following the peak value. The relaxin peak was determined in the same fashion as the progesterone peak. Additionally, if the highest relaxin value occurred on L10 and the next menses occurred within the next 1-3 days, the L10 value was accepted as the peak relaxin. This exception was based on prior research [16,19] reporting serial samples and observation of our own data which shows that relaxin starts to decrease no less than two days before the next menses and returns to nadir levels by M1.
Standard summary statistics such as means and standard deviations for continuous variables (age, BMI, luteal length, cycle length) were calculated for the study sample. Line plots were developed to illustrate the relationship between the relaxin or progesterone peak concentration and day of the menstrual cycle, day of the luteal phase and the onset of the next menses. An ANOVA in repeated measures was used to generate means and 95% confidence intervals of the mean (standard error) for the line plots. The mean, standard deviation and range of days between the peak progesterone and relaxin as well as the median difference between peak relaxin concentrations and concentrations on any given luteal phase day were calculated. Finally, to compare the variability between progesterone and relaxin concentrations on the same scale, we plotted the z-scores for each of the daily means [1]. Analyses were performed using SAS statistical software (version 9.4, SAS Institute, Cary NC) and R statistical software (R Development Core Team, version 3.3.2, 2014).
The 26 women included in this study were similar to the larger study population with regard to age (Table 1). Definite peak concentrations of relaxin and progesterone were observed in 15 women. The remaining 11 women demonstrated the highest relaxin concentration on L10 and began their next menstrual cycle < 3 days, so this was also considered the peak. Peak relaxin values ranged from 15-118 pg/mL (mean 49.6 pg/mL), and the magnitude of change from follicular levels varied from 12.5-113.8 pg/mL. All peak relaxin values were captured between L3 to L10 and 65% of subjects demonstrated their highest concentrations on days L9 and L10 (Figure 2). Further analysis showed the median difference between peak relaxin concentrations and concentrations on any given luteal phase day progressively decreased throughout the luteal phase and was lowest at L10 (Figure 3). The median magnitude of difference on L10 was 0.45 pg/ml, compared to 25.1 pg/ml and 17.83 pg/ml on L6 and L8 respectively. Peak relaxin was delayed relative to the progesterone peak with 14/26 (54%) of progesterone peaks occurring on L6 or L7 and 17/26 subjects (65%) of relaxin peaks occurring on L9 or L10. At the individual level, the mean delay between peak progesterone and peak relaxin was 3.03 ± 1.66 days (range 0-6 days). When characterizing peak relaxin based on cycle day (day from M1) and days before the next menses (days minus M1 of next cycle), we did not observe a strong relationship with a specific day. The majority of subjects had peak relaxin concentration between cycle days 21-25, which corresponded to 4-6 days before the start of the next menstrual cycle (Figure 4a and Figure 4b). There is significant variability of both progesterone and relaxin across the menstrual cycle (Table 2). Compared to progesterone, relaxin peaked later and demonstrated greater variability throughout the majority of the menstrual cycle (Figure 5).
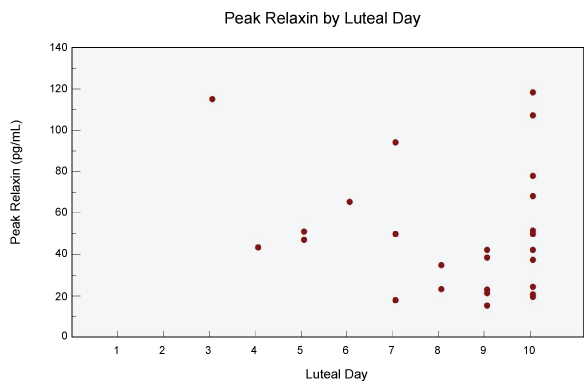 Figure 2: Timing of relaxin peak in the luteal phase.
View Figure 2
Figure 2: Timing of relaxin peak in the luteal phase.
View Figure 2
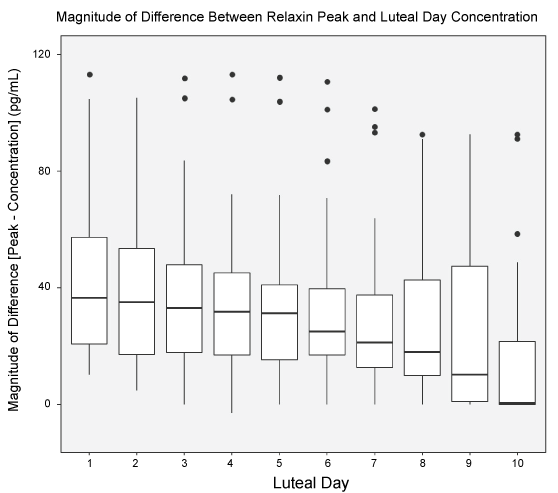 Figure 3: The magnitude of difference between peak and relaxin concentrations for each luteal phase day. This figure demonstrates the smallest difference is achieved on L10, progressively decreasing throughout the luteal phase.
View Figure 3
Figure 3: The magnitude of difference between peak and relaxin concentrations for each luteal phase day. This figure demonstrates the smallest difference is achieved on L10, progressively decreasing throughout the luteal phase.
View Figure 3
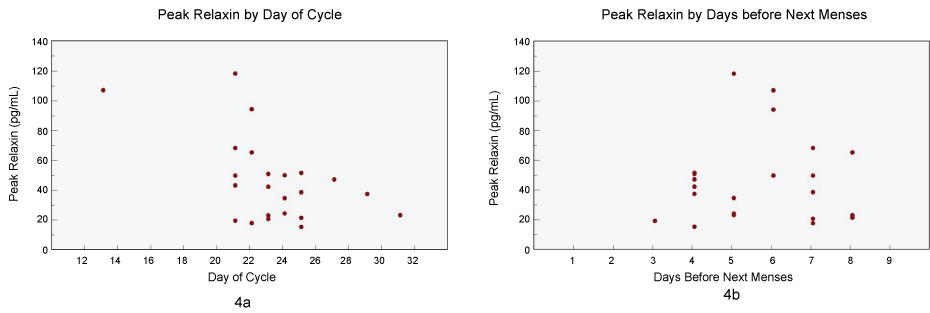 Figure 4: a) Timing of relaxin peak across the menstrual cycle; b) Timing of relaxin peak with the onset of the next menses.
View Figure 4
Figure 4: a) Timing of relaxin peak across the menstrual cycle; b) Timing of relaxin peak with the onset of the next menses.
View Figure 4
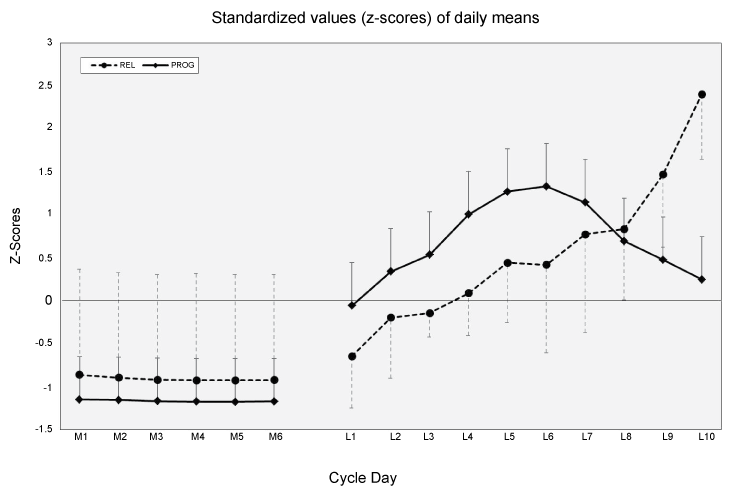 Figure 5: Magnitude of difference between peak relaxin concentration and relaxin concentrations for each day of the menstrual cycle.
View Figure 5
Figure 5: Magnitude of difference between peak relaxin concentration and relaxin concentrations for each day of the menstrual cycle.
View Figure 5
Table 1: Subject demographics and menstrual cycle characteristics. View Table 1
Table 2: Means, standard deviations (SD) and rages for delta scores of relaxin and progesterone. View Table 2
In order to determine the relationship between sex hormones and ACL injury and subsequently screen for those at risk, we need to be able to accurately capture serum concentrations across the menstrual cycle in a feasible manner. To our knowledge, this is the first study to utilize serial serum samples to comprehensively characterize relaxin concentration fluctuations in recreationally active, eumenorrheic women. Our primary hypothesis was that consistent with the variability in sex steroid hormone profiles [17] and with the priming of relaxin by the rise in estrogen at ovulation [11,20], there would be considerable inter-subject variability of relaxin concentration. In regards to the magnitude of relaxin fluctuations across the menstrual cycle, this hypothesis was largely upheld. Peak relaxin values observed in our study were similar to work by Stewart, et al. [16], which showed values ranging from 30-150 pg/mL (mean of 50 pg/mL), and Johnson, et al. [15], which showed values ranging from 20-103 pg/mL (mean of 37 pg/mL). It is reassuring that our magnitudes of relaxin concentration fluctuations were similar across three studies despite the fact that our cohort was younger and likely more physically active than the women studied previously, and that there were some differences in the assays and technique employed across studies. We also compared the variability of the magnitude of relative change in concentration between progesterone and relaxin. The z-scores allow us to compare the daily fluctuations around their means of both hormones on the same graph. From these data it appears that the inter-individual variability in relaxin is generally greater than that of progesterone on most days measured (with the possible exception of L3 and L4), but the overall magnitude of values fluctuate less around the mean in relaxin than progesterone until days L9 and L10 when relaxin rises rapidly toward its peak values.
Our hypothesis that there would be variability in the timing of peak relaxin concentration was also upheld. Prior studies reference the mid-luteal phase to be the window to capture the relaxin peak [20-22]. However, work by Stewart, et al. [16] and Johnson, et al. [15] demonstrate a rise in relaxin around days L6-8 with peak values occurring around L10 to 12. Our data, using more current assays with greater sensitivity at the lower end of the curve, support this later-than-expected relaxin peak by demonstrating a slow rise in relaxin a few days after ovulation, a steep rise around days L6-9, and peak values typically occurring around days L9-10. Both progesterone and relaxin are secreted by the corpus luteum and some prior work suggests that relaxin and progesterone peak around the same time in the mid-to-late luteal phase [21]. However, Stewart, et al. found the relaxin secretion pattern was delayed 6-9 days after progesterone [16]. Our data supports a delayed relaxin peak relative to progesterone as the mean delay between peak progesterone and peak relaxin was 3.03 ± 1.66 days (range 0-6 days), demonstrating that relaxin does not peak until the latter portion of the luteal phase. The rise in serum relaxin concentrations at the end of the luteal phase may be, at least partly, due to relaxin leakage from apoptotic luteal cells [16]. This later peak in relaxin concentration was further demonstrated by the magnitude of difference observed between concentrations on most luteal days and the eventual peak concentrations. Of all of the variables we analyzed, it appears that the most feasible way to predict the timing of the peak relaxin concentration is by the postovulatory day. The postovulatory day is determined by measuring urinary luteinizing hormone (LH) and designating the day of a positive test as the day of ovulation. While this is the most practical way to predict ovulation in this type of study, the variability in the LH surge duration, which can be characterized by multiple peaks, can make detection challenging [23]. It is likely that we incorrectly identified ovulation in at least 13 of our subjects who had to be excluded because they had an ovulatory cycle (+ urine LH surge) with a confirmed progesterone peak, but the relaxin peak was never observed (Figure 1). This resulted in starting the luteal testing too early or too late to capture the peak relaxin concentration.
Our findings of a later-than-expected relaxin peak and significant inter-subject variability in relaxin concentration fluctuations provide important context when interpreting prior studies investigating the link between relaxin and musculoskeletal injury. For example, in the previously mentioned prospective study of female collegiate athletes conducted by Dragoo, et al. subjects with serum relaxin concentration > 6.0 pg/mL had more than a 4-fold increased risk of ACL injury during their collegiate athletic career [7]. In this study, subjects underwent venipuncture on day L6 and L8 (as determined by similar home ovulation kits used in our study). The peak relaxin concentration in this study (5.30 ± 9.08) was lower than the relaxin concentrations displayed by our subjects (46.11 ± 28.72). While it is possible that the peak relaxin concentration was lower in Dragoo's cohort due to their increased levels of physical activity as collegiate level athletes, it is more likely that the disparity is related to differences in sampling technique. In our data, sampling on days L6 and L8 did not correlate well with the peak relaxin concentration and the magnitude of relaxin was significantly lower than if sampling had occurred on days L9 and L10 [(17.7 ± 15.34) vs. (29.2 ± 23.83)]. Since the collegiate level athletes in Dragoo's study are more likely to have menstrual dysfunction than the recreationally active women in our study [24], the variability of the relaxin peak in high-level athletes might be even greater than what we observed. Also unknown is how the timing of these peaks relates to changes in ligamentous laxity and injury risk across the menstrual cycle. While relaxin rises and peaks in the late luteal phase, which is consistent with one of the time points when knee laxity is elevated, epidemiological studies of ACL injury report that a higher proportion of ACL injuries occur in the follicular (pre-ovulatory) as opposed to luteal (postovulatory) phase, with some reporting highest risk near the time of menses while others report highest risk in the peri-ovulatory days [3]. Work by Shultz, et al. [25] demonstrated that changes in estradiol, progesterone and testosterone modulated knee laxity across the menstrual cycle with a 3 to 5 day lag time effect. If a similar delay in alterations of tissue mechanical properties occurs with relaxin fluctuations, then the peak relaxin in the late luteal phase might be the link between the changes in ligament laxity, motor control strategies and increased risk of ACL injury observed in the pre-ovulatory phase [2]. Furthermore, since estradiol appears to enhance the action of relaxin [26], the relationship of these two hormones and knee stability should be investigated.
Our findings are limited by use of a home ovulation kit to predict ovulation, stopping data collection on L10 and only including serial measurements across one menstrual cycle. Future work should build on this study by addressing these limitations to more completely characterize how relaxin fluctuates at the end of one cycle and into the next cycle. Serial samples across multiple cycles will also enable us to determine the month-to-month consistency of relaxin within and between women. If daily testing is prohibitive, then daily testing on days L8-12 would increase the likelihood of capturing the true peak values. Sequential testing would also allow future investigation of which relaxin variable is most important when determining the ultimate threshold for ACL injury: Peak concentration, total relaxin exposure or the magnitude of change from the follicular to the luteal phase. Once relaxin can accurately be measured and characterized, we can begin to link relaxin to changes in connective tissue properties and injury risk so that we can better understand the mechanistic links between hormonal changes and risk of ACL rupture. If relaxin is important in sex disparities in ACL (and other) injuries, we can use this information to risk-stratify athletes and potentially modulate serum relaxin concentration, with hormonal contraceptives, for example [4].
Our purpose was to determine the timing and magnitude of relaxin concentration changes across the menstrual cycle in recreationally active, eumenorrheic women. Our study demonstrates that relaxin peaks on average 3 days later than progesterone with 65% of peak values occurring on L9 and L10 in our population. Based on the variability in the timing and magnitude of relaxin peaks, we recommend serial sampling from postovulatory days L8-12 to increase the likelihood of obtaining the highest concentration.
E. Casey - Drafting and critical revising of the manuscript; substantial contribution to the analysis and interpretation of data.
T. Anderson - Critical revising of the manuscript; substantial contribution to the analysis and interpretation of data.
L. Wideman - Substantial contribution to the data acquisition; critical revising of the manuscript; substantial contribution to the analysis and interpretation of data.
F. Shofer - Substantial contribution to the analysis and interpretation of data; critical manuscript revision.
S.J. Shultz - Substantial contribution to the research design and data acquisition; critical revising of the manuscript; substantial contribution to the analysis and interpretation of data.
The project described was supported by grant no. R01-AR53172 NIH-NIAMS and through a cooperative agreement (NICHD/NIH U54 HD28934) as part of the Specialized Cooperative Centers Program in Reproductive Research.
This investigation was support by an internal institutional grant. The authors have no conflict of interest to declare. The results of the study are presented clearly, honestly, and without fabrication, falsification, or inappropriate data manipulation. The results of the present study do not constitute endorsement by the American College of Sports Medicine.