Models for predicting the success of labor induction can be used to improve clinical decision. The aim of this study was to create nomogram based on clinical, sonographic and biochemical parameters, to provide an individualized prediction of success of induction.
A prospective cohort study was conducted on 201 consecutive pregnant women who underwent induction of labor. This study includes singleton, cephalic, term (≥ 37 gestational weeks) and intact amniotic membranes pregnancies. Clinical characteristics, fetal fibronectin, transvaginal ultrasound and Bishop score were used to construct a nomogram for predicting induction success. Independent risk factors were identified by logistic regression.
Vaginal delivery within 24 hours occurred in 141 (70.1%) women. Upon multivariate analysis, parity and Bishop score were significantly associated with vaginal delivery within 24 hours. The nomogram constructed had a concordance index of 0.78 (area under the curve). Calibration found no difference between the predicted probabilities and the observed results.
We developed nomogram to predict the success of induction labor. Discrimination and calibration of model were statically good. External validation of this nomogram in different groups of patients is needed before recommending their use in clinical practice.
Induction of labor, Parity, Fetal fibronectin, Bishop score, Ultrasound
Induction of labor is one of the most frequent procedures in obstetrics, representing 23.3% of all deliveries [1]. Induction is indicated when either maternal and/or fetal benefit to ending the pregnancy are supposed. However, it is not a complication-free procedure and is usually associated with an increase in caesarean section rates (20%) [2]. Evaluation of the probabilities of success is one of the most relevant aspects in clinical practice, as it would permit identifying patients with a high risk of induction failure who would hypothetically benefit from the termination of pregnancy by caesarean section.
Several clinical factors have been related to induction success: Maternal age, maternal size, parity, weight, height, BMI (body mass index), weight of the new-born or gestational age [3,4]. Nowadays, the standard method for predicting induction success is the assessment of cervical conditions using the Bishop score [5]. However, this evaluation is subjective and has shown a poor predictive value of induction success in different studies [6]. Two other methods that have been analyzed for assessing induction success were transvaginal ultrasound and fetal fibronectin (fFN). Ultrasound is an objective and reproducible technique that permits evaluating the cervical length and the characteristics of the internal cervical os. The results obtained regarding the usefulness of ultrasound for predicting the success of induction are contradictory [7]. fFN has been related to spontaneous onset of labor and induction success in different studies [8,9].
The development of risk prediction models is becoming more important in the medical literature [10]. These models allow the stratification the risk of caesarean section delivery before the start of the procedure, using risk factors with statistical significance, which enables clinicians to offer proper advice to patients.
The objective of this study is to develop prediction models (nomogram) using clinical, sonographic and biochemical parameters to predict outcome of induction of labor and it could be useful for individualized counseling.
The STROBE guidelines were followed for this prospective observational cohort study of 201 women, which was developed from September 2014 to September 2015 at Puerta de Hierro Majadahonda University Hospital, Spain.
Patients were admitted consecutively for induction of labor. Eligibility criteria were: gestational age ≥ 37 weeks, maternal age ≥ 18 years, singleton pregnancy, live fetus, cephalic presentation, intact amniotic membranes and no contraindication of vaginal delivery. The exclusion criteria were: Fetal malformations, vaginal bleeding, previous uterine surgery (myomectomy or caesarean section), fetal compromise, beginning of the active phase of labor and digital exploration 24 hours prior to induction. Ultrasound and examination were performed by only one expert in order to reduce inter-observer variability.
The following data were obtained from electronic medical record for each woman who was undergoing induction of labor: Maternal age, parity, BMI, previous abortion, gestational age, fFN, cervical length, presence or absence of funneling and Bishop score.
We assessed fFN in cervical secretions using the qualitative fast-reacting immunoassay test, QuikCheck® fFN test (Adeza Biomedical Corporation, Sunnyvale, California, USA) with a cut-off value set at > 50 ng/mL. The sample was obtained using a sterile polyester swab from posterior vaginal fornix before digital and sonograhpic examination. A positive result produced two visible lines (control and test) after 10 minutes.
A cervical transvaginal ultrasound examination was then performed using a HD11XE ultrasound machine (Philips Medical System Netherlands) with a 6.0 MHz vaginal transducer. We utilized the technique described by Andersen [11]. The ultrasound examination lasted 3 minutes. The probe was placed in the vagina 3 cm proximal to the cervix and a sagittal view of the cervix was obtained. The calipers were used to measure the distance between the internal os and external os. Three measurements were performed, and the smallest of them was recorded. Subsequently, the characteristics of the internal cervical os were evaluated, describing presence or absence of funneling.
Finally, Bishop score was calculated based on the clinical assessment of cervical dilation, cervical consistency, cervical effacement, cervical position and station of fetal presentation. Fetal heart rate and uterine contractions were performed within the next 30 minutes. Patients with regular uterine contractile activity or abnormal fetal heart rate pattern were excluded from the study.
Induction of labor was performed according to the standard labor induction protocol of the Puerta de Hierro Majadahonda University Hospital. Patients with an unfavorable cervix (Bishop score ≤ 5) received 10 mg Dinoprostone vaginal pessary (Propess®, Ferring SAS, Gentilly, France) for 24 hours. Patients with Bishop score > 5 or contraindications for use of Dinoprostone had artificial rupture of the membranes. Oxytocin augmentation was indicated in case with unsatisfactory progress of labor or following amniotomy. On the other hand, there were several indications to remove the device before 24 hours: Beginning active phase of labor, hyperdynamics and alteration of fetal heart rate pattern.
The primary outcome in this analysis was vaginal delivery within 24 hours after induction of labor.
Sample size performed before the study. We based on study developed by Bajpai [12]. They calculated minimum required sample size according to method described by Jones [13] and Radeka prospective study based on Bishop score to evaluate successful of induction with prostaglandins.
To determinate independent predictive factors associated with vaginal delivery within 24 hours we used Chi-Square test for categorical variables, and Mann-Whitney and Kruskal-Wallis test for continuous variables. Factors with a degree of significance were entered in the multivariate analysis model using a non-automatic backward strategy. The final predictive model performance was evaluated by discrimination (area under the receiver operating characteristic (ROC) curve). Calibration was study by the Hosmer-Lemeshow goodness-of-fit statistics. Finally, we develop nomogram with the final predictive model to facilitate the interpretation of the results. A p-value < 0.05 was considered statistically significant. Statistical analysis was conducted using STATA version 14.1 (StataCorp, College Station, Texas, USA).
The ethical approval for this study was gained from Clinical Research Ethics Committee of Puerta de Hierro Majadahonda Hospital in Madrid (Act number 311). Women were informed of the objectives of the study, as well as they signed the informed consent to participate in the study.
The baseline characteristics of the 201 pregnant women included in the study are shown in Table 1. The indications for induction were prolonged pregnancy (n = 100; 49.8%), diabetes with and without insulin (n = 36; 18.0%), intrauterine growth restriction (n = 19; 9.5%), pregnancy induced hypertension (n = 10; 5.0%), large for gestational age without diabetes between 37 and 39 weeks of gestation (n = 13; 6.5%), oligohydramnios or polyhydramnios (n = 8, 4.0%), and other indications (n = 15; 7.2%).
Table 1: Characteristic of the study population. View Table 1
Vaginal delivery occurred in 169 (84.1%) women and, in 141 (70.1%) of theses, delivery was within 24 hours. Vaginal instrumental delivery was performed in 46 (22.9%) women. There were 32 (15.9%) deliveries by caesarean section, 12 (37.5%) for suspected fetal distress, 11 (34.4%) for failure to progress and 9 (28.1%) for cephalopelvic disproportion.
There were 140 (69.6%) women with a Bishop score ≤ 5. Distribution of the Bishop score is shown in Figure 1. The median sonographically measure cervical length was 21.30 mm (range 5-45 mm) and the distribution is shown in Figure 2. Of the total patients, 118 (58.7%) women had cervical funneling. In 59 (29.4%) women fFN test produce a positive result. In 123 (61.2%) women 10 mg Dinoprostone vaginal (Propess®, Ferring SAS, Gentilly, France) was applied for ripening of the cervix.
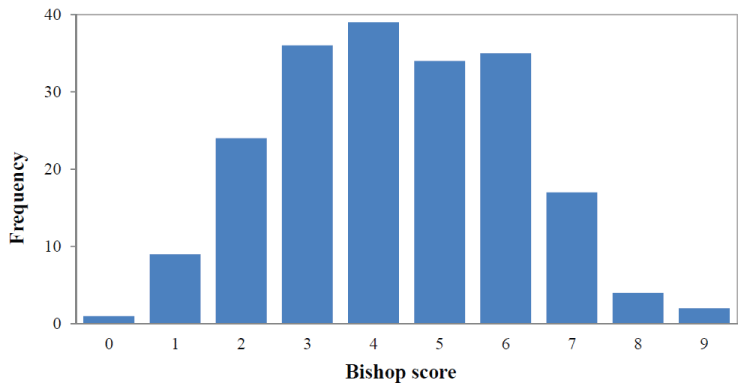 Figure 1: Frequency distribution of Bishop score. View Figure 1
Figure 1: Frequency distribution of Bishop score. View Figure 1
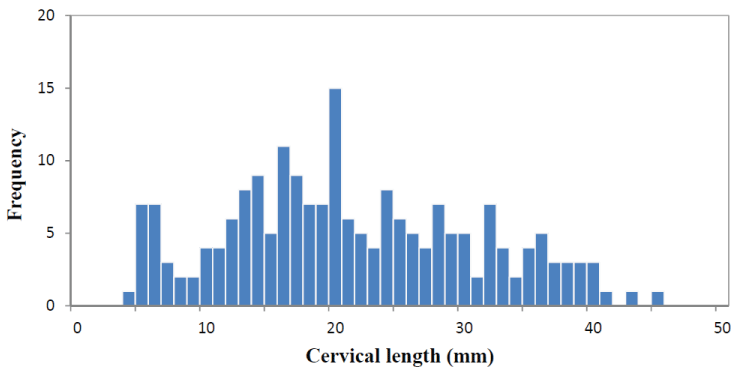 Figure 2: Frequency distribution of sonographically measured cervical length. View Figure 2
Figure 2: Frequency distribution of sonographically measured cervical length. View Figure 2
On univariate analysis only Bishop score and parity were found significantly associated with independent variables with vaginal delivery within 24 hours (Table 2). The result of multivariable logistic regression revealed that parity and Bishop score provided significant independent contribution in the prediction of vaginal delivery within 24 hours after induction (Table 2). A nomogram was constructed from the final multivariable logistic model (Figure 3). This is a pictorially nomogram where each variable (parity and Bishop score) is listed separately, with a corresponding number of points assigned to a given magnitude of the variable. Then, the cumulative point score for all the variables is matched to a scale of outcome of vaginal delivery within 24 hours.
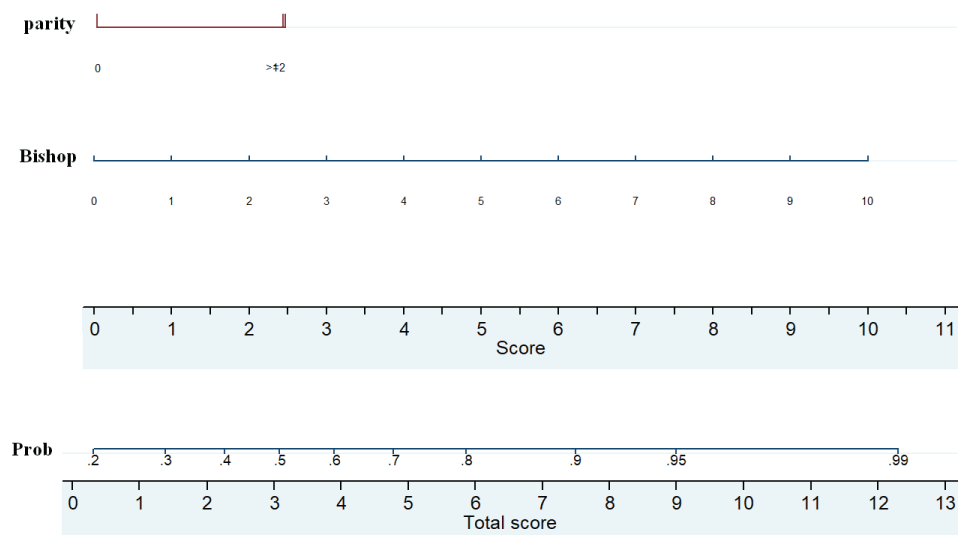 Figure 3: Nomogram predicting the probability of vaginal delivery within 24 hours. The probability is calculated by drawing a vertical line from each variable (parity and Bishop score) to the points horizontal axis so as to obtain a number of point for each variable. The total score for each patient is obtaining by summing up the 2 scores. Finally, a vertical line is projected from the “total score” line to the predicted probability scale to obtain individual probability of vaginal delivery within 24 hours after of induction. View Figure 3
Figure 3: Nomogram predicting the probability of vaginal delivery within 24 hours. The probability is calculated by drawing a vertical line from each variable (parity and Bishop score) to the points horizontal axis so as to obtain a number of point for each variable. The total score for each patient is obtaining by summing up the 2 scores. Finally, a vertical line is projected from the “total score” line to the predicted probability scale to obtain individual probability of vaginal delivery within 24 hours after of induction. View Figure 3
Table 2: Univariate and multivariate analysis for vaginal delivery within 24 hours. View Table 2
Discrimination was measured by the concordance index. This prediction model showed an area under ROC curve 0.78, which corresponded to the concordance index (Figure 4). The calibration performed using the Hosmer-Lemeshow test shows that the model is very close to the ideal.
 Figure 4: Area under the curve for the nomogram corresponding to Concordance Index. View Figure 4
Figure 4: Area under the curve for the nomogram corresponding to Concordance Index. View Figure 4
Induction of labor is a common procedure in current obstetrics. However approximately 20% of woman having induction labor end up having a caesarean delivery [2,14]. We developed a nomogram integrating both parity and Bishop score to improve and individualize prediction. However, ultrasound and fetal fibronectin were not found to be associated with the outcome of induction in our study.
Several studies in the past have evaluated a combination of ultrasound, maternal characteristics and Bishop score to predict the success of induction labor [14-20]. We found that both parity and Bishop score are parameters that predict the success of labor induction. Some authors have found result similar to ours and conclude that parity and Bishop score are important predictors [14-17,21]. Pelvic examination with calculation of a Bishop score is the current standard of care for induction planning rather than sonographic assessment of cervical length. However, some models concluded that transvaginal ultrasonography is a better predictor of outcome than Bishop score [19,22-25].
This study aims to identify the potential value of preinduction cervical length and funneling in prediction of successful vaginal delivery to develop a nomogram. This study did not analyze the relationship between Doppler flows in pregnancy. Recent study evaluated the relationship between Doppler flows in fetal vessels (middle cerebra artery, umbilical artery and venous system) and uterine artery and the incidence of cesarean section in pregnancy after 41 week with induction labor [26]. In our study, determining the presence of fetal fibronectin was not associated with successful labor induction. This is a contrast to studies showing an association between fetal fibronectin to the prediction of successful labor induction [8,27,28].
The success of induction can be measured in different ways. We, as well as Rane [14], considered the success of labor induction as vaginal delivery within 24 hours. Data reported in literature are divergent regarding the definition of successful induction; hence it is difficult to compare different studies.
Variables with statistical significance in the multivariate logistic regression analysis, parity and Bishop score, constitute the basis for the development of our nomogram. This nomogram can be applied for patient counseling and clinical management. Such a tool may be useful for deciding induction or caesarean delivery. For example, a woman who has predicted high risk of failure induction may elect to avoid the potential risk of intrapartum caesarean. This is the second report, to our knowledge, which describes a nomogram for predicting the success of labor induction. The nomogram constructed by Tolcher, et al. [20] utilizing maternal age, parity, height, BMI, maternal weight gain during pregnancy, gestational age, arterial hypertension, diabetes, and initial cervical dilatation. However, this model differed from ours in that it includes only nulliparous patients. We chose to include both nulliparous and multiparous woman, because several studies reported that parity predict independently the mode of delivery [14-17]. Our nomogram included Bishop score based on the clinical assessment of cervical dilatation, cervical effacement, cervical consistency, cervical position and the station of fetal presentation. However, Tolcher, et al. [20] in their model included initial cervical dilatation < 3 cm. Our nomogram used very basic data to predict the risk of cesarean section.
The rate of instrumental delivery in our study was considerable higher (22.9%). This is close to the results reported by Crane (20.8%) [29]. However, our cesarean rate was 15.9% compared to 24.3% in Crane's study [29]. On the other hand, the lowest rate of caesarean section and instrumental delivery are those obtained by Verhoeven [30], with an instrumental delivery rate of 13.0% and a cesarean section rate of 11.0%.
The major strength of this prospective study is the adequate sample size. Method of induction was the same in women undergoing cervical ripening. We only used prostaglandins (Dinoprostone) to avoid biases.
Our study is not free from limitations. A potential limitation was the inclusion of a homogeneous group of patients. Our study population is majority Caucasian and we do not have data on socioeconomic status. Although the nomogram has been internally validated, but it has not been externally validated in other populations.
In conclusion, the present study demonstrated that parity and Bishop score are independent prognostic factor for success of induction labor. This predictive nomogram based on clinical variables can be applied for patient counseling and clinical management of induction. It will help to decrease maternal and perinatal morbidity of prolonged labor and intrapartum caesarean delivery. Our nomogram should be validated in other populations prior to use in clinical practice.
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
The authors report no conflict of interest.