Background: Chronic knee pain, primarily associated with knee osteoarthritis (OA), is a leading cause of disability among older adults and place an immense burden on patients and healthcare systems, exceeding $80 billion in direct medical costs annually. Current non-surgical solutions to treat chronic knee pain provide limited relief of symptoms. The present study aims to assess the long-term effect of a biomechanical, non-invasive intervention on total knee replacement (TKR) incidence and the utilization of healthcare resources over five years.
Methods: This was a retrospective study among older adults with chronic knee pain, primarily due to OA, who received the biomechanical intervention between 2014 and 2017. An independent survey institute contacted patients between August and September 2022 to confirm their surgical status and utilization of other healthcare services, covering at least five years of follow-up. During the intervention, patients were provided with a FDA-cleared shoe-like device, which includes two convex pods attached to the sole. The pods are attached to the shoe in a customized manner based on the patient's gait and pain (i.e., calibrated). The device shifts the foot's center of pressure to reduce the knee joint load and alleviate pain. The device also creates controlled perturbation that challenges gait and posture stability and creates neuromuscular training. Patients were instructed to wear the device for up to 2-3 hours a day during their daily routine and invited to follow-up sessions for treatment modifications.
Results: Four hundred and fourteen patients responded to the survey. Their mean (SD) age was 66.5 (4.2) years, 65% were women and 82% of patients were diagnosed with knee OA. At five years since inception, the TKR incidence rate was 18.4% with 95% CI (14.6%, 22.1%). Most responders (79%) reported using other healthcare resources and services to treat their knee pain before the intervention. Of them, 64% ceased receiving intra-articular injections to the knee, 44% discontinued using pain medication and 46% stopped receiving physiotherapy.
Conclusion: This biomechanical, non-invasive intervention is a beneficial and sustainable intervention that reduces healthcare resource utilization, including avoidance of TKR and chronic knee pain. With the constant increase in the prevalence of chronic knee pain, offering this intervention to patients can help address the increasing burden on the healthcare system and society.
Trial registration: NIH protocol no. NCT00767780
Total knee replacement, Knee, Osteoarthritis, Pain, Healthcare resource, Biomechanical device
OA: Osteoarthritis; TKR: Total Knee Replacement
Chronic knee pain is a leading cause of disability among older adults. Between 18.1% and 23.5% of Americans older than 60-years-old report having significant knee pain, which was also associated with difficulties in performing physical function activities [1,2]. Loss of function and persistent pain generate substantial social and economic burden, accumulating to approximately $128 billion a year [3] from direct costs of medication and medical procedures, alongside the indirect costs of work loss and early exit from work by patients [4] and their caregivers [3,5]. In most cases, and specifically with aging, chronic knee pain is caused by knee osteoarthritis (OA), but may result from other conditions, such as knee sprains, meniscal injuries, and chronic inflammation [6,7]. It is estimated that about 50% of the patients who sustain knee pain caused by OA will ultimately have a total knee replacement (TKR) after exhausting non-surgical treatment solutions [8] and post-surgically patients may present with pain that persists beyond the expected recovery phase [7].
Healthcare resource utilization is significantly higher in patients with chronic knee pain as compared with their healthy peers [4,9]. Patients who have more annual outpatient visits are twice as likely to use pain medication and generate significantly higher medical costs, nearly double the mean per-person-per-year cost of matched controls [4]. Most non-surgical interventions to treat chronic knee pain offer only temporary relief. This leads patients to use recurrent procedures such as intra-articular injections or persist with medication intake frequently and over extended periods of time [7,8] thereby exposing them to a higher risk of side effects and medical complications [10]. Progression from first-line treatment into TKR significantly increases the medical cost per patient, with a national US average price for complete episode of care estimated at about $34,000 [11]. There is an unmet need for a viable, safe, and long-lasting non-surgical solution for chronic knee pain to improve patients' clinical outcomes and reduce the economic burden on the healthcare system. Recent evidence suggests that a FDA-cleared biomechanical, non-invasive, home-based treatment (AposHealth®) can address chronic knee pain with a long-term positive effect on both pain and the need for surgery [12,13]. This intervention uses a shoe-like device to manipulate the foot's center of pressure, alter ground reaction force trajectory to reduce loads on the affected knee joint, while inducing controlled gait and posture instability (i.e., perturbation) to alleviate pain, improve patients' function and gait pattern [14-16]. The present study aimed to assess this intervention's effect on TKR incidence and on the utilization of medical, non-surgical treatments over 5 years.
The study was a retrospective analysis of patients who received treatment for knee pain at the AposTherapy Center in Herzliya, Israel between 2014 and 2017. Patients between 60 to 75 years (on the day of their initial evaluation) who presented with primary knee pain were included in the study registry. We excluded patients if they (1) Had previous knee or hip replacement, (2) Sustained other, serious medical conditions that would affect their pain, function, or gait or (3) Reported acute symptoms that persisted less than three months prior to treatment. The study was approved by the Ethics committee at Assaf Harofeh Medical Center, Zerifin, Israel. The study is registered in clinicaltrials.gov (NIH protocol no. NCT00767780). All patients who started treatment were informed that their clinical data may be used anonymously for research purposes. Patients surveyed at follow-up were informed of the study goals and gave their written or verbal consent before responding.
The study had two primary outcomes: (1) The rate of TKR incidence at five years after starting treatment and (2) The change in patients' utilization of medical procedures and services to treat knee pain before and after the treatment. The AposTherapy database was used to confirm patients' eligibility, obtain demographic characteristics (i.e., age, gender) and clinical diagnosis (determined by the physiotherapist), and compile patient registry based on their initial evaluation visit. An external, independent research service (Sarid Research Institute Ltd, Haifa, IL) contacted all eligible patients between August and September 2022, to cover at least five years of follow-up since starting treatment. Patients' responses were collected through an online structured survey or by telephone interview with a human representative. To establish patients' surgical status, the survey included questions regarding the type of orthopedic surgery that occurred after starting treatment and the time of surgery. Patients were presented with multiple choice questions regarding their adherence to the treatment plan and utilization of other, non-surgical solutions to treat their knee pain before and after the treatment.
All patients received a personalized, non-invasive, home-based, biomechanical treatment that aims to alleviate knee pain and improve function (AposHealth®). The treatment is delivered via unique FDA-cleared footwear (Apos device- see Figure 1) and uses two convex pods attached to the plantar surface of the sole. A specially trained physiotherapist calibrates (i.e., customizes the location and size of the pods) the devices to reduce pain in the knee while walking. Calibration is determined based on patients' symptoms, physical examination, and computerized gait assessment. The adjustment of pod location manipulates the foot's center of pressure (COP), shifts the ground reaction force (GRF) vector, and reduces pressure on the knee joint immediately [17,18]. The convexity of the pods induces controlled perturbation by decreasing the foot base of support, therefore creating a challenge to gait and posture stability. This controlled instability triggers neuromuscular training response and alters patients' gait pattern [19,20]. After the initial calibration, patients are asked to wear the device for short periods of time while performing task-specific activities (i.e., daily living) and gradually increase wear time from approximately 20 minutes per day initially, reaching up to 2-3 hours per day. This home-based training allows patients to manage their symptoms and integrate the training into their daily routine, further inducing a carry-over beneficial effect to usual walking (without the device) over time [21]. Patients were requested to return to follow-up appointments in which re-assessment of clinical outcomes and gait patterns was performed and the calibration of the Apos device was adjusted as indicated. Treatment plans were adjusted at each appointment, and patients were encouraged to wear the device regularly at home as prescribed.
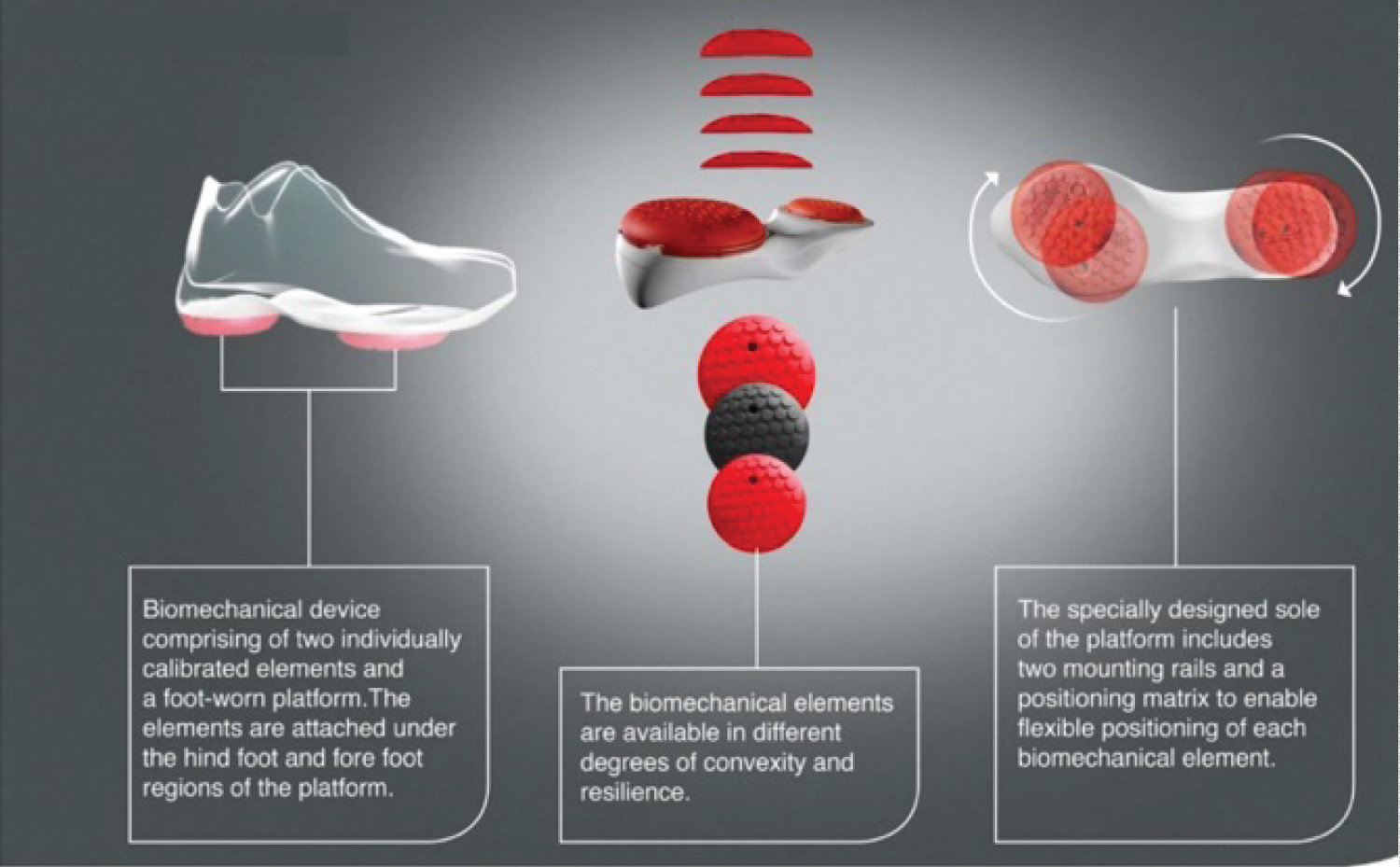 Figure 1: The biomechanical device.
View Figure 1
Figure 1: The biomechanical device.
View Figure 1
All statistical procedures were administered by an independent assessor (Sarid Research Institute Ltd) using R statistical software (version 3.6.3) [22]. Means and standard deviations were calculated for all dependent variables. To examine incidence rates of TKR over time, for each patient that reported having a TKR, we computed the duration (days) between the treatment inception and TKR surgery date. The proportion of patients who had a TKR out of the total number of patients was then used to obtain the TKR incidence rate at a daily resolution. The 95% confidence interval was computed using a two-sided exact binomial test. Changes in the utilization of other medical solutions before and after the intervention were described as rates (%) out of available responses. Alpha level of significance was set as 0.05 for all statistical procedures.
Invitation to participate in the survey was sent to 2521 patients who met the eligibility criteria. Of them, 414 patients (16%) responded to the survey, covering a mean (SD) follow-up duration of 2528.2 (377.1) days since their initial evaluation. Participants were mostly women (65%), with a mean (SD) age of 66.5 (4.2) years when they started treatment. Most of the participants (82%) had a clinical diagnosis of knee OA, 1% had Osteonecrosis of the knee, 7% sustained chronic pain due to ligament tears and fractures and an additional 10% of the participants presented with patellofemoral joint pain. Participants reported high adherence to the treatment, with 90% attending all follow-up visits and 82% of the patients who recalled following treatment plan as indicated.
The incidence rate of TKR since inception is presented in Figure 2. At five years, the incidence rate of TKR (covering all responders) was 18.4% with a 95% CI [14.6%, 22.1%].
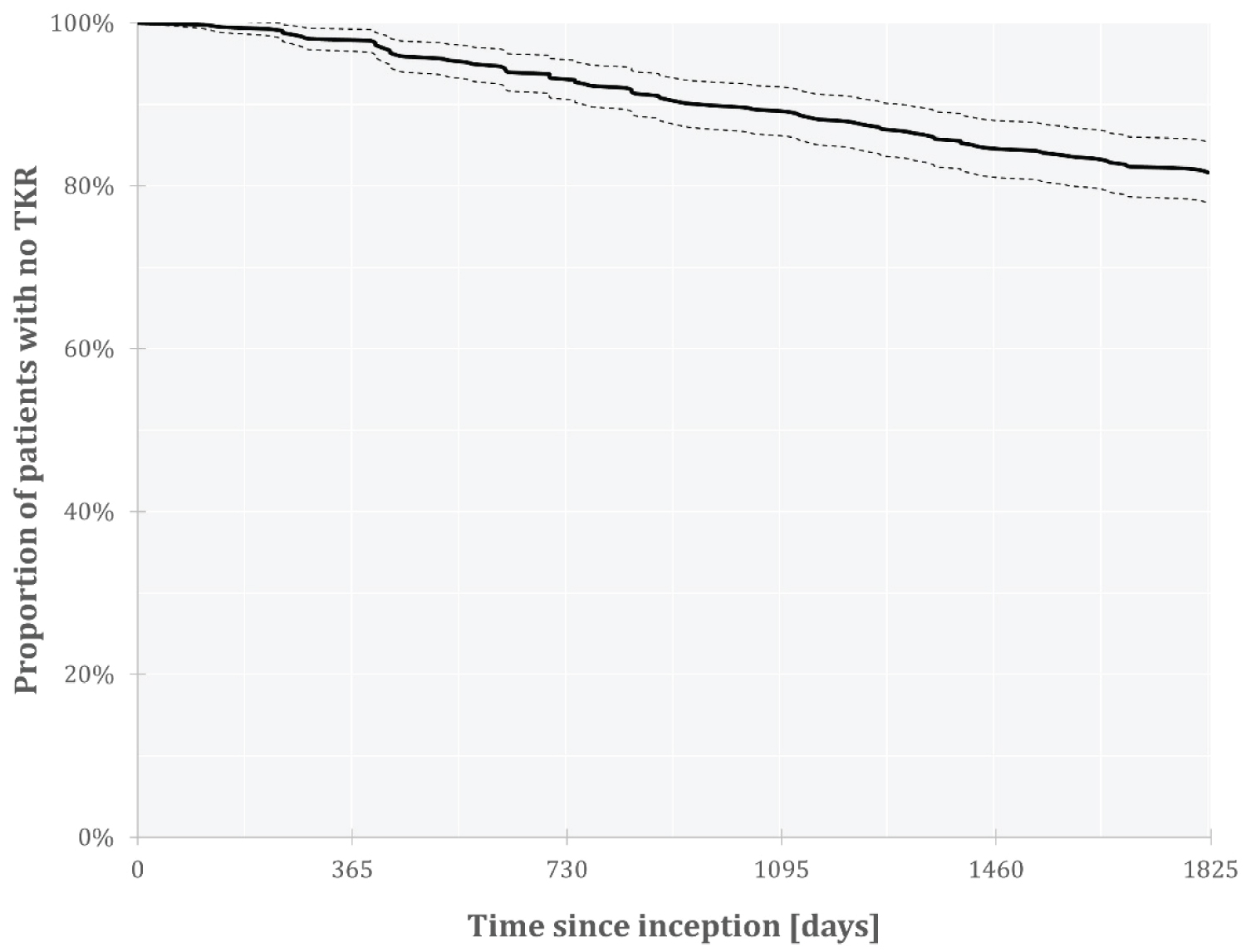 Figure 2: TKR incidence rates over time.
View Figure 2
Figure 2: TKR incidence rates over time.
View Figure 2
Table 1 presents the data of 326 participants who reported on their utilization of other medical solutions for knee pain before and after starting the biomechanical intervention. Almost 80% of the patients (n = 258) reported they used other medical treatments prior to the intervention. Of patients using these various therapies, over time, 44% of the patients ceased using pain medication, 64% of the patients ceased using intra-articular injections to the knee, and 46% of the patients discontinued using physiotherapy (see Table 1). Another portion of patients continued using alternative solutions for their pain but reported on reduction in the use of pain medication (22% of the patients), intra-articular injections (17% of the patients) and physiotherapy (26% of the patients) after starting treatment with the biomechanical device.
Table 1: Changes in utilization of other healthcare resources before and after the intervention. View Table 1
The current study examined the long-term effect of a non-invasive, home-based biomechanical intervention on the TKR incidence and the utilization of healthcare resources among patients with chronic knee pain, primarily due to knee OA. Our key findings suggest that patients who received the biomechanical intervention had a relatively low incidence rate of TKR over five years since starting treatment. These patients significantly reduced the utilization of pain medication, intra-articular injections, and physiotherapy to treat their chronic knee pain. These findings extend previous reports that demonstrated 2-year outcomes of low incidence of TKR, improved pain, function, and gait among patients with end-stage knee OA [12,13].
The survey results indicate that this biomechanical intervention reduced the use of pain medication and intra-articular injections among patients with chronic knee pain. This is significant for a few reasons. Firstly, it suggests that the intervention helped patients avoid the potential complications and side effects linked to prolonged analgesic use and recurrent injections [7], which may contribute to the long-term progression of knee joint degeneration [10,23-25]. Secondly, it implies that patients who stopped other treatments found the biomechanical intervention to be a more sustainable solution for their knee pain, a conclusion that is also supported by the low incidence of TKR over five years. We did not evaluate the treatment effect on patients' symptoms in the scope of the present study. However, previous reports have shown that the use of this biomechanical device led to a significant reduction in pain and improvement in function among patients with knee OA that persisted over at least two years [12,13,15]. Interestingly, participants have also reported decreased physiotherapy use after starting treatment. It is possible that this home-based intervention allowed patients to be more active, incorporate training into their daily routine and reduce the need for supervised training or manual therapy. Together, these findings indicate that the biomechanical intervention may supply a cost-effective solution for the management of knee pain.
The 5-year TKR incidence rate that was found in the study was 18.4%, which is significantly lower than the natural decay rate reported among patients presenting with similar symptoms [26-28]. As a conservative comparison, Larsen, et al. demonstrated a 30% to 34% TKR incidence over five years among newly diagnosed patients with knee OA who were not initially eligible for surgery [25]. Similar results were obtained in a New-Zealand based study that showed a 38.5% TKR incidence at five years since diagnosis among 186 patients who received first-line, non-operative treatment for their knee pain [28]. Most of the participants in the present study were diagnosed with knee OA, and while we could not confirm their radiological diagnosis, it is safe to assume that they were similar in nature to patients with moderate to severe symptomatic knee OA. Patients' age, clinical diagnosis, symptom duration and the fact that almost 80% of them reported using other treatment alternatives prior to the biomechanical intervention suggest that the participants in the study were at increased likelihood to progress to TKR. Overall, these findings highlight the potential effect of the biomechanical device and intervention on improving patients' status and reducing the risk of progression to TKR. For healthcare systems this is significantly important, as TKR average market cost, according to a Blue Cross Blue Shield report, may rise up to $61,750 per patient in the New York metropolitan area [11]. Therefore, this customized device and intervention may help to reduce the costs of knee OA management significantly.
The present study has several limitations. First, this retrospective analysis and survey did not include a control group. To address this, we compared the results of the current study with other publications to provide reference to the outcomes for similar patients, albeit the patient population in the current study seems more severe than in previous publications. Hence, we assume a reduction of 50% likelihood of TKR is a conservative assumption. Second, this survey was based on participants' reports and was subject to recall bias. This should not create a major bias as the severity and rehabilitation associated with TKR should produce a very high rate of patient recall. Third, patient records were limited and did not include a standardized severity measure, and the group may have varied in their symptoms and diagnosis.
In conclusion, the current study suggests that a non-invasive, home-based biomechanical intervention is a beneficial and sustainable treatment option for patients with chronic knee pain. The study found that patients who received the intervention had a low and significantly reduced incidence rate of TKR over five years and also reduced their utilization of related pain medication, intra-articular injections, and physiotherapy therefore may be a cost-effective solution for managing knee pain. This innovative therapy appears to be a very effective solution for moderate to severe knee OA as it reduces the need for surgery in patients with this common condition and can potentially reduce costs to the healthcare system.
The study was approved by the Ethics committee at Assaf Harofeh Medical Center, Zerifin, Israel and registered in clinicaltrials.gov (NIH protocol no. NCT00767780). Participants gave their informed consent upon starting treatment and when responding to the survey.
Not applicable.
The data that support the findings of this study are available on request from the corresponding author (SSS). The data are not publicly available due to them containing information that could compromise research participant privacy/consent.
One or more of the authors have received or will receive benefits for personal or professional use from a commercial party related directly or indirectly to the subject of this article.
Avi Elbaz and Amit Mor hold shares in AposHealth.
Shirley Shema-Shiratzky is asalaried employee of AposHealth.
AposHealth used the service of an external survey company to perform the projectto ensure independent data collection and analysis.
Conceptualization: SSS, AM, AA; Methodology: SSS, AM, AE; Writing-original draft preparation: SSS; Writing-review and editing, AM, AE. All authors have read and agreed to the published version of the manuscript.
The authors wish to thank Sarid Research Institute for their role in data collection and analysis.