Idebenone (IDBN) is an antioxidant compound, structurally related to coenzyme Q10. It's therapeutic potential is growing in different areas. Over the two decades ago, for potential treatments of mitochondrial diseases have been suggested, relatively few have undergone controlled clinical trials. This review focuses on the recent history of manuscripts of idebenone, coenzyme Q10 and exercise in both primary disorders and secondary disorders. Despite prior clinical impressions that IDBN had a positive effect and significant benefit on Mitochondrial disorders.
Leber hereditary optic neuropathy (LHON), Idebenone (IDBN), Mitochondrial disease, Mitochondrial DNA, Coenzyme Q10, Friedreich ataxia (FA), Dichloroacetate
Mitochondrial disorders are a group of rare hereditary diseases that affect energy metabolism. They are caused by impairment of the mitochondrial oxidative phosphorylation system [1]. Mitochondria perform a multitude of tasks, including combating the production of reactive oxygen species, initiating apoptosis, and generating energy as Adenosine Triphosphate (ATP) using the electron-transport chain and the oxidative phosphorylation system. While numerous treatments for mitochondrial disorders have been suggested, treatment of these disorders is challenging, as only symptomatic therapy is available. In this review we will focus on drugs (IDBN) for and treatment of Mitochondrial diseases.
Idebenone is a rapidly absorbed, safe and well-tolerated drug and is currently the only clinically proven treatment for mitochondrial disease and Leber's hereditary optic neuropathy and neurological disease.
This study will determine that a drug called idebenone is safe and effective in reducing the level of oxidants that are believed to damage the nervous system and hearts in patients with mitochondrial disease. Mitochondrial disease may be caused by alterations of the mitochondrial genome by an abnormality in the gene that makes a certain protein.
Idebenone (IDBN), first synthesized in Japan in the 1980s, (Figure 1) is an analogue of coenzyme Q10 (CoQ10), the lipophilic electron carrier and endogenous antioxidant found in all cellular mitochondrial membranes [2].
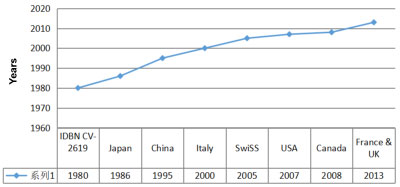 Figure 1: The history of idebenone (IDBN) supplements. View Figure 1
Figure 1: The history of idebenone (IDBN) supplements. View Figure 1
Structurally, Coenzyme Q10 and IDBN share the same substituted 1,4-benzoquinone ring but they have a different side chain than carbon 2 (Figure 2).
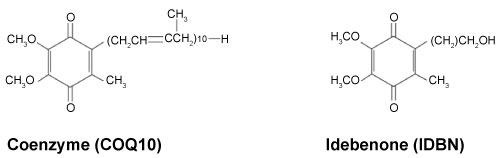 Figure 2: Chemical structures of Coenzyme Q10 and its short-chain analog, Idebenone. View Figure 2
Figure 2: Chemical structures of Coenzyme Q10 and its short-chain analog, Idebenone. View Figure 2
In fact that the hydroxydecyl side chain makes IDE less lipophilic than CoQ10. However, the hydroxydecyl side chain of IDBN seems to be of the ideal length for partitioning into the mitochondrial membrane and for a better blood brain barrier (BBB) permeation in comparison with CoQ10 [3].
In advance studies given its mechanism of action, IDBN was employed for the treatment of neurodegenerative disorders and diseases that exhibited mitochondrial etiology [4]. IDBN was tested with conflicting results in Friedreich's ataxia (FA) [5], a genetic, progressive disease that usually begins in childhood and affects multiple systems, including the nervous system and the heart. It also demonstrated that bile acids inhibit mitochondrial electron transport [6] and that hepatic mitochondria generate increased quantities of oxygen free radicals when exposed to hydrophobic bile acids [6,7]. This data suggest that hydrophobic bile acids may cause increased electron leak from the altered electron transport chain, resulting in the generation of superoxide and hydrogen peroxide, as observed with other chemical blockers of mitochondrial electron transport [8], therefore, targeting antioxidant therapy to the mitochondrion may be an effective strategy to minimize free radical generation and toxicity in the hepatocyte exposed to hydrophobic bile acids.
In IDBN-treated Friedreich's ataxia [FA] patients, generally, a reduction in oxidative stress markers was reported and many patients showed an improvement of non-neurological symptoms, as well. However, in a subsequent randomized, double-blind, placebo-controlled trial, no statistically significant difference between the placebo and IDBN was revealed on the International Cooperative Ataxia Rating Scale [9,10].
Furthermore, IDBN has been proposed for the treatment of skin disorders that could benefit from topical supplementation with antioxidants. Regarding its topical application, IDBN showed the highest oxidative protection capacity when compared to, CoQ10 [11]. Hence, the cosmetic industry has considered IDBN an attractive molecule for developing anti-ageing products.
A recent study shed light onto the mechanism of IDBN and provided more insight into its possible therapeutic use for treating or preventing atherosclerosis by ameliorating mitochondrial dysfunctions in endothelial cells [12].
For the classification, clarification, and presentation of the available scientific evidence about use of Idebenone in management of Mitochondrial Disorder (MD) in this systematic review, one of the most accredited scientific databases, PubMed, was explored with keywords (MeSH terms) 'Idebenone and Co-enzyme Q10" Drugs and 'Mitochondrial Disease, Leber's hereditary optic neuropathy and nervous system disease'. The search order was ("Nervous system" and "Idebenone" [Supplementary concepts]. Only journal articles were acceptable for this review. There was no language restriction in our methodology, but all articles had English abstracts. According to the low number of search results, no time limit was planned for the included articles.
Key finding in this Literature Review some articles that assessed the efficacy of Mitochondrial disease Friedreich ataxia (FA) and Leber hereditary optic neuropathy (LHON). The present study was planned to gain further evidence supporting the efficacy and safety of idebenone in the treatment neurological degeneration, cardiomyopathy, and diabetes.
As discussed earlier, idebenone is similar to Coenzem Q10 and tacrine in its ability to function within the mitochondrial electron transport chain (ETC) and to be a scavenger for ROS, and may be better transported into the brain. This has led to trials of idebenone in Mitochondrial dieases Friedreich ataxia (FA), MELAS, and Leber hereditary optic neuropathy (LHON) [13,14].
In summary, none of the controlled clinical trials have described clearly established efficacy and safety for the interventions tested for therapy of mitochondrial diseases, including ''primary'' genetic disorders or ''secondary'' degenerative diseases associated with mitochondrial dysfunction. However, it is clear from the experience with successive trials of administering dichloroacetate (an activator of pyruvate dehydrogenase complex) (DCA) [15], and from trials of idebenone that controlled clinical trials are necessary to establish efficacy and safety, and that repetitive collaborative trials may be necessary to establish even relatively small incremental benefits.
Friedreich ataxia (FA) is due to mutations in the FXN gene, which encodes frataxin, an iron carrier protein and donor for iron-sulfur cluster biosynthesis, which is critical for components of the mitochondrial ETC. Deficiency of frataxin results in intramitochondrial iron accumulation and progressive neurological degeneration, cardiomyopathy, and diabetes.
Over the last 13 years there have been 13 clinical trials of idebenone in FA, using progressively larger doses, emerging to phase III trials, indicating that higher doses of idebenone are well tolerated and may stabilize neurological function in young patients with FA [16,17].
Again, these successive trials are instructive for smaller trials in less common mitochondrial disorders in that they involve multicenter collaboration, carefully controlled trial design, agreed upon primary outcomes, and build upon the experience of previous trials [18].
An ongoing trial of idebenone in MELAS is being conducted at Columbia University. This is a phase IIa (dose-finding), double-blind, randomized, placebo-controlled study [14]. The study design randomizes patients to 1 of 3 groups: Group A receives 900 mg/d of idebenone for 1 month, group B receives idebenone at 2250 mg/d, and group C receives a placebo.
Idebenone also has been evaluated in a controlled, international collaborative treatment trial of LHON [19]. LHON is associated with mutations in mtDNA affecting complex I of the ETC, and is the most common mitochondrial disorder causing progressive blindness, and is most prevalent in young adult males. This was the first adequately powered, multicenter, randomized, placebo-controlled trial for this disorder, which followed earlier uncontrolled reports that idebenone resulted in improvement of vision in LHON.
Finally many papers on the baseline of visual acuity (VA), indicated a beneficial effect of idebenone in patients. Discordant VA in LHON has been thought to be related to early stages of the disease, although that was not confirmed by history in this subgroup. Color-contrast for blue-yellow also improved significantly in those receiving idebenone, but not red-green, indicating a benefit for the larger stratified nerve fibers. Based on this finding, a follow-up phase III clinical trial has been proposed [20].
Most of studies on the idebenone drugs so far have compared active drugs to therapeutic value and measured the benefits after a few weeks after treatment. In our approach, we followed the clinical rationale that safety influences the efficacy of idebenone.
On the other hand, one of the aims was to show that drugs with low to moderate clinical safety could not be effective in large cohorts of patients.
Idebenone is the drug approved for the treatment of patients with (Mitochondrial, LHON, FA). Treatment with idebenone has been shown to be effective, safe, and well tolerated.
Idebenone, an analog of coenzyme Q, has a more favorable pharmacokinetic profile than other Co Q analogs [21]. However, idebenone may represent an inhibitor of both the redox and proton-pumping activity of complex I [22] and may cause mitochondrial depolarization and reduced nicotinamide adenine dinucleotide (NADH) depletion [21,23].
Most recently, idebenone has been shown to be effective in improving recovery after the onset of vision loss in Leber's hereditary optic neuropathy (LHON) [24]. The current clinically relevant benefit from treatment was established by evaluating the treatment's effect in three dimensions of efficacy.
Finally, there are general advantages to using idebenone, but according to a series of clinical summarized case reports and common medical practice of specific therapies for mitochondrial disorders, these advantages are insufficient for evidence-based medicine. Formerly advocated therapies have usually been proven to be effective and may have acceptable results. Clearly, follow up of great clinical work is unnecessary to describe the effectiveness and safety of current therapies for Michondrial and FV and LHON disorders and hopefully in the future idebnone will lead to more effective drugs with fewer side effects.
In our conclusion some of the studies have demonstrated the potential benefits of IDB, many researchers and doctors attempted different strategies to increase IDE efficacy and safety in the treatment of neurodegenerative diseases as well as skin disorders. IDB chemical modifications mainly focused on increasing IDB activity at the cellular or mitochondrial level, while nanotechnology approaches aimed at designing novel formulations to improve IDB systemic and local efficacy. The identification of quantifiable disease-specific biomarkers will aid in the quest for rational treatment strategies in the future.
Not applicable.
Not applicable.
The authors have no conflict of interest.