Certain drugs can induce ventricular tachycardia (VT) in a different mechanism [1]. Most knowed mechanisms are creating new reentry, enhancing ventricular after potentials or exaggerating the slope of phase 4. These kind of arrhythmias can or can not be symptomatic, sustained or non-sustained and have variable ECG changes: Monomorphic or polymorphic ventriculer extras, bidirectional or torsades de pointes ventricular tachyarrhythmias. Zuclopenthixol decanoate blocks postsynaptic Dopamine receptor in the limbic system. It is a potent antagonist of D1 and D2 receptors. Zuclopenthixol decanoate also has high affinity for alpha1-adrenergic and 5-HT2 receptors. Zuclopenthixol decanoate may cause QT prolongation like other drugs belonging to the therapeutic class of antipsychotics [2]. Persistently prolonged QT intervals may increase the risk of malignant arrhythmias. This is first drug induced VT case at literature which was developed a after zuclopenthixol decanoate (Clopixol® Depot) IM injection.
A 64-years-old male patient was admitted to the emergency department with complaints of palpitation, chest pain, feeling dizzies and presyncope. ECG records showed ventricular tachycardia of 169 beats per minute (Figure 1C). Ventricular tachycardia (VT) rapidly changed to sinus rhythm with medical cardioversion by using amiodarone infusion with the dosage of 5 mg/kg in the form of a short term infusion within 30 minutes in the emergency department (Figure 1B).
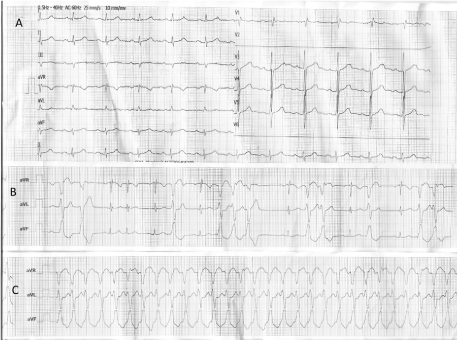 Figure 1: A) ECG record in intensive care ünit follow-up and before hospital discharge; B) ECG record in emergency department after amiodarone infusion, ECG; C) First ECG record in admission to emergency department. View Figure 1
Figure 1: A) ECG record in intensive care ünit follow-up and before hospital discharge; B) ECG record in emergency department after amiodarone infusion, ECG; C) First ECG record in admission to emergency department. View Figure 1
The QTc value of the patient was evaluated as 460 ms in ECG when sinus rhytm (Figure 1A). Left ventricular ejection fraction was 50% with septal paradoxic movement, minimal pericardial effusion, systolic pulmonary arterial pressure 40 mmHg, minimally mitral insufficiency, 1-2 degree tricuspid insufficiency had found by transthoracic echocardiography assessment. All procedures were performed after patient's consent taken. Coronary aniography performed and noncritical stenosis had been found. Patient was discharged without any symptoms after four days coronary intensive care unit follow-up.
Ventricular tachycardia is an extremely dangerous rhythm. It refers to any rhythm with wide QRS complexes faster than 100 beats per minute arising distal from the bundle of His. VT lasting over 30 seconds is called sustained tachycardia [3]. Ischemic heart disease is the most common reason, in which myocardial scar is the substrate for reentry. Commonly it is associated with hemodynamic unstable, particularly if the left ventricle ejection fraction low. Ventricular tachycardia is common in structural and valvular diseases of the heart, utilization of drugs such as isoproterenol, digoxin, quinidine, epinephrine, physostigmine, theophylline, cyclic antidepressants, antihistaminic, and thyroxin, metabolic problems such as hypoxia, electrolytes disturbances, alkalosis or deep acidosis [4]. VT may cause important symptoms such as acute palpitations, dyspnea and syncope. Weakness, chest pain and dizziness can occur [4]. Similarly our case presented to the emergency unit with weakness, dizziness, and dyspnea. Zuclopenthixol decanoate may cause QT prolongation like other drugs belonging to the therapeutic class of antipsychotics. Persistently prolonged QT intervals may increase the risk of malignant arrhythmias. Therefore, zuclopenthixol decanoate should be used with caution in susceptible individuals (with hypokalemia, hypomagnesaemia or genetic predisposition) and in patients with a history of cardiovascular disorders, e.g. QT prolongation, significant bradycardia (< 50 beats per minute), recent acute myocardial infarction, uncompensated heart failure, or cardiac arrhythmia. Concomitant treatment with other antipsychotics should be avoided.
The length of the QTc interval has been associated with the risk of sudden death after myocardial infarction [5,6]. QTc interval prolongation is a flag that warns of the possibility of torsade de pointes and sudden death. However, it is important to realize that QTc interval prolongation is a warning, not the risk itself. Not all drugs that prolong the QTc interval produce torsade de pointes and sudden death. Amiodarone, a class III antiarrhythmic drug, produces substantial prolongation of the QTc interval but very rarely produces torsade de pointes. Conversely, quinidine, with less severe prolongation, is more often a cause of torsade de pointes.
When our case admitted to the emergency department, he was hemodynamically stable. Cardioversion was succeed with amiodarone of 5 mg/kg in the form of a short term infusion with in 30 minutes and sinus rhythm was provided. After echocardiography, coronary anjiography and monitored 4-days intensive care unit flow-up any cause (coronary artery disease, electrolyte imbalance) about VT had not been diagnosed.
We do not have any idea about ECG findings before zuclopenthixol decanoate treatment but there was no prolonged QT with 460 ms on ECG after cardioversion and in hospital follow period (Figure 1).
We could not exclude an idiopathic VT because the patient refused electrophysiological study but in the case of arrhythmic event did not develop again after the zuclopenthixol decnoate medication stopped and there is no QT interval change with comparison ECG before hospital discharge with after discontinuation of zuclopenthixol, so these are making far away us from an idiopathic VT.
On litarature research about zuclopenthixol decanoate (Clopixol® Depot) cardiac advers events are < 1% chest pain and very rarely palpitation [7]. Naranjo Adverse Drug Reaction (ADR) probability scale performed for patient and 5 points found as result (events judged as probable ADEs if Naranjo ADR probability scale score of > 4), that means probably VT had been because of zuclopenthixol decanoate (Clopixol® Depot). Other wise there are strong evidences with other typical antipsychotic medical scan cause ventricular arrhythmia and sudden cardiac death [8], But there is no evidence on litarature records VT due to zuclopenthixol decanoate (Clopixol® Depot) usage.
There were certain drugs can induce side effects in a different mechanism but uncertain drugs and unexpected side effects always must keep in mind.
All authors decline there is no conflict of interest. There is no need to ethic commitee approval at case report but all procedures were conducted in accordance with ethical rules.
This case presented as a poster on 05-08 October 2017 for the occasion of our 33th Turkish Cardiology Congress with International Participation.
Bu vaka 05-08 Ekim 2017 tarihleri arasındaki Uluslararası Katılımlı Türk Kardiyoloji Kongresinde Poster olarak sunulmuştur.