The role of the tongue in the pathophysiology of anterior open bite (AOB) is poorly understood. This study characterized tongue morphology and functional motion, craniofacial skeletal pattern and upper airway space, and speech function in East African children with and without AOB.
Ten 9-12 year-old children of East African descent were recruited, five with AOB and five as control. Cone-beam CT scans, ultrasound imaging of the tongue during chewing and swallowing, and inter-labial pressure and lip muscle activity during lip closure and speech were taken in all subjects.
No significant differences were identified in most linear and angular measurements between the two groups except for the smaller mandibular and combined airway volumes in AOB (p < 0.10). Mandibular volume was positively associated with total face and ramus heights, and bilateral condylar width. The oral airway volume was associated with total face and anterior lower face heights. The combined oral and pharyngeal airway volume was associated with anterior lower face height. No significant differences were identified for the average tongue thicknesses between the two groups during swallowing and chewing. However, AOBs had a larger range of motion and average maximal velocity than controls. Significant associations between the tongue and skeletal variables were most often seen in tongue thickness during chewing. The average raw score of articulation tests was higher in AOBs than controls, and more isolated efforts of sound production with an extended duration and less corresponding muscle activity in speech intelligibility tests were identified in AOBs than those of controls. AOBs also presented significantly lower lip muscle activity during lip closure.
1) Craniofacial skeletal patterns are similar between AOBs and controls, but AOBs have smaller volumes of mandibular and upper airway spaces; 2) The tongue moves farther and faster during chewing and swallowing in AOBs; 3) The tongue thickness in chewing is more related to skeletal variables than other tongue features; 4) AOBs are more likely to have articulatory deficiencies and speech disorders.
Open bite, Tongue, Cone-beam CT, Ultrasound, Speech disorders
AOB: Anterior Open Bite; CBCT: Cone-Beam CT; OO: Orbicularis Oris; iEMG: Integrated Electromyogram
Anterior open bite (AOB) is defined as no vertical overlap of the maxillary and mandibular incisors when the posterior teeth are in occlusion [1-3]. Although AOB may not be the most frequent type of malocclusion, it is one of the most challenging malocclusions to treat because of its multifactorial etiology and its high relapse rate [4-10]. The overall prevalence of AOB is approximately 3.5% in Americans aged 8 to 17 [11], but 3-4 times more in East African adolescent and adult populations when compared to Caucasians [12]. Although the general clinical appearances may be similar across the various ethnic groups, AOB in the black population is expected to have specific features in facial profile, occlusion, alveolar relationship, skeletal pattern, and shape/structure of oral-facial soft tissues, particularly the tongue and jaw muscles [13,14].
In addition to genetics, etiology of AOB involves both morphological and functional factors, along with the interaction of environmental factors such as diet, tongue habits, eruption disturbances, and vertical facial growth pattern [15]. Morphological factors such as skeletal pattern and soft tissue shape, and functional disorders such as immature swallowing patterns, problems with mastication, abnormal tongue movement, and speech impediments, are also considered to be important consequences as a physiologic adaption [16]. Identifying and characterizing these morphological and functional problems can lead to better understanding of mechanisms and more successful treatment plans for patients with AOB in general, and to better explain why AOB is more common and more severe in East Africans in particular.
The tongue involves various oropharyngeal functions such as respiration, speech, chewing, and deglutition. This muscular organ protects the airway from spillage and maintains tonic muscle contraction in order to keep the airway patent, but also places significantly sustained and instant forces on teeth and their support structures. Because the tongue is positioned deep within the oral cavity and is inaccessible to many instruments, measuring its morphology and function is often a challenge. Ultrasound imaging has been proven to be a valid and reliable way to measure tongue morphology and motion in the last three decades [17-19]. This unobtrusive and non-invasive technique creates real-time images that map tongue surface motions using high-frequency sound waves. Additionally, ultrasonography has the advantage of being rapid, easily repeatable, radiation-free, and relatively inexpensive [20]. Therefore, the present study was to apply this technique to characterize tongue morphology and functional motion, and to further correlate tongue features with craniofacial skeletal variables. Although it is widely accepted that teeth alignment and inter-labial closure (pressure) play important roles in functional articulation and speech production [21-23], their relationships to the type and severity of malocclusions remain controversial and few studies have addressed this issue [24-27]. Therefore, these evaluations were included in the present study as well.
The hypothesis of the present study is that the functional motion of the tongue is more pronounced in AOBs than that in controls during chewing and swallowing in order to accommodate for increased facial vertical dimension. Additionally, some unique of craniofacial features, lip function, and speech characteristics may exist in AOBs.
Ten East African children (4 males and 6 females, aged 9 to 12 years.) were recruited for the study. All were the first generation immigrants from Somalia (7), Ethiopia (2), and Eritrea (1), and had no history of orthodontic treatment. Five (2 males, 3 females) with negative overbite were categorized as AOB group; the other 5 with overbite larger than 1.0 mm were taken as controls. Incisal contact was determined cephalometrically and verified with the subject's pre-orthodontic casts as reported previously, along with other dental features and habits upon clinical examinations [28].
All subjects sat in the CBCT chair with the Frankfort horizontal plane parallel to the ground and received cone-beam computed tomography (CBCT) scans (Hitachi Dental Cone-Beam X-ray System, CB Mercury, Hitachi Medical Corp. Tokyo, Japan). The exposure parameters were: 90 kV, 5.0 mA, and a voxel resolution of 270 µm. These CBCT scans were first used to compare the craniofacial skeletal morphology by landmarking 10 linear and 3 angular measurements (Figure 1A), and then to establish 3D models of upper airway (oral and pharyngeal airway spaces) and the mandible for volumetric measurements (Figure 1B) by using CB-Works (Hitachi Medical Corp, Tokyo, Japan) and OnDemand3D (Kavo Dental, Charlotte, NC) software through the segmentation technique. The reliability of CBCT measurement methods were examined through the inter- and intra-investigator agreement tests as did previously [29].
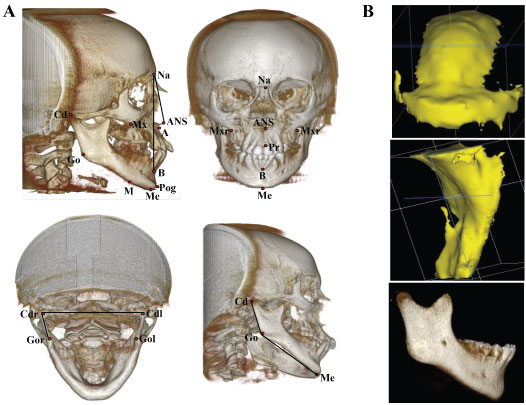 Figure 1: A) Skeletal landmarks and definitions in CBCT images; B) 3D models of intraoral (upper) and pharyngeal air (middle) spaces and mandible (lower) through segmentation of CBCT images.
Figure 1: A) Skeletal landmarks and definitions in CBCT images; B) 3D models of intraoral (upper) and pharyngeal air (middle) spaces and mandible (lower) through segmentation of CBCT images.
Points: Na (Nasion) - The deepest point on the skull between frontal bone and nasal bone in sagittal plane; Me (Menton) - The lowest point on the mandible; ANS (Anterior Nasal Spine) - The most anterior point of the anterior nasal spine; A (Subspinale) and B (Supramentale) - The deepest points on the curvature located below the ANS and above the Pog; Pog (Pogonion) - The most anterior point on the mandible; Pr - The most inferior point between the upper central incisors; Mx - Zygomaticoalveolar crest, the deepest point on the curvature of maxilla around molars; M - Point is located on the mandible so that the line that M-Mx makes is parallel to the Pog-Na line; Go - The most anterior point of the on the curvature of the mandibular angle; Cd - The most superior and posterior point of condyle.
Ten linear measurements: Heights (5): Na-Me - Total anterior face; ANS-Me - Anterior face; Pr-Me - Anterior lower face; Mx-M - Middle face; Cd-Go - Mandibular ramus. Widths (3): Mxr-Mxl - Anterior face; Cdl-Cdr - Bilateral condyles; Gol-Gor - Mid-width of mandible. Lengths (2): Me-Go - Mandibular body; Cd-Pog - Total mandible. Three angular measurements: ANB - Antero-posterior relationship of jaws; Cd-Go-Me - Gonial angle; Cdl-Go-Cdr - Internal ramal inclination, angle formed by the ramal and mandible width.
View Figure 1
Ultrasound scanning was performed using a Philips HDI 5000 clinical scanner equipped with a custom-designed image capture system [30] that includes 3D-tracking and saves the instrument video output to a computer workstation as JPEG images at a rate of 30 frames/s. The subject sat with the Frankfort plane parallel to the floor, and a 4-2 MHz phased array transducer was placed on the sub-mandibular region to acquire gray-scale (B-mode) images of the tongue in both sagittal and coronal planes. Subjects were instructed to chew gum on a single side at voluntary speed for several minutes and to swallow water (20 ml) six times while ultrasound scanning was performed.
Ultrasound images were captured at several locations and orientations for each task. The scanning protocol was defined as follows: 1) The chewing phase was imaged at 8 locations with the ultrasound transducer held at a single location for 5 seconds (5 locations for sagittal and 3 locations for coronal planes, 30 frames/s, total of 150 images each). The sagittal scans were specified by left, left-mid, mid, right-mid, and right positions, and the coronal scans were specified by anterior, mid, and posterior positions; and 2) The swallowing phase was imaged at 6 locations, with the ultrasound transducer held at a single location for 6 seconds (30 frames/s, a total of 180 images each). The sagittal scans were located at the left, mid, and right, and the coronal scans at the anterior, mid, and posterior positions.
Tongue analysis was performed by manually outlining the tongue surface using custom image segmentation software [31], and then calculating tongue thickness changes as a function of time within a single image plane. The tongue surface was identified as the bright border at the tongue-air boundary (Figure 2). The outlined borders were then exported to the custom software developed with the MATLAB engineering software package (MathWorks, Natick, MA). Tongue thickness was calculated in millimeters from the base of the ultrasound image to the tongue-air boundary.
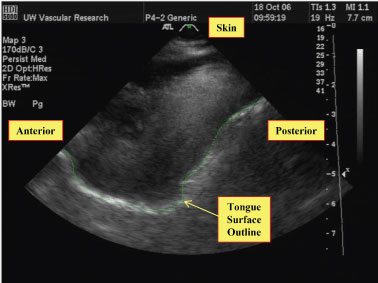 Figure 2: Example of the tongue surface outlining procedure for ultrasound images. The ultrasound transducer is at the top of the image, and the tongue surface contour is manually outlined (green line) on each image of interest using custom software.
View Figure 2
Figure 2: Example of the tongue surface outlining procedure for ultrasound images. The ultrasound transducer is at the top of the image, and the tongue surface contour is manually outlined (green line) on each image of interest using custom software.
View Figure 2
For ultrasound motion analysis, outlining was performed only for the images captured in the middle region of the tongue for two reasons: 1) The middle region generally had the clearest images of the tongue surface; and 2) The chewing side was not instructed to the patient when the gum-chewing was imaged, thus the chewing side was unknown for image analysis. Therefore, the middle region of the tongue is the least confounded position for data analysis because it is not side-dependent.
For chewing and swallowing, every frame within a time period of interest was outlined. This provided a constant time spacing of 33 milliseconds between outlines. The chewing phase was defined as an open and close cycle of the jaw. The jaw motion was derived from the output of a 3D tracker (Flock of Birds, Ascension Technology, Burlington, VT) attached to the ultrasound probe, which moved with the jaw during imaging. Swallowing was defined as starting during a stable period before swallow, and tracing every image until the motion stopped.
Additional custom MATLAB software was used to analyze the tongue motion during chewing and swallowing. Both the sagittal and coronal views were analyzed to generate sequences of tongue surface motion as a function of time. Average maximum thickness and velocity, and the range of motion were derived from this time-motion data. Measurements for the tongue thickness at rest were made from image frames preceding motion associated with chewing or swallowing.
Custom MATLAB software was also used to create correlation maps of tongue motion from these tracing contours (Figure 3). First, tongue thickness as a function of time was calculated for individual spatial segments of the tongue along each traced contour (Figure 3A and Figure 3B). Correlations of these time series were then calculated for each tongue segment relative all other segments (Figure 3C). The correlation coefficients were then compiled into a 2D map for all tongue segments (Figure 3D). The darker the red, the more consistent the motion (positive correlation) between the two regions. Therefore, the diagonal line from 1 to 48 is always the darkest because it represents the correlation for the same tongue segment. In contrast, the darker the blue, the more negative the correlation between the two regions, signifying motion in opposite directions. These correlation maps were used to explore the directional consistency of tongue segments in a time domain approach [32].
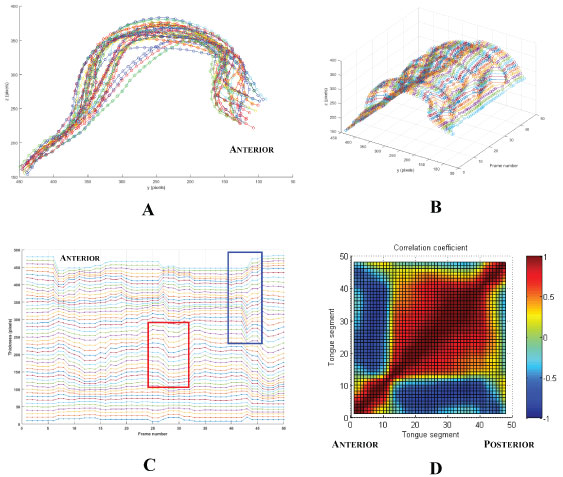 Figure 3: Derivation of regional correlation maps of tongue motion. A) Superimposed tongue surface outlines over time with 48 tongue segments shown (circles); color changes represent contours at different times; B) Plot of tongue thickness showing the path over time of each of the 48 regional segments; C) Tongue thickness as a function of time for each of the 48 segments stacked to show the differences in regional motion. The trace for the anterior segment is at the top of the stack. The red box shows an example of positively correlated motion between segments of the tongue; the blue box shows an example of negative correlation; D) 2D map of the correlation coefficients for each segment relative to all of the following segments. The upper left region is a mirror image of the lower right.
View Figure 3
Figure 3: Derivation of regional correlation maps of tongue motion. A) Superimposed tongue surface outlines over time with 48 tongue segments shown (circles); color changes represent contours at different times; B) Plot of tongue thickness showing the path over time of each of the 48 regional segments; C) Tongue thickness as a function of time for each of the 48 segments stacked to show the differences in regional motion. The trace for the anterior segment is at the top of the stack. The red box shows an example of positively correlated motion between segments of the tongue; the blue box shows an example of negative correlation; D) 2D map of the correlation coefficients for each segment relative to all of the following segments. The upper left region is a mirror image of the lower right.
View Figure 3
By using a popular assessment of articulation error, Pat-3 Photo Articulation Test (the 3rd edition) [33], articulation was first examined and scored by the two speech pathologists who were unfamiliar with the subjects, and the mean score of the two was used. This test used a series of 72 color photographs to assess all American English consonants, vowels, and diphthongs, as well as connected speech. The child identified each photo of an object presented by the examiner who recorded the responses directly onto the summary/record forms. All elicited sounds were arranged by age of acquisition and grouped by the age at which the sounds were correctly articulated by 90% of the normative sample. Scores were reported as standard scores, percentiles, and age-equivalents [33,34].
Two miniature pressure transducers (Konigsberg Instruments, Pasadena, CA) were placed bilaterally on the lower lip opposite to the canines with tissue glue (Sonometrics Co. London, Canada), and two pairs of 10-mm round surface electrodes (NeoTrace Kitty Cat Electrode, Kappa Medical, Prescott, AZ) were adhered bilaterally in the region of the superior orbicularis oris (OO) (Figure 4). Each subject was asked to complete maximal lip closure, involving squeezing the lips together as hard and as long as possible, followed by a period of relaxation (3 cycles); and then fast maximal lip closure, involving squeezing the lips together as hard and as fast as possible, as many times as possible, within 10 seconds (3 trials). Inter-labial pressures and integrated electromyogram (iEMG) of the OO during above efforts were recorded and analyzed using a MP-150 system and Acknowledge software (BioPac Co. Santa Barbara, CA), as previously published [35-37]. Finally, a series of speech tasks were performed. These tasks included three speech intelligibility tests: 1) Syllables, by reading, "pa" and "papapa"; 2) Single-words, by reading "pop" and "poppy"; and 3) Sentences, by reading "see poppy again" which was set in a carrier phrase "I can say ..." [27]. All tasks were repeated 3 times, and speech tasks and co-occurring inter-labial pressures and muscle EMG were recorded simultaneously.
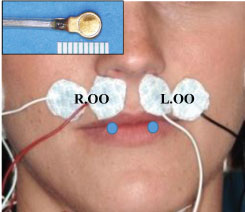 Figure 4: Set-up for inter-labial pressure and muscle EMG. Insert: Pressure transducer. White patches: Surface EMG electrodes for Superior orbicularis oris (OO). Blue circles: Locations of the two pressure transducers.
View Figure 4
Figure 4: Set-up for inter-labial pressure and muscle EMG. Insert: Pressure transducer. White patches: Surface EMG electrodes for Superior orbicularis oris (OO). Blue circles: Locations of the two pressure transducers.
View Figure 4
Since all quantitative data (CBCT, ultrasound, EMG, and lip pressure) are normally distributed, non-paired t-tests were performed to examine the significance between AOBs and controls. Associations between craniofacial skeletal measurements from CBCT and tongue shape/motion measurements from ultrasound were assessed by Spearman correlation coefficients. Due to the small sample size, the level of significance was set as p < 0.10. Because of certain language barriers (non-native English speakers), a few subjects had difficulty completing articulation and/or speech tests. Thus, these data were processed qualitatively without statistical analysis.
No significant differences in linear and angular measures were identified between the two groups. However, AOBs tended to have smaller mandibular body length (Go-Me), ramal height (Cd-Go), and bilateral condyle width (Cdl-Cdr). The gonial angle (Cd-Go-Me) and internal ramal inclination (Cdl-Go-Cdr) tended to be larger in AOBs than controls. The volume of the mandible and the combined volume of oral and pharyngeal airways were significantly smaller in AOBs than controls (p ≤ 0.10, Table 1).
Table 1: Comparison of CBCT measurements. View Table 1
Mandibular volume positively correlated with anterior face height (Na-Me), ramal height (Cd-Go), and bilateral condyle width (Cdl-Cdr). Oral airway volume was positively associated with anterior face height (Na-Me) and anterior lower face height (Pr-Me). Combined volume of oral and pharyngeal airway was positively associated with anterior lower face height (Pr-Me) (Figure 5).
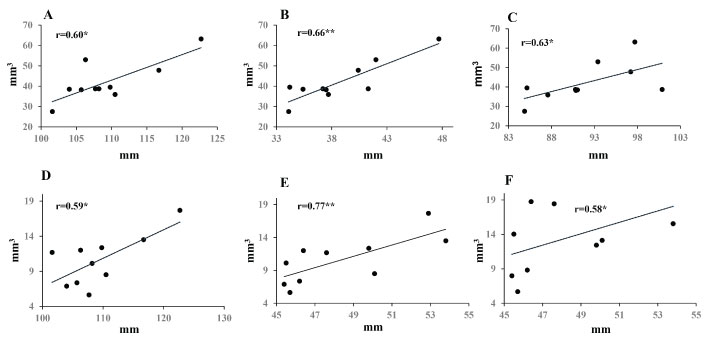 Figure 5: Correlations between craniofacial skeleton and the volumes of mandible and airways. A,B,C) Mandibular volume vs. anterior face height (Na-Me), ramal height (Cd-Go), and bilateral condyle width (Cd-Cd), respectively; D,E) Oral and pharyngeal airway volumes vs. anterior face height (Na-Me) and lower anterior faceheight (Pr-Me), respectively; F) Combined airspace volume vs. lower anterior face height (Pr-Me).
Figure 5: Correlations between craniofacial skeleton and the volumes of mandible and airways. A,B,C) Mandibular volume vs. anterior face height (Na-Me), ramal height (Cd-Go), and bilateral condyle width (Cd-Cd), respectively; D,E) Oral and pharyngeal airway volumes vs. anterior face height (Na-Me) and lower anterior faceheight (Pr-Me), respectively; F) Combined airspace volume vs. lower anterior face height (Pr-Me).
r: Correlation coefficient; *p < 0.10; **p < 0.05.
View Figure 5
Motion display of tongue thickness over time demonstrated more dynamic fluctuations in the mid-tongue dorsal surfaces for both the sagittal and coronal views during the instructed swallowing in AOBs. By contrast, the motion pattern was flatter in both the anterior and posterior regions for the majority of the swallowing images in controls (Figure 6).
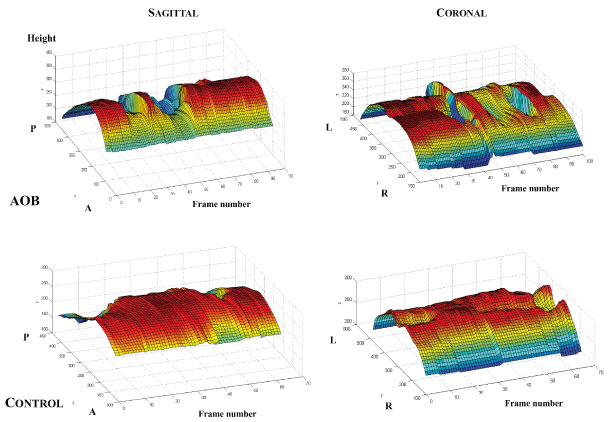 Figure 6: Representative plots of mid-tongue motion patterns during instructed swallowing in AOB (Top) and control (Bottom) subjects for sagittal and coronal imaging planes.
Figure 6: Representative plots of mid-tongue motion patterns during instructed swallowing in AOB (Top) and control (Bottom) subjects for sagittal and coronal imaging planes.
A: Anterior; P: Posterior; L: Left; R: Right.
View Figure 6
The average thickness of the middle portion of the tongue at rest was nearly the same for both AOBs and controls in both sagittal and coronal views (Figure 7A). For both groups, motion range was larger for swallowing than for chewing. AOBs displayed a trend to have a larger range of motion and a faster velocity compared to controls, in particular during swallowing (Figure 7B and Figure 7C).
 Figure 7: A) Comparisons of average thickness of mid-tongue at rest in AOB and control subjects; B) Comparisons of mid-tongue motion range during swallowing in AOB and control subjects; C) Comparisons of mid-tongue motion velocity during swallowing in AOB and control subjects.
View Figure 7
Figure 7: A) Comparisons of average thickness of mid-tongue at rest in AOB and control subjects; B) Comparisons of mid-tongue motion range during swallowing in AOB and control subjects; C) Comparisons of mid-tongue motion velocity during swallowing in AOB and control subjects.
View Figure 7
Correlation results for the subjects with the highest degrees of AOB and overbite of controls are shown in Figure 8. In the sagittal view, the control demonstrated a relatively clear pivot point in both chewing and swallowing where the anterior and posterior segments are inversely correlated, representing a change in direction of motion (Figure 8B). The AOB shows an extreme anterior pivot point in chewing, but a tendency toward bulk motion of the tongue in swallowing (Figure 8A). In the coronal view, the tongue motion from left to right is consistent for both the AOB and control while chewing, demonstrating bulk motion with minimal bending (Figure 8C and Figure 8D). However, while swallowing, the control demonstrates a well-defined bending pattern, with the center of the tongue moving in the opposite direction compared to the left and right sides. In addition, the left and right sides are strongly correlated with each other, showing a coordinated motion of the two sides. The sharp correlation transitions seen in the control, signifying clear bending points, were absent in the AOB. In general, there appears to be finer regional motions of the tongue for the control compared with the AOB.
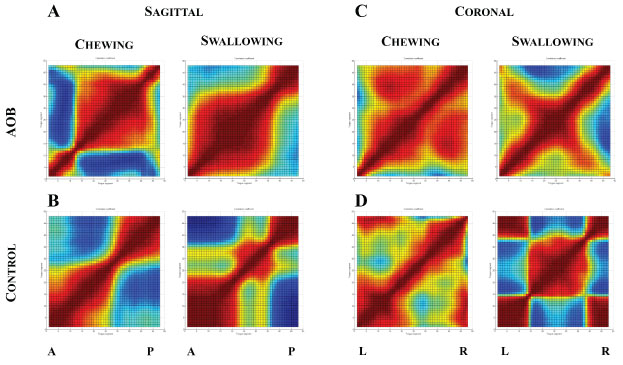 Figure 8: Regional correlation maps of tongue motion for the AOB subject with maximum open bite (Top) and the control subject with maximum overbite (Bottom). Results are shown for sagittal and coronal views for both chewing and swallowing.
Figure 8: Regional correlation maps of tongue motion for the AOB subject with maximum open bite (Top) and the control subject with maximum overbite (Bottom). Results are shown for sagittal and coronal views for both chewing and swallowing.
A: Anterior; P: Posterior; L: Left; R: Right.
View Figure 8
As shown in Table 2, the thickness of the mid-tongue in sagittal view during chewing was positively associated with the heights of anterior total (Na-Me) and lower (Pr-Me) faces and mandibular ramus (Cd-Go), the width of bilateral mandibular angles (Go-Go), the lengths of total mandible (Cd-Pog) and mandibular body (Me-Go), antero-posterior relationship of jaws (ANB), and upper airway volume (CA). The thickness of mid tongue during swallowing was positively associated with the gonial angle (Cd-Go-Me) in sagittal view, and with internal ramal inclination (Cdl-Go-Gdr) in coronal view. At rest, the thickness was associated with bilateral condyle width (Cdl-Cdr) in sagittal view and mandibular body length (Me-Go) in coronal view.
Table 2: Correlations between tongue features and skeletal/airway measures. View Table 2
The ranges of tongue motion during chewing were positively associated with bilateral condylar width (Cdl-Cdr) in both sagittal and coronal views, but negatively associated with internal ramal inclination (Cdl-Go-Cdr) in coronal view. During swallowing, the range of motion was negatively associated with the width of middle face (Mxl-Mxr) in sagittal view and with heights of middle and anterior lower face (Mx-M and ANS-Me) in coronal view.
During chewing, maximal motion velocities were positively associated with the width of mandibular angles (Gol-Gor) and internal ramal inclination (Cdl-Go-Cdr) in both the sagittal and coronal views. During swallowing, maximum velocity was positively associated with the gonial angle (Cd-Go-Me) in sagittal view and with internal ramal inclination (Cdl-Go-Gor) in coronal view.
The average raw score from AOBs was 1.2, as compared with 0.40 from controls. All AOBs scored one or more, but 3 out of 5 controls scored 0. The other variables from the test, such as age equivalent and standard scores, were not available due to uncompleted tasks by 2 AOBs and controls. Nevertheless, the higher raw score implies the possibility of an articulation deficiency in AOBs.
While there was no statistical difference of inter-labial pressure during fast and maximal lip closure between the two groups, AOBs presented a significantly lower iEMG of OOs during the effort of maximal lip closure (Table 3).
Table 3: iEMG and pressures of fast and maximal lip closures. View Table 3
The speech intelligibility tests identified the following characteristics of the speech function in AOBs as compared with controls: 1) Syllables: More activity of lip (OOs) and jaw opening (digastric, DAs) muscles, and more isolated efforts of sound production with an extended duration (Figure 9A); 2) Single-words: More extended activity of the muscles, extended duration of speech effort, and unseparated syllables, such as "Pop" (Figure 9B); 3) Sentences: Extended and isolated effort for each word, with less corresponding muscle activity (Figure 9C). These features indicate that AOB subjects presented a certain degree of speech disorder.
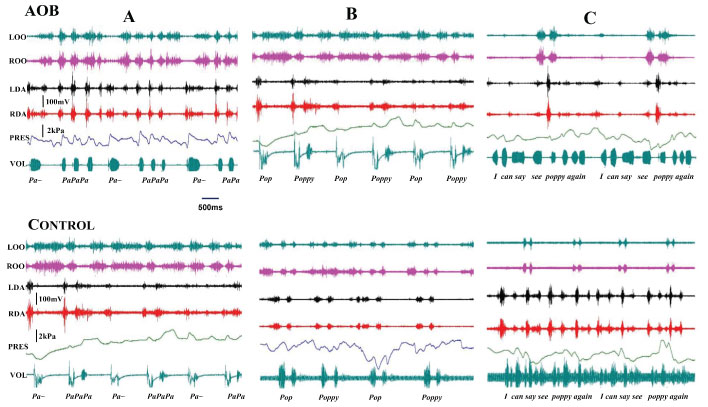 Figure 9: : Raw tracing of speech intelligibility tests. A) Syllables; B) Single-word; C) Sentence.
Figure 9: : Raw tracing of speech intelligibility tests. A) Syllables; B) Single-word; C) Sentence.
LOO and ROO: Left and right orbicularis oris; LDA and RDA: Left and right anterior digastric; PRES: Inter-labial pressure; VOL: Voice recording.
View Figure 9
Although the present study is comprehensive and involves in understudied areas concerning AOB, several limitations might have prevented us from reaching the definitive results. These limitations include: 1) The sample sizes is relatively small, and the controls are not normal (with malocclusion but no AOB); 2) Some possible confounding factors were not excluded or controlled, such as Angle Classification and environmental factors; 3) No East African norms and standard scores are available to use for comparing all measurements. Nevertheless, the present study does produce enlightening findings and is a foundation for future research on AOB in a large East African population.
The common skeletal characteristics associated with AOB include negative overbite, augmented anterior face height, steep mandibular plane, larger gonial angle, increased inter-basal angle, and proclined incisors [10,38-40]. Racial variations of skeletal features exist in the presentation of AOB [11]. Considerable evidence indicates that AOB has higher prevalence and severity in the black population [12]. However, there are only three published studies which describe and characterize the skeletal and dental features of AOB in the North American black population [12,28,41]. These findings generally coincided with those for white subjects. However, particularly longer lower facial heights and smaller mandibular plane angles were seen in AOB in the black population, as compared to their white counterparts [41].
As a potential etiology for AOB, airway obstruction, such as adenoid hyperplasia or anatomic blockages, may result in mouth breathing with a more backward head and a lower tongue position [42-44]. Mouth breathing is closely associated with a more hyper-divergent growth pattern, dolichol-facial-type face and open bite [3,43-45].
In this study, CBCT were used to characterize dental/skeletal patterns and upper airway because of its high accuracy and the capacity to demonstrate 3D anatomic structures [46,47]. In addition, more parameters, such as volumes and angles in different planes, can be measured in CBCT.
The present study did not find unique skeletal characteristics in AOBs, as reported in previous cephalometric study [28]. However, the mandibular length and ramal height, and the width of bilateral condyles were smaller, although the difference was not statistically significant. In addition, AOBs showed significantly smaller mandible than that of controls. These differences may suggest a smaller mandible in AOB, which could have led to a more Class II skeletal type. Clinically, Class II patients with smaller mandibles have higher risk for AOB [44]. AOBs also tended to have slightly larger gonial and internal inclination angles, indicating a downward and backward rotation of the mandible.
Greater vertical facial height is associated with AOB in some reports [2,10,48]. However, the values of vertical facial height measured in this study, including total anterior face height, anterior face height, anterior lower face height and middle face height, were similar between AOBs and controls. Given the fact that all subjects were 9-12 year-old East Africans in this study, greater differences might be found with older patients who have completed their pubertal growth spurt. The results also indicate that oral airway volume and combined volume of oral and pharyngeal airway spaces were significantly correlated with total anterior face height and/or anterior lower face height. Therefore, it is reasonable to assume that vertical dimension of the facial skeleton and upper airway volume are positively associated.
Tongue habits and abnormal tongue movement have been associated with AOB, but the cause-effect relationship between the tongue and AOB remain controversial [20,49]. It is also unclear whether the differences exist in various ethnic groups. The present study did not show a difference in middle tongue thickness at rest between AOBs and controls. However, AOBs exhibited a greater range and velocity of motion and a larger area of consistent movement of tongue segments, especially during swallowing. The later indicates that there may be less regional coordination or fine motor control of functional motion of the tongue in AOBs. In addition, the correlation mapping methods details the tongue motion pattern in function and is an area of future research.
The present study also revealed that tongue thickness and motion during chewing and swallowing were associated with skeletal features to a certain extent. For example, mandible size may influence the changes of tongue thickness during chewing because the tongue thickness was positively associated with mandibular body length, mandibular length, and mid-width of mandible both in sagittal and coronal views. The tongue thickness and maximal velocity during swallowing may be related to the inclination of the mandible because an increased gonial angle was significantly related to a greater tongue thickness in sagittal view. In the coronal view, the tongue thickness and maximal velocity during swallowing were also positively associated with the internal ramal inclination. The motion velocity during chewing showed associations with some features of the mandible, including its mid-width internal ramal inclination. These results further indicate that the tongue thickness at rest and during chewing, as well as the chewing motion range, were mainly related to the dimensions of mandible, while the swallowing motion was more depended upon the facial height, in particular in coronal view.
The differences in functional motions of the tongue between the two groups and their close relationships with craniofacial skeletal patterns imply that AOB may be associated with these unusual tongue features. Since the tongue exerts short- or long-term forces on the surrounding dentition, these forces could result in the development of an AOB [49,50]. A number of authors have noted an association between the development of AOB and a tongue thrust swallowing pattern [20,51-53]. Another explanation for the difference in tongue functional motion between the two groups is that the tongue might modified to adapt the dental and craniofacial features, in turn modulate the neuromuscular system to meet functional demands, such as tongue thrusting.
Malocclusion can lead to unsuccessful speech function due to morphological abnormalities, and AOB is one malocclusion that can be highly associated with articulatory speech disorders [54-59].
AOB subjects have some characteristic anatomic variations as discussed above, which may modify the interplay of the tongue with the lips, teeth, palate, rugae, and oropharynx, thus compromising articulation. Aberrant tongue function is related with speech dysfunction, and the relationship between AOB and abnormal swallowing/sigmatisms was reported as high as 75% [55,56,60,61].
Lips are also of great importance in speech function. Increased vertical height, proclination of incisors, and incompetent lips are common in AOB patients, which make it very difficult for patients to close the lips and to achieve normal forms of bilabial consonants. Thus, lower lip pressure is most likely to be seen in AOB patients, which may lead to phonation alterations. Further, weak lip pressure on anterior teeth allows a greater vestibular inclination, in turn exacerbating AOB. However, some hold the opinion that the association between AOB and functional speech disorders is not clear [62]. Lopez-Perez, et al. found no association between speech disorders and AOB in Mexican children with Down syndrome [63].
Little information is available about real-time lip pressure and articulation condition in AOB. Even though weaker inter-labial pressures during lip closure were not identified in the present study, the lip muscle (OO) did show significantly lower activity, indicating a deficiency of lip function. The articulation examination in this study also supports the hypothesis that AOB is more likely to have disordered articulation.
However, the relation between AOB and articulatory deficits is not direct. In the speech intelligibility tests, most AOBs were able to pronounce normally and produce fully comprehensive sentences, although there were more unseparated syllables as compared with controls. The EMG result further showed more isolated effort and extended duration of lip and jaw-opening muscle activity during sound production in AOBs. These features indicate that AOBs might be able to compensate for their occlusal defects and achieve acceptable pronunciation through more effort of articulatory components - lip and jaw-opening muscles. It has been demonstrated that the more severe or handicapping the malocclusion, the more likely a speech sound error occurs [64]. Therefore, more subjects with different degrees of AOB are needed for future research on the influences of AOB on articulation and speech function.
The results of the present study suggest that craniofacial skeletal patterns of East Africans children with and without AOB are similar, but AOBs have smaller volumes of mandible and upper airway space. The tongue moves more and faster during swallowing in AOBs, and tongue thickness during chewing is more related to skeletal variables than other tongue functional features (motion range and velocity). AOBs may present weak lip muscle activity during lip closure, deficiency of articulation, enhanced activity of lip and jaw-opening muscles during speech, and speech disorders.
The authors would like to thank Drs. Volodymyr Shcherbatyy, Fereshteh Yousefzadeh, Inna Piskorska, and Ilona Khosh for their help with data collection and analysis, and Dr. Christopher A. Moore and his laboratory in Speech and Hearing Sciences at the University of Washington for their help with evaluation of articulation and speech. Special thank goes to Dr. Gregory J. King for his thoughtful vision on the initiation of this project.
The authors declare that they have no competing interests.
This study was supported by the Royalty Research Fund of the University of Washington to Dr. Zi-Jun Liu. The participation of Drs. Meng-Zhao Deng and Zhi-He Zhao was supported by the VISER program (International Visiting Student Engaged in Research) of the University of Washington, USA, and West China Hospital of Stomatology, Sichuan University, China.